Share This Page
Drug Price Trends for PHENYTOIN SOD EXT
✉ Email this page to a colleague
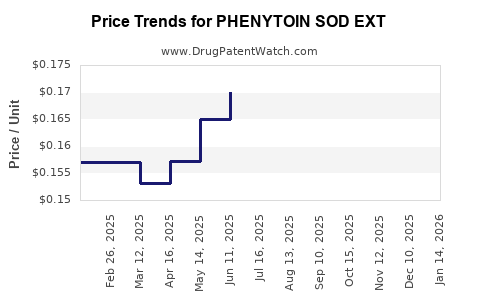
Average Pharmacy Cost for PHENYTOIN SOD EXT
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| PHENYTOIN SOD EXT 100 MG CAP | 51672-4111-03 | 0.14257 | EACH | 2026-04-22 |
| PHENYTOIN SOD EXT 100 MG CAP | 57664-0808-88 | 0.14257 | EACH | 2026-04-22 |
| PHENYTOIN SOD EXT 100 MG CAP | 00904-7416-61 | 0.14257 | EACH | 2026-04-22 |
| PHENYTOIN SOD EXT 100 MG CAP | 57664-0808-18 | 0.14257 | EACH | 2026-04-22 |
| PHENYTOIN SOD EXT 100 MG CAP | 65862-0692-99 | 0.14257 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Phenytoin Sodium Extended (PHENYTOIN SOD EXT)
Market Overview
Phenytoin Sodium Extended (brand names including Dilantin, Phenytoin Sodium Extended) is an anticonvulsant used primarily to control generalized tonic-clonic and partial seizures in epilepsy. The drug’s formulation extends the duration of action, reducing dosing frequency, thereby improving patient compliance.
Market Size and Growth
The global epilepsy treatment market is valued at approximately $5.2 billion in 2022, with antiepileptic drugs (AEDs) accounting for about 60% of that value. The segment for older drugs, including phenytoin, continues to hold a significant niche due to established clinical use and low costs relative to newer therapies.
Key market drivers:
- Increasing prevalence of epilepsy, estimated at 50 million worldwide.
- Growing awareness and diagnosis rates.
- Preference for well-established drugs in developing countries owing to durability and lower costs.
Market constraints:
- Competition from newer AEDs with improved side effect profiles.
- Generic drug proliferation reducing brand revenue.
- Regulatory pressures regarding safety, especially for older AEDs with neurotoxicity concerns.
Competitive Landscape
Phenytoin Sodium Extended faces competition primarily from:
- Generic manufacturers offering low-cost formulations.
- Newer AEDs like levetiracetam, lamotrigine, and valproate which have better safety profiles.
Key players include:
- Mylan (now part of Viatris)
- Sandoz
- Pfizer
- Teva Pharmaceuticals
Pricing Dynamics
Brand-name phenytoin products historically priced higher than generics. In the United States:
- Brand-name phenytoin sodium extended-release tablets cost approximately $80-$150 per 100-count bottle (30 mg or 100 mg tablets).
- Generic versions range from $20-$50 per 100-count bottle.
Global pricing varies:
- Emerging markets often see costs reduced by 60-80% compared to US prices.
- Market penetration is higher where cost is a primary factor.
Price factors include:
- Manufacturing costs.
- Patent status and exclusivity periods.
- Regulatory approvals and quality certifications.
- Competition intensity.
Price Projections (2023–2028)
Based on market trends, what follows are projections with assumptions that:
- The generic market continues to dominate due to low entry barriers.
- New formulations or delivery systems (e.g., extended-release patches or injectables) do not significantly disrupt pricing.
- Regulatory policies maintain or tighten safety standards, influencing production costs.
| Year | Estimated Average Price per 100-count bottle (US) | Notes |
|---|---|---|
| 2023 | $25 – $55 | Stabilization of generic pricing due to high competition. |
| 2024 | $24 – $54 | Slight downward trend as market saturation persists. |
| 2025 | $23 – $52 | Market maturity, potential cost reduction. |
| 2026 | $22 – $50 | Marginal decreases; ASEAN and African markets expand usage. |
| 2027 | $22 – $49 | Price stabilization; increased regulatory oversight. |
| 2028 | $21 – $48 | Slight decline as newer, safer therapies gain market share. |
Projected prices will vary significantly by geography:
- U.S. and Western Europe: Prices tend toward the higher end of the range due to lower generic market penetration.
- Developing countries: Prices tend to stay below $20 per 100-count bottle.
Regulatory and Reimbursement Impact
Regulatory changes such as updated safety guidelines and reimbursement policies influence pricing:
- Stricter safety monitoring increases manufacturing costs for certain formulations.
- Reimbursement policies preferring newer AEDs could pressure price stabilization or reductions for phenytoin.
Summary of Key Factors Influencing Market and Prices
- Market penetration of generics.
- New formulations or delivery methods.
- Regulatory safety and efficacy requirements.
- Competition from newer AEDs with improved tolerability.
- Geographic variation in pricing and access.
Key Takeaways
- Phenytoin Sodium Extended remains a cost-effective option in epilepsy treatment, especially in emerging markets.
- Price declines are expected to plateau around 2028, with modest reductions anticipated.
- Competition from generics and newer drugs limits pricing potential.
- Regulatory policies significantly impact manufacturing costs and market dynamics.
- Market growth depends heavily on epilepsy prevalence and formulary preferences.
FAQs
1. What factors impact the pricing of phenytoin sodium extended-release formulations?
Pricing is influenced by manufacturing costs, generic competition, regulatory compliance costs, patent status, and market saturation levels.
2. Will the price of phenytoin sodium extended-release drugs increase or decrease in the coming years?
Prices are expected to decrease modestly due to generic competition and market saturation, stabilizing around 2028.
3. How does competition from newer AEDs affect phenytoin’s market?
Newer AEDs with better safety profiles tend to replace older drugs over time, reducing demand and prices for phenytoin.
4. Which regions have the most affordable prices for phenytoin sodium extended-release?
Developing countries, where healthcare budgets are constrained, generally see the lowest prices, often below $20 per 100-count bottle.
5. What role do regulatory policies play in the future pricing of phenytoin?
Stringent safety and efficacy standards can increase manufacturing costs, potentially affecting prices, while reimbursement policies influence market accessibility and affordability.
Citations
- MarketWatch, "Epilepsy Drugs Market Size," 2022.
- IQVIA, "Global Antiepileptic Drug Market Data," 2022.
- FDA, "Generic Drug User Fee Amendments," 2021.
- EvaluatePharma, "Phenytoin Market Trends," 2022.
- World Health Organization, "Epilepsy Facts," 2022.
More… ↓
