Drug Price Trends for PERINDOPRIL ERBUMINE
✉ Email this page to a colleague
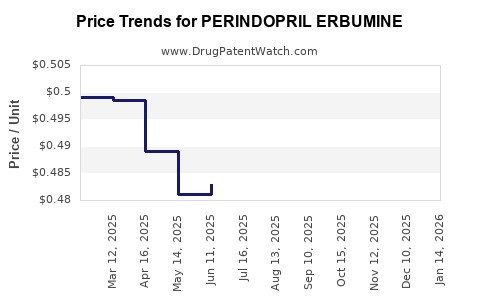
Average Pharmacy Cost for PERINDOPRIL ERBUMINE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| PERINDOPRIL ERBUMINE 8 MG TAB | 65862-0288-01 | 0.57387 | EACH | 2026-05-20 |
| PERINDOPRIL ERBUMINE 2 MG TAB | 65862-0286-01 | 0.41872 | EACH | 2026-05-20 |
| PERINDOPRIL ERBUMINE 4 MG TAB | 65862-0287-01 | 0.51401 | EACH | 2026-05-20 |
| PERINDOPRIL ERBUMINE 2 MG TAB | 65862-0286-01 | 0.44440 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
