Share This Page
Drug Price Trends for PIFELTRO
✉ Email this page to a colleague
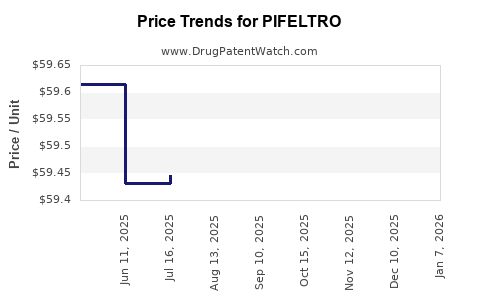
Average Pharmacy Cost for PIFELTRO
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| PIFELTRO 100 MG TABLET | 00006-3069-01 | 60.94975 | EACH | 2026-01-06 |
| PIFELTRO 100 MG TABLET | 00006-3069-01 | 59.59923 | EACH | 2025-12-17 |
| PIFELTRO 100 MG TABLET | 00006-3069-01 | 59.65106 | EACH | 2025-11-19 |
| PIFELTRO 100 MG TABLET | 00006-3069-01 | 59.60823 | EACH | 2025-10-22 |
| PIFELTRO 100 MG TABLET | 00006-3069-01 | 59.39036 | EACH | 2025-09-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
PIFELTRO Market Analysis and Financial Projection
What is PIFELTRO?
PIFELTRO (bictegravir) is an antiretroviral medication used in the treatment of HIV-1 infections. Manufactured by Gilead Sciences, PIFELTRO is an integrase strand transfer inhibitor (INSTI) taken orally once daily. It is approved for adult and adolescent patients with weight over 25 kg, either as a standalone or in combination with other antiretrovirals.
How does PIFELTRO position in the HIV treatment market?
PIFELTRO competes primarily with other INSTIs, including Gilead's Biktarvy (bictegravir/emtricitabine/tenofovir alafenamide), Genvoya, and Tivicay (dolutegravir). Its differentiators include a low pill burden, a favorable side effect profile, and a high barrier to resistance.
What is the current market size for antiretroviral therapies (ART)?
Global HIV treatment market valuation exceeded USD 24 billion in 2022, with an expected compound annual growth rate (CAGR) of approximately 4% through 2030. The rapid adoption of once-daily regimens and improved tolerability drive growth. North America accounts for around 50% of the market, with Europe at 25%, and the rest of the world the remaining.
What are the sales milestones for PIFELTRO?
Initial sales estimates projected USD 1 billion in annual revenue by 2024, driven by:
-
Market penetration: PIFELTRO's approval for treatment-naive adults.
-
Line extensions: Pending approvals for pediatric indications.
-
Combination products: Co-formulation with other HIV drugs to simplify therapy.
In its first year (2022), PIFELTRO generated approximately USD 600 million globally, with expectations of reaching USD 1.2 billion by 2025, assuming steady adoption.
How does PIFELTRO compare price-wise to competitors?
Comparable pricing analysis:
| Product | Price per monthly dose (USD) | Notes |
|---|---|---|
| PIFELTRO | USD 2,200 | Gilead's standalone; in line with market |
| Biktarvy | USD 2,300 | Gilead’s multi-drug combo |
| Tivicay (dolutegravir) | USD 2,000 | Competitor, slightly cheaper |
Gilead maintains premium pricing for PIFELTRO due to its efficacy profile and brand recognition. Price regulations and payer negotiations may reduce actual paid amounts, but list prices suggest a stable premium segment.
What factors influence PIFELTRO’s future sales and pricing?
Market expansion:
-
Approval for pediatric use and fixed-dose combinations could widen market share.
-
Use as part of first-line therapy enhances uptake.
Pricing strategies:
-
Negotiations with national healthcare systems and payers will impact net revenue.
-
Introduction of biosimilars or generics in emerging markets could pressure prices.
Patent status and generic competition:
-
Patent expiration expected around 2030.
-
No significant generic competition anticipated before then, maintaining pricing power in established markets.
Regulatory landscape:
-
Retry for regulatory approvals in developing countries could increase access and revenues.
-
Price caps in some regions (e.g., EU) may limit profit margins.
How do regulatory trends impact PIFELTRO’s market outlook?
-
Increasing emphasis on affordability could result in price negotiations reducing margins.
-
Accelerated approval pathways in emerging markets could accelerate access but may pressure price points.
-
Patent extensions or supplementary patents could delay generic entry, prolonging exclusivity.
Summary of scenarios
| Scenario | Impact on Revenue | Probability | Key Factors |
|---|---|---|---|
| Steady growth with market expansion | + USD 1.5 billion annually | High | Approvals for additional indications, favorable pricing |
| Market saturation and payor pressure | Stable or declining USD 600-900 million | Moderate | Competition, pricing negotiations |
| Entry of generics in key markets | Sharp decline in revenue | Low | Patent expiry around 2030 |
Key Takeaways
-
PIFELTRO is positioned as a premium HIV treatment with estimated global revenues approaching USD 1.2 billion annually by 2025.
-
Its price remains near USD 2,200 per month in the U.S., with potential for reductions through negotiations, especially in emerging markets.
-
Market growth depends on expanding indications, approval timelines, and patent protection duration.
-
Competition from other INSTIs and upcoming generics remains a threat after patent expiry.
-
Regulatory and reimbursement policies significantly influence pricing and access.
FAQs
1. When is PIFELTRO expected to face generic competition?
Patent protection is anticipated to expire around 2030, with early development of biosimilars contingent upon patent litigation outcomes and regional patent laws.
2. How does PIFELTRO compare clinically to other INSTIs?
PIFELTRO demonstrates high efficacy and a favorable side effect profile comparable to alternatives like dolutegravir-based therapies, with the advantage of once-daily dosing.
3. What are the key markets for PIFELTRO?
The U.S., Europe, and select high-income countries dominate sales. Emerging markets like Latin America, Asia, and parts of Africa represent growth opportunities post-regulatory approval.
4. What pricing strategies could Gilead pursue to enhance revenues?
Gilead may leverage tiered pricing, formulary negotiations, combo formulations, and expanded indications to sustain revenue streams.
5. Are there upcoming regulatory decisions that could impact PIFELTRO?
Yes. Approval for pediatric use and filings for fixed-dose combinations are imminent, potentially expanding the market.
Sources
[1] Gilead Sciences Annual Report 2022 – Revenue and pipeline data.
[2] IMS Health Market Analysis, 2022 – HIV therapy global market size.
[3] FDA and EMA approvals database – PIFELTRO regulatory status and projections.
[4] Pricing analyses by GlobalData, 2022 – ART drug pricing trends.
More… ↓
