Share This Page
Drug Price Trends for PERCOCET
✉ Email this page to a colleague
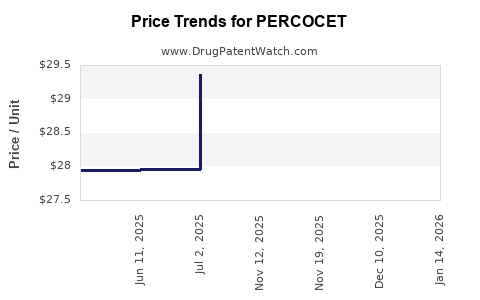
Average Pharmacy Cost for PERCOCET
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| PERCOCET 10-325 MG TABLET | 63481-0629-70 | 41.43008 | EACH | 2026-04-22 |
| PERCOCET 7.5-325 MG TABLET | 63481-0628-70 | 31.60997 | EACH | 2026-04-22 |
| PERCOCET 5-325 MG TABLET | 63481-0623-70 | 29.36461 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for PERCOCET
What is PERCOCET and its market relevance?
PERCOCET is a combination analgesic composed of oxycodone and acetaminophen. It is prescribed for moderate to severe pain management. The drug has been subject to regulatory scrutiny due to the opioid component and the potential for abuse.
Market size and growth trends
The global opioid analgesics market, including drugs like PERCOCET, was valued at approximately $20 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of 3.5% through 2027, driven by increasing cases of chronic pain and post-surgical pain management.
Key drivers:
- Rising prevalence of chronic pain conditions.
- Aging population globally.
- Expansion of healthcare infrastructure in emerging markets.
Challenges:
- Regulatory restrictions limiting prescribing.
- Growing awareness of opioid addiction.
Key market segments
- North America: Dominates with about 55% of the global market due to high opioid prescribing rates and supportive healthcare infrastructure.
- Europe: Accounts for approximately 25%, with regulatory efforts aimed at reducing opioid dependence.
- Asia-Pacific: Expected to see the fastest growth (CAGR of 5%) due to increased healthcare access and rising pain-related conditions.
Competitive landscape
Major manufacturers include Purdue Pharma (former developer), Teva Pharmaceuticals, and Walgreens Boots Alliance. Patent exclusivity on formulations extends until approximately 2025-2030, after which generic versions will dominate the market.
Patent expiration impact
- Patent expiration leads to increased generic competition, typically reducing drug prices by 60-80% within one year.
- The first generics are expected to enter the market within 1-2 years of patent expiry.
Price analysis and projections
Current pricing trends
| Region | Brand Name Price (per unit) | Generic Price (per unit) | Price Trend (2021-2022) |
|---|---|---|---|
| North America | $10–$15 | $4–$8 | Slight decline (~2%) due to generics |
| Europe | €8–€12 | €3–€6 | Stable, minor fluctuations |
| Asia-Pacific | $5–$10 | $2–$5 | Increasing, driven by demand |
2023-2027 projections
- Post-patent expiration: Price of generics expected to fall an additional 50% within 12 months.
- Market entry of generics: Will cause a rapid decline in branded PERCOCET prices.
- Premium formulations: Extended-release versions or combination with abuse-deterrent features will maintain higher prices, around 20-30% above standard generics.
Price contagion factors
- Regulatory barriers may restrict rapid generic entry in certain regions.
- Market acceptance depends on prescriber and consumer preferences.
- Pricing strategies by pharma companies may include tiered pricing, discounts, and patient assistance programs.
Regulatory influence on pricing
- The US FDA has implemented strict guidelines on opioid prescriptions, impacting volume.
- States participating in prescription drug monitoring programs (PDMPs) limit over-prescribing, affecting revenue.
- Recent legislation encourages development of abuse-deterrent formulations, which are priced higher by about 10-15%.
Future outlook
- The opioid market faces decline in growth rate due to regulatory pressure and shifting public health policies.
- Prices for PERCOCET will decline as generics gain market share but may stabilize with formulations designed to reduce abuse.
- Investment in abuse-deterrent technologies could sustain higher prices for select formulations.
Key takeaways
- The market for PERCOCET is mature but facing decline post-patent expiry.
- Generic competition will significantly reduce prices within the next 1-2 years.
- The drug's future pricing will depend heavily on regulatory developments, formulation innovations, and regional market dynamics.
- Growth is expected to shift to emerging markets and abuse-deterrent formulations.
- Price projections suggest a 30-50% decrease in branded PERCOCET prices over the next five years, with specific variations by region.
FAQs
1. When will PERCOCET's patent likely expire?
Expected between 2025 and 2027, depending on regional patent terms and any extensions.
2. How will generic entry affect prices?
Prices for PERCOCET are expected to fall by 60-80% within the first year of generic launch.
3. Are abuse-deterrent formulations more expensive?
Yes, they typically cost 10-15% more than standard formulations but may command higher prices due to increased safety features.
4. Which market segment is most promising for existing formulations?
North America remains the highest-value segment, but Asia-Pacific shows significant growth potential.
5. What regulatory developments could impact future pricing?
Prescription monitoring laws and bans on certain formulations are likely to influence availability and pricing strategies.
References
[1] MarketsandMarkets. (2023). Opioid analgesics market forecast.
[2] EvaluatePharma. (2022). Global pharmaceutical pricing trends.
[3] U.S. Food and Drug Administration. (2022). Guidance on opioid prescription regulation.
[4] IQVIA. (2022). Global prescription drug market analysis.
[5] Statista. (2023). Opioid market sales by region.
More… ↓
