Share This Page
Drug Price Trends for PIRFENIDONE
✉ Email this page to a colleague
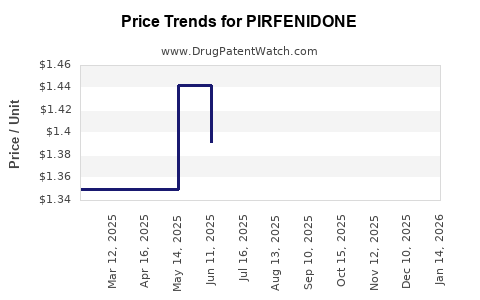
Average Pharmacy Cost for PIRFENIDONE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| PIRFENIDONE 267 MG TABLET | 00904-7397-53 | 1.33365 | EACH | 2026-04-22 |
| PIRFENIDONE 267 MG TABLET | 00480-3610-87 | 1.33365 | EACH | 2026-04-22 |
| PIRFENIDONE 267 MG TABLET | 00781-8085-32 | 1.33365 | EACH | 2026-04-22 |
| PIRFENIDONE 801 MG TABLET | 76282-0716-90 | 3.18104 | EACH | 2026-04-22 |
| PIRFENIDONE 267 MG TABLET | 31722-0872-27 | 1.33365 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for PIRFENIDONE
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| ESBRIET 801MG TAB | Genentech USA, Inc. | 50242-0123-01 | 90 | 10291.90 | 114.35444 | EACH | 2024-01-01 - 2028-09-14 | FSS |
| ESBRIET 267MG TAB,ORAL | Genentech USA, Inc. | 50242-0122-06 | 270 | 10291.90 | 38.11815 | EACH | 2023-09-15 - 2028-09-14 | FSS |
| ESBRIET 267MG TAB,ORAL | Genentech USA, Inc. | 50242-0122-06 | 270 | 7871.98 | 29.15548 | EACH | 2024-01-01 - 2028-09-14 | Big4 |
| ESBRIET | Genentech USA, Inc. | 50242-0121-01 | 270 | 7833.55 | 29.01315 | EACH | 2023-09-15 - 2028-09-14 | Big4 |
| ESBRIET 267MG TAB,ORAL | Genentech USA, Inc. | 50242-0122-06 | 270 | 10291.90 | 38.11815 | EACH | 2024-01-01 - 2028-09-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Pirfenidone Market Analysis and Price Projections
Pirfenidone, a small molecule with antifibrotic and anti-inflammatory properties, is primarily approved for the treatment of idiopathic pulmonary fibrosis (IPF). The global pirfenidone market is projected to experience moderate growth driven by an increasing prevalence of IPF, rising healthcare expenditures, and the drug's established efficacy. However, the market faces challenges including patent expirations and the emergence of generic competition, which are expected to exert downward pressure on pricing.
What is the current global market size and projected growth rate for pirfenidone?
The global pirfenidone market was valued at approximately $1.2 billion in 2023. Projections indicate a compound annual growth rate (CAGR) of 4.5% to 5.5% over the next five to seven years, reaching an estimated $1.6 billion to $1.8 billion by 2030. This growth is primarily attributed to the expanding diagnosis of IPF, particularly in developed economies, and the sustained demand for effective treatment options. Asia-Pacific is expected to be the fastest-growing region due to increasing healthcare infrastructure and awareness.
What are the key drivers and restraints impacting the pirfenidone market?
Market Drivers:
- Increasing Prevalence of Idiopathic Pulmonary Fibrosis (IPF): IPF is a chronic, progressive lung disease with a rising incidence and prevalence globally. Estimates suggest that IPF affects approximately 30 to 100 individuals per 100,000 people worldwide, with higher rates in North America and Europe. This growing patient population directly fuels demand for pirfenidone.
- Established Efficacy and Safety Profile: Pirfenidone has demonstrated clinical benefits in slowing disease progression and improving lung function in IPF patients. Clinical trials, such as the ASCEND and CAPACITY studies, have shown that pirfenidone reduces the rate of decline in forced vital capacity (FVC). Its safety profile, while not without side effects, is generally considered manageable for many patients.
- Growing Healthcare Expenditure and Access: Increased investment in healthcare systems and improved access to advanced medical treatments in emerging economies contribute to market expansion. As more patients gain access to diagnostic tools and treatment options, the demand for pirfenidone rises.
- New Drug Formulations and Delivery Methods: Development of alternative pirfenidone formulations, such as orally disintegrating tablets (ODTs), aims to improve patient adherence and convenience, potentially driving market adoption.
Market Restraints:
- Patent Expirations and Generic Competition: The expiration of key patents for originator pirfenidone products has opened the door for generic manufacturers. This has led to increased market fragmentation and significant price erosion as generics enter the market. For instance, the primary patent for Esbriet (pirfenidone) in the U.S. expired in 2020, leading to the introduction of several generic versions.
- High Cost of Treatment: Despite generic availability, the overall cost of pirfenidone therapy remains substantial, posing a financial burden for patients and healthcare systems. This can limit uptake in price-sensitive markets.
- Side Effects and Patient Adherence: Common side effects of pirfenidone, including gastrointestinal issues (nausea, diarrhea), fatigue, and photosensitivity, can impact patient adherence and necessitate dose adjustments or discontinuation.
- Emergence of Novel Therapies: Ongoing research and development in antifibrotic therapies, including new drug candidates and combination approaches, may introduce alternative treatment options that could compete with pirfenidone.
What is the current pricing landscape for pirfenidone and what are the projected price trends?
The pricing of pirfenidone varies significantly between branded and generic versions, as well as by region.
Branded Pirfenidone (e.g., Esbriet):
- Prior to generic entry, the annual cost for branded pirfenidone in the U.S. could range from $75,000 to over $90,000 per patient, based on standard dosing regimens and without considering patient assistance programs.
- In Europe, pricing also varied by country, but annual costs were generally in a similar range, often subject to national health system negotiations and formularies.
Generic Pirfenidone:
- Following patent expirations, the price of generic pirfenidone has decreased substantially. In the U.S. market, the price of generic pirfenidone has fallen by an estimated 50-70% compared to the branded product.
- A 28-day supply of generic pirfenidone can now be found for prices ranging from $3,000 to $6,000, depending on the manufacturer, dosage, and pharmacy.
- This price reduction is a direct consequence of increased competition from multiple generic manufacturers.
Projected Price Trends:
- Downward Pressure: The trend for pirfenidone pricing is expected to continue downward in the medium term due to ongoing generic competition. As more generic manufacturers establish market presence and production scales, competitive pricing will likely intensify.
- Regional Variations: Price disparities will persist across regions, influenced by local regulatory environments, market dynamics, and the presence of authorized generics or biosimil-like competition.
- Stabilization Potential: While significant price declines have occurred, the rate of decrease may stabilize as the market reaches a competitive equilibrium. However, prices are unlikely to rebound to pre-generic levels.
- Impact of New Formulations: New, potentially patented, formulations of pirfenidone (e.g., ODTs) may command premium pricing initially, but their long-term price trajectory will also be influenced by generic competition for these novel versions.
Who are the key players in the pirfenidone market?
The pirfenidone market comprises originator companies, generic manufacturers, and contract manufacturing organizations (CMOs).
Originator and Major Branded Players:
- Roche (Genentech): Owns the original brand, Esbriet. Roche acquired InterMune, which developed pirfenidone.
- Prometheus Laboratories: Had a role in the early development and commercialization in specific territories.
Key Generic Manufacturers: The landscape of generic pirfenidone manufacturers is dynamic and includes:
- Cipla Ltd.
- Sun Pharmaceutical Industries Ltd.
- Dr. Reddy's Laboratories
- Lupin Ltd.
- Torrent Pharmaceuticals Ltd.
- Teva Pharmaceutical Industries Ltd.
- Viatris Inc.
- Aurobindo Pharma
- Cadila Healthcare (Zydus Cadila)
Contract Manufacturing Organizations (CMOs): A number of CMOs are involved in the manufacturing of Active Pharmaceutical Ingredients (APIs) and finished dosage forms for various pirfenidone brands, both branded and generic. Specific CMO names are often proprietary or subject to confidential agreements but represent a crucial segment of the supply chain.
What is the patent landscape and its impact on market exclusivity for pirfenidone?
The patent landscape for pirfenidone is characterized by expiring composition of matter patents and the ongoing pursuit of new patents for formulations, manufacturing processes, and new indications.
- Composition of Matter Patents: The primary patents covering the pirfenidone molecule itself have largely expired in major markets such as the U.S. (around 2020) and Europe (varying dates, generally expiring in the early 2020s). These expirations were the critical trigger for generic entry.
- Formulation Patents: Companies have sought and obtained patents for improved formulations, such as orally disintegrating tablets (ODTs), which offer benefits in terms of ease of administration and potentially reduced gastrointestinal side effects. These patents can extend market exclusivity for specific product types. For example, patents related to ODT formulations may extend protection into the mid-to-late 2020s.
- Manufacturing Process Patents: Proprietary methods for synthesizing pirfenidone or its intermediates can also be patented, potentially creating barriers for generic competitors who must develop non-infringing processes.
- Polymorph Patents: Patents related to specific crystalline forms (polymorphs) of pirfenidone can also offer a degree of protection.
- New Indication Patents: While pirfenidone is primarily approved for IPF, ongoing research into its efficacy for other fibrotic diseases (e.g., liver fibrosis, kidney fibrosis, systemic sclerosis) could lead to new patents if successful. However, the regulatory hurdles and development timelines for new indications are substantial.
The impact of this patent landscape is direct:
- Generic Entry: The expiration of composition of matter patents has led to a flood of generic pirfenidone products, significantly increasing competition and driving down prices.
- Extended Exclusivity: Formulation and process patents can allow originator companies or new entrants to maintain some market exclusivity for improved versions of the drug, albeit for a limited period and often at a lower premium than initial branded exclusivity.
- Legal Challenges: The patent landscape can also be a source of litigation, as generic companies challenge existing patents or originator companies defend their intellectual property.
What are the regulatory considerations for pirfenidone in key global markets?
Regulatory considerations for pirfenidone involve approvals for its use, pharmacovigilance, and market access policies.
- United States (FDA):
- Approval: Pirfenidone (Esbriet) was first approved by the U.S. Food and Drug Administration (FDA) in 2014 for the treatment of IPF.
- Generic Approvals: The FDA has approved multiple abbreviated new drug applications (ANDAs) for generic versions of pirfenidone.
- Labeling and Post-Market Surveillance: Generic products must demonstrate bioequivalence to the reference listed drug and adhere to the same labeling requirements. Post-market surveillance continues to monitor safety.
- European Union (EMA):
- Approval: Pirfenidone (Esbriet) received a marketing authorization from the European Medicines Agency (EMA) in 2017.
- National Authorizations: Approval by the EMA leads to a centralized procedure, but market access and pricing are subsequently determined at the national level by individual member states' health authorities and reimbursement bodies.
- Generic Approvals: Similar to the U.S., generic pirfenidone products have been authorized in the EU following patent expiries.
- Japan (PMDA):
- Approval: Pirfenidone (Esbriet, marketed as Pirespa) was approved in Japan in 2008, making it one of the earlier markets for the drug.
- Pricing and Reimbursement: Japan's healthcare system involves strict price negotiations and reimbursement reviews.
- Other Key Markets (e.g., Canada, Australia, China, India):
- Regulatory Pathways: Each country has its own regulatory agency (e.g., Health Canada, TGA in Australia, NMPA in China) with specific requirements for drug approval, including efficacy and safety data.
- Generic Market Entry: The timing of generic entry and the associated price reductions are influenced by local patent laws and regulatory processes.
- Market Access: Reimbursement policies, formulary inclusion, and pricing regulations vary significantly, impacting the accessibility and cost-effectiveness of pirfenidone in these regions.
What is the competitive landscape and outlook for pirfenidone?
The competitive landscape for pirfenidone has fundamentally shifted from a monopolistic market dominated by a single branded product to a highly competitive generic market.
- Originator vs. Generics: The primary competition is now between originator brands (primarily Esbriet) and a wide array of generic pirfenidone manufacturers. Originator brands are focusing on maintaining market share through brand loyalty, patient support programs, and potentially differentiating new formulations.
- Price Wars: Generic manufacturers are engaged in price competition to capture market share. This has led to substantial price erosion, making pirfenidone more accessible but reducing overall revenue for the drug class.
- Market Penetration: The increased affordability of generic pirfenidone is expected to improve market penetration, particularly in emerging markets and among patient populations with limited insurance coverage.
- Pipeline Competition: While pirfenidone is established, the development of next-generation antifibrotic agents or therapies targeting different mechanisms of fibrosis could pose future competition. Drugs targeting specific pathways involved in fibrosis are in various stages of clinical development. Examples include nintedanib (another approved IPF drug) and investigational agents.
- Combination Therapies: Research into the potential benefits of combining pirfenidone with other antifibrotic agents or therapies is ongoing. Successful combination strategies could create new market segments or alter the competitive dynamics.
The outlook for pirfenidone is one of sustained demand for IPF treatment but with significantly diminished revenue potential per unit due to genericization. The market's growth will be driven by volume rather than price increases.
What are the key therapeutic areas and indications for pirfenidone?
The primary and currently approved therapeutic area for pirfenidone is Idiopathic Pulmonary Fibrosis (IPF).
- Idiopathic Pulmonary Fibrosis (IPF): This is a chronic, progressive, and irreversible lung disease characterized by scarring (fibrosis) of the lung tissue. Pirfenidone is approved to slow the rate of disease decline in patients with IPF. It has demonstrated efficacy in reducing the annual rate of decline in forced vital capacity (FVC).
Investigational or Emerging Indications: Research is exploring the potential efficacy of pirfenidone in other fibrotic conditions. These are generally considered investigational and not yet approved indications:
- Systemic Sclerosis-associated Interstitial Lung Disease (SSc-ILD): Clinical trials have investigated pirfenidone for this condition, though its approval and market penetration in this area are less established than for IPF.
- Other Fibrotic Diseases: Preclinical and early-stage clinical research has examined pirfenidone's potential role in other fibrotic disorders, including:
- Liver fibrosis (e.g., non-alcoholic steatohepatitis - NASH)
- Kidney fibrosis
- Pulmonary fibrosis not related to IPF (e.g., connective tissue disease-associated ILD)
Currently, the significant market value and volume for pirfenidone are overwhelmingly tied to its approved use in IPF. Any expansion into new indications would represent a significant future growth opportunity but involves substantial clinical development and regulatory hurdles.
Key Takeaways
- The global pirfenidone market is projected to grow at a CAGR of 4.5-5.5% to reach $1.6-$1.8 billion by 2030, driven by increasing IPF prevalence.
- Patent expirations have led to significant generic competition, causing a 50-70% price reduction compared to branded products.
- The market is now characterized by price competition among multiple generic manufacturers, with downward pricing pressure expected to continue.
- Roche (Genentech) remains the key originator player, while numerous companies, including Cipla, Sun Pharma, and Dr. Reddy's, are major generic suppliers.
- Regulatory approvals for pirfenidone and its generics are well-established in major markets like the U.S., EU, and Japan, with varying national reimbursement policies influencing market access.
- While pirfenidone is primarily approved for IPF, research is ongoing for other fibrotic diseases, representing potential future expansion areas.
Frequently Asked Questions
1. What is the primary mechanism of action for pirfenidone?
Pirfenidone is believed to exert its antifibrotic and anti-inflammatory effects through multiple pathways. It has been shown to inhibit the synthesis of pro-fibrotic mediators such as transforming growth factor-beta (TGF-β) and tumor necrosis factor-alpha (TNF-α). It also reduces the production of extracellular matrix components, including collagen.
2. How does pirfenidone compare to other IPF treatments like nintedanib?
Pirfenidone and nintedanib are both approved first-line treatments for IPF. Clinical trial data suggests comparable efficacy in slowing disease progression, with both drugs reducing the rate of decline in FVC. However, they differ in their mechanisms of action and side effect profiles. Pirfenidone is primarily associated with gastrointestinal issues and photosensitivity, while nintedanib commonly causes diarrhea and nausea. Treatment choice is often individualized based on patient tolerance and physician preference.
3. Can pirfenidone be used in pediatric IPF patients?
Currently, pirfenidone is not approved for use in pediatric populations. Clinical trials and regulatory approvals have focused on adult patients with IPF. Safety and efficacy data in children are limited, and its use in this age group is generally considered off-label and requires careful consideration of risks and benefits.
4. What are the typical side effects associated with pirfenidone therapy?
The most common side effects of pirfenidone include gastrointestinal disturbances such as nausea, diarrhea, and abdominal pain. Other frequently reported side effects are fatigue, headache, decreased appetite, and photosensitivity (increased sensitivity to sunlight), which necessitates sun protection. Less common but potentially serious side effects can occur, and patients are closely monitored by their healthcare providers.
5. How is the pricing of pirfenidone expected to evolve in the next five years, considering generic competition?
The pricing of pirfenidone is expected to continue its downward trend over the next five years, driven by intensified competition among generic manufacturers. While significant price drops have already occurred, further erosion is anticipated as market saturation increases and companies vie for market share. Pricing will likely stabilize at a considerably lower level than that of the original branded product, with regional variations persisting due to differing market access and reimbursement policies.
Citations
[1] Global IPF Treatment Market 2023-2030. (n.d.). Grand View Research. [2] U.S. FDA Approves First Generic Versions of Pirfenidone Capsules. (2020, March 31). U.S. Food & Drug Administration. [3] Pirfenidone Market Size & Share Analysis - Growth Trends & Forecasts. (n.d.). Mordor Intelligence. [4] Drug Price Information for Pirfenidone. (n.d.). GoodRx. [5] Esbriet (pirfenidone) Prescribing Information. (2023). Genentech, Inc. [6] Global Pirfenidone Market Trends and Opportunities. (n.d.). Market Research Future. [7] European Medicines Agency (EMA). (n.d.). Various assessment reports and product information for pirfenidone. [8] Pharmaceuticals and Medical Devices Agency (PMDA). (n.d.). Approval information for pirfenidone (Pirespa).
More… ↓
