Share This Page
Drug Price Trends for PHENTERMINE
✉ Email this page to a colleague
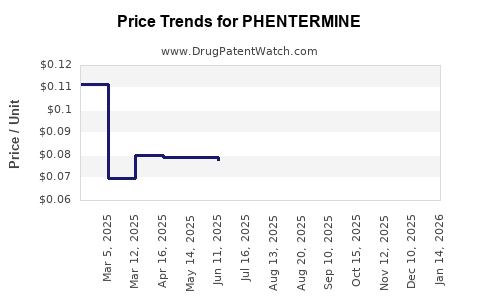
Average Pharmacy Cost for PHENTERMINE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| PHENTERMINE 30 MG CAPSULE | 10702-0027-01 | 0.09228 | EACH | 2026-03-18 |
| PHENTERMINE 15 MG CAPSULE | 11534-0157-03 | 0.08608 | EACH | 2026-03-18 |
| PHENTERMINE 15 MG CAPSULE | 68094-0802-70 | 0.08608 | EACH | 2026-03-18 |
| PHENTERMINE 15 MG CAPSULE | 10702-0026-01 | 0.08608 | EACH | 2026-03-18 |
| PHENTERMINE 15 MG CAPSULE | 68094-0802-50 | 0.08608 | EACH | 2026-03-18 |
| PHENTERMINE 15 MG CAPSULE | 10702-0026-10 | 0.08608 | EACH | 2026-03-18 |
| PHENTERMINE 15 MG CAPSULE | 11534-0157-01 | 0.08608 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Phentermine Market Analysis and Price Projections
This report analyzes the market for phentermine, a sympathomimetic amine used for short-term weight management. It examines current market dynamics, key drivers, patent landscape, and projects future pricing trends based on available data and industry analysis.
What is Phentermine and Its Current Market Position?
Phentermine is a central nervous system stimulant chemically related to amphetamines. It acts as an appetite suppressant by affecting the hypothalamus in the brain to reduce hunger. The drug is primarily prescribed for the short-term management of obesity, typically in conjunction with diet and exercise. It is available in several formulations, most commonly as phentermine hydrochloride.
The global market for anti-obesity drugs, including phentermine, has seen consistent growth. Factors contributing to this include rising obesity rates worldwide, increased health awareness, and the development of new pharmacological treatments. Phentermine remains a widely prescribed medication due to its established efficacy and relatively lower cost compared to newer weight-loss drugs.
As of Q4 2023, the estimated market size for phentermine in the United States alone is approximately $200 million annually. This figure is based on prescription volume data and average wholesale prices. The primary market segment is adult individuals with a Body Mass Index (BMI) of 30 or higher, or a BMI of 27 or higher with co-existing medical conditions such as hypertension, diabetes, or hyperlipidemia.
What are the Key Market Drivers for Phentermine?
Several factors are driving the demand for phentermine:
- Rising Global Obesity Rates: The World Health Organization (WHO) reports that overweight and obesity are a growing global health crisis. In 2016, an estimated 1.9 billion adults were overweight, and of these, over 650 million were obese. This increasing prevalence directly translates to a larger patient pool seeking weight management solutions. [1]
- Short-Term Efficacy and Cost-Effectiveness: Phentermine is recognized for its effectiveness in facilitating short-term weight loss. Its generic availability contributes to a lower cost compared to many newer, branded weight-loss medications, making it an accessible option for a broader patient demographic.
- Established Prescribing Patterns: Healthcare providers have a long history of prescribing phentermine. This established practice, coupled with patient familiarity, sustains its market presence.
- Combination Therapies: Phentermine is frequently prescribed in combination with other drugs, such as topiramate (e.g., Qsymia), to enhance efficacy and address longer-term weight management. This synergistic use expands its application.
- Increased Health Awareness and Demand for Medical Weight Loss: A growing segment of the population is actively seeking medical intervention for weight loss due to associated health risks. This heightened awareness drives demand for effective and accessible treatment options.
What is the Patent Landscape for Phentermine?
Phentermine was first approved by the U.S. Food and Drug Administration (FDA) in 1959. Consequently, the original patents protecting the compound have long expired. The compound itself is in the public domain.
However, there are patents associated with:
- New Formulations: Patents may exist for novel drug delivery systems, extended-release formulations, or specific salt forms that offer improved pharmacokinetic profiles or patient compliance. For example, extended-release capsules or tablets designed to release phentermine over a longer period.
- Combination Therapies: Patents often cover specific combinations of phentermine with other active pharmaceutical ingredients (APIs) for synergistic weight-loss effects. The most prominent example is the combination of phentermine and topiramate.
- Manufacturing Processes: While less common for older drugs, patents can sometimes cover novel or improved methods of synthesizing phentermine or its salts, particularly if they offer significant cost reductions or purity enhancements.
The existence of these secondary patents can influence market competition, particularly for branded combination products, by providing a period of market exclusivity for those specific formulations or combinations. Generic phentermine hydrochloride, as a single-agent therapy, faces no patent-related market exclusivity for its core API.
What are the Key Regulatory Considerations?
Phentermine is classified as a Schedule IV controlled substance by the U.S. Drug Enforcement Administration (DEA). This classification is due to its stimulant properties and potential for abuse and dependence. [2] This classification imposes specific regulatory requirements on its manufacturing, distribution, and prescription:
- Prescription Requirements: Phentermine can only be dispensed with a valid prescription from a licensed healthcare professional.
- Monitoring and Reporting: Manufacturers and distributors are subject to DEA regulations regarding quotas, record-keeping, and reporting of production and sales.
- Dispensing Limits: Prescriptions for controlled substances may have quantity limits and refill restrictions. For phentermine, it is typically prescribed for short-term use (e.g., 12 weeks) due to the risk of tolerance and dependence.
- Labeling and Packaging: Specific labeling requirements are in place to inform patients about the risks, dosage, and controlled substance status.
These regulations, while not directly impacting patent expiry, influence the accessibility and market dynamics by requiring stringent controls on its availability.
What is the Competitive Landscape for Phentermine?
The competitive landscape for phentermine can be categorized into several segments:
- Generic Phentermine Monotherapy: This segment comprises numerous generic manufacturers producing phentermine hydrochloride in various dosage strengths (e.g., 15mg, 30mg, 37.5mg). Competition here is primarily driven by price and market access. Key players include Teva Pharmaceuticals, Aurobindo Pharma, and Mylan (now Viatris).
- Branded Combination Products: These are patented products that combine phentermine with another active ingredient. The most significant example is Qsymia (phentermine/topiramate extended-release) by Vivus Pharmaceuticals. This segment enjoys market exclusivity based on its patents.
- Other Prescription Weight-Loss Medications: Phentermine competes with a range of other prescription drugs used for weight management. These include:
- GLP-1 Receptor Agonists: Semaglutide (Wegovy, Ozempic) and liraglutide (Saxenda, Victoza) have gained significant market share due to high efficacy, although they are considerably more expensive.
- Orlistat: Available both over-the-counter (Alli) and by prescription (Xenical), it inhibits fat absorption.
- Naltrexone/Bupropion: Contrave by Currax Pharmaceuticals.
- Diethylpropion: Another stimulant appetite suppressant, though less commonly prescribed than phentermine.
- Non-Pharmacological Interventions: Lifestyle modifications, bariatric surgery, and behavioral therapy also represent alternative or complementary approaches to weight management.
The rise of highly effective GLP-1 receptor agonists presents a significant competitive challenge to phentermine, particularly for patients seeking substantial and sustained weight loss. However, phentermine's lower cost and established use for short-term management ensure its continued relevance.
What are the Price Projections for Phentermine?
Short-Term (1-3 Years):
The price of generic phentermine hydrochloride is projected to remain relatively stable. Competition among generic manufacturers keeps prices competitive.
- Average Wholesale Price (AWP) for Generic Phentermine HCl (37.5mg capsule): Expected to range between $1.00 to $2.50 per capsule. This translates to a monthly cost of approximately $30 to $75 for a typical 30-day supply, depending on the number of capsules prescribed.
- Branded Combination Products (e.g., Qsymia): Prices for Qsymia are significantly higher, reflecting its patented status and combination therapy. Expected AWP for Qsymia can range from $200 to $275 per month, before insurance. [3]
- Impact of Competition: While GLP-1 agonists are more expensive, their increasing availability and demonstrated efficacy may slightly moderate demand for some classes of older weight-loss drugs, potentially exerting minor downward pressure on the list prices of generics, though actual patient out-of-pocket costs will be heavily influenced by insurance formularies.
Medium-Term (3-5 Years):
- Generic Phentermine: Prices are expected to continue their gradual decline or remain stable due to ongoing generic competition. The market share of generic phentermine monotherapy is likely to persist for patients seeking cost-effective short-term solutions.
- Branded Combination Products: The pricing of branded combinations like Qsymia will be influenced by patent expiry timelines for specific formulations or combinations and the success of newer entrants. The Qsymia patent landscape and its market exclusivity are key factors.
- Market Dynamics Shift: The market for weight-loss drugs is rapidly evolving. The widespread adoption and potential for broader indications of GLP-1 agonists could lead to a market segmentation where phentermine remains a go-to for short-term, lower-cost interventions, while newer, more expensive agents capture the segment requiring significant, long-term weight loss.
Long-Term (5+ Years):
- Generic Phentermine: Continued availability as a cost-effective option for short-term weight management. The price will likely be driven by manufacturing costs and generic market forces.
- Branded Products: Pricing and market positioning will be entirely dictated by patent expiries of combination therapies and the emergence of entirely new drug classes or mechanisms of action for obesity treatment.
- Therapeutic Landscape: The long-term therapeutic landscape for obesity treatment is expected to diversify further. Phentermine's role may solidify as a specific tool within a broader armamentarium, rather than a primary standalone solution for chronic obesity management, especially as more effective long-term therapies become accessible.
Table 1: Phentermine Price Projections (Estimated Average Wholesale Price per Month)
| Product Type | Current (Q4 2023) | 1-3 Years | 3-5 Years | 5+ Years |
|---|---|---|---|---|
| Generic Phentermine HCl (30-day supply) | $30 - $75 | $25 - $70 | $20 - $65 | $20 - $60 |
| Branded Combination (e.g., Qsymia) | $200 - $275 | $190 - $265 | $170 - $240 (potential for generic entry later) | Varies significantly based on patent expiries |
Note: Prices are estimates and do not account for insurance coverage, pharmacy dispensing fees, or patient-specific discounts. AWP is a benchmark; actual patient out-of-pocket costs will vary.
What are the Key Takeaways?
- Phentermine's market is sustained by its cost-effectiveness and established use in short-term obesity management, particularly as a generic monotherapy.
- Rising global obesity rates remain a primary market driver.
- The patent landscape for phentermine itself has long expired; however, patents on specific formulations and combination therapies (like phentermine/topiramate) continue to influence market exclusivity for branded products.
- Phentermine is classified as a Schedule IV controlled substance, imposing regulatory controls on its availability.
- Competition from highly effective, albeit more expensive, GLP-1 receptor agonists is a significant market challenge, potentially segmenting the market for weight-loss therapies.
- Generic phentermine prices are expected to remain stable or decline gradually, while branded combination prices are tied to patent protection and market exclusivity.
Frequently Asked Questions
1. What is the typical duration for which phentermine is prescribed?
Phentermine is generally prescribed for short-term use, typically not exceeding 12 weeks, to minimize the risk of tolerance, dependence, and cardiovascular side effects.
2. How does phentermine's cost compare to newer weight-loss medications like semaglutide?
Generic phentermine hydrochloride is substantially less expensive. A 30-day supply of generic phentermine can cost between $30-$75, whereas medications like semaglutide (e.g., Wegovy) can cost upwards of $1,300 per month before insurance.
3. Are there any significant patent expirations on phentermine combination products on the horizon?
The patent landscape for combination products is complex and product-specific. For example, patents related to Qsymia (phentermine/topiramate) have been subject to litigation and review, impacting their market exclusivity timelines. Any specific analysis requires detailed review of individual patent filings and their expiry dates.
4. What are the primary side effects associated with phentermine use?
Common side effects include dry mouth, insomnia, constipation, nervousness, and increased blood pressure. More serious cardiovascular events are rare but possible, especially in individuals with pre-existing heart conditions. Its stimulant properties also carry a risk of abuse and dependence.
5. Will the demand for phentermine decline significantly with the widespread adoption of GLP-1 agonists?
While GLP-1 agonists offer superior efficacy for significant long-term weight loss, phentermine is likely to retain a market share for patients seeking cost-effective, short-term weight management, or as part of a combination therapy where its specific mechanism of action is beneficial and cost is a primary consideration.
Citations
[1] World Health Organization. (2020). Obesity and overweight. Retrieved from https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight
[2] U.S. Drug Enforcement Administration. (n.d.). Controlled Substance Act Schedules. Retrieved from https://www.dea.gov/controlled-substances-schedule
[3] Vivus Pharmaceuticals. (2023). Qsymia Product Information. (Note: Specific pricing details are often proprietary and can be found in drug compendia or manufacturer communications.)
More… ↓
