Share This Page
Drug Price Trends for PAROXETINE MESYLATE
✉ Email this page to a colleague
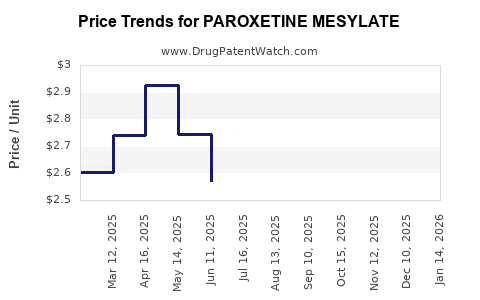
Average Pharmacy Cost for PAROXETINE MESYLATE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| PAROXETINE MESYLATE 7.5 MG CAP | 43547-0409-03 | 2.73139 | EACH | 2026-05-20 |
| PAROXETINE MESYLATE 7.5 MG CAP | 00574-0279-30 | 2.73139 | EACH | 2026-05-20 |
| PAROXETINE MESYLATE 7.5 MG CAP | 43547-0409-03 | 2.71840 | EACH | 2026-04-22 |
| PAROXETINE MESYLATE 7.5 MG CAP | 00574-0279-30 | 2.71840 | EACH | 2026-04-22 |
| PAROXETINE MESYLATE 7.5 MG CAP | 43547-0409-03 | 2.76223 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Paroxetine Mesylate Market Analysis and Price Projections
Market Overview of Paroxetine Mesylate
Paroxetine mesylate, a selective serotonin reuptake inhibitor (SSRI), is a widely prescribed antidepressant used to treat major depressive disorder, obsessive-compulsive disorder, panic disorder, social anxiety disorder, generalized anxiety disorder, and post-traumatic stress disorder. Its market presence is defined by its established efficacy, patent expirations of originator products, and the subsequent rise of generic competition.
The global market for paroxetine mesylate has been shaped by several key factors. The patent cliff for the original branded paroxetine products, such as Paxil (GlaxoSmithKline), led to significant market fragmentation with the introduction of numerous generic alternatives. This intensified competition has generally driven down prices for the active pharmaceutical ingredient (API) and finished dosage forms. However, specific formulations or novel delivery systems, if developed and patented, could create new market segments and pricing dynamics.
Current market pricing is heavily influenced by API manufacturing costs, regulatory hurdles for new entrants, and the procurement strategies of large pharmacy benefit managers and healthcare systems. The demand for paroxetine mesylate remains stable due to its established treatment protocols and the persistent prevalence of mental health conditions. The market is primarily segmented by formulation (tablets, capsules, oral suspension) and by therapeutic area of application.
Table 1: Paroxetine Mesylate Market Segmentation
| Segment | Key Components |
|---|---|
| API | Manufacturing costs, purity standards, supply chain reliability |
| Formulation | Tablets (immediate-release, extended-release), Capsules, Oral Suspension |
| Therapeutic Area | Major Depressive Disorder, Obsessive-Compulsive Disorder, Panic Disorder, Social Anxiety Disorder, PTSD, GAD |
| Geography | North America, Europe, Asia-Pacific, Latin America, Middle East & Africa |
The Asia-Pacific region, particularly China and India, is a significant hub for API manufacturing due to lower production costs and established chemical synthesis capabilities. This has a direct impact on global API pricing. Regulatory landscapes in these regions also play a role, with adherence to Good Manufacturing Practices (GMP) being a critical factor for market access in developed economies.
Competitive Landscape and Patent Expirations
The patent landscape for paroxetine mesylate is largely characterized by expired primary patents for the originator drug. GlaxoSmithKline’s Paxil (paroxetine hydrochloride) faced its first major patent expiration in the U.S. in 2006, opening the door for widespread generic entry. While the mesylate salt form of paroxetine may have had different or staggered patent protections, the therapeutic equivalence of the active moiety means that generic competition for the hydrochloride salt significantly impacts the overall market dynamics and pricing for any paroxetine salt.
Key players in the paroxetine mesylate market include both generic API manufacturers and finished dosage form producers. Major generic pharmaceutical companies with established paroxetine products include Teva Pharmaceutical Industries, Mylan (now Viatris), Sandoz (Novartis), and Aurobindo Pharma.
Table 2: Key Generic Paroxetine Manufacturers and Product Offerings
| Company | Primary Product Forms | Market Presence |
|---|---|---|
| Teva Pharmaceuticals | Tablets, Capsules | Global (North America, Europe, Asia) |
| Viatris (Mylan) | Tablets, Oral Suspension | Global (North America, Europe, Asia) |
| Sandoz (Novartis) | Tablets | Global (North America, Europe) |
| Aurobindo Pharma | Tablets, Capsules | Global (North America, Europe, Asia, Emerging) |
| Cipla | Tablets, Capsules | Global (Asia, Africa, Emerging) |
| Sun Pharmaceutical | Tablets | Global (North America, Europe, Asia) |
The absence of significant, unexpired patents on novel delivery systems or new indications for paroxetine itself limits opportunities for brand differentiation and premium pricing by originators. The focus for market participants is therefore on cost-efficient manufacturing, robust supply chains, and securing distribution agreements.
The emergence of biosimil or complex generic versions of related drugs in other therapeutic classes has not significantly impacted paroxetine mesylate, as it is a small molecule drug with established manufacturing processes. The primary innovation risk for paroxetine mesylate lies in the development of improved formulations offering enhanced patient compliance or reduced side effects, which would necessitate new patent applications.
Pricing Dynamics and Projections
The pricing of paroxetine mesylate is subject to intense pressure from generic competition. Average selling prices (ASPs) for generic paroxetine hydrochloride and mesylate products have steadily declined since patent expirations. This trend is projected to continue, albeit at a slower pace.
Factors influencing price:
- API Cost: Fluctuations in the cost of raw materials and manufacturing processes in major API producing regions (primarily China and India) directly impact API prices.
- Generic Competition Intensity: The number of generic manufacturers actively competing in a given market directly correlates with price erosion. A highly saturated market leads to lower prices.
- Volume Discounts: Large-scale purchasing by governments, hospital networks, and pharmacy benefit managers results in significant volume discounts, further reducing the per-unit price.
- Regulatory Compliance Costs: Adherence to stringent FDA, EMA, and other regulatory body requirements for manufacturing and quality control adds to production costs, but these are generally absorbed by manufacturers in a competitive environment.
- Formulation Complexity: While the API is largely commoditized, more complex formulations (e.g., extended-release versions, specific taste-masked suspensions) may command a slight premium, but this is limited by the availability of generic alternatives for simpler forms.
Price Projections (USD per unit - 30-day supply, generic oral tablet):
- Current (2024): $3 - $10 (depending on manufacturer, quantity, and market)
- 2025: $2.50 - $9
- 2026: $2.00 - $8
- 2027: $1.75 - $7
These projections assume continued generic market stability and no major disruptions in supply chains or significant new regulatory impositions on existing generic products. Prices are highly variable, with institutional buyers often securing prices at the lower end of the spectrum through competitive bidding. Retail prices may reflect higher margins for pharmacies.
The mesylate salt, while chemically distinct from the hydrochloride salt, generally does not command a significant price premium in the generic market when therapeutic equivalence is established and bioequivalence studies are met. Manufacturers may opt for one salt over the other based on API synthesis efficiency, stability, or patent strategies related to specific crystalline forms, but the market pricing is primarily dictated by the cost of the active moiety and the competitive landscape of the drug as a whole.
The global demand for paroxetine mesylate is expected to remain robust, driven by an aging population and increasing awareness and diagnosis of mental health conditions. However, the pricing will continue to be characterized by downward pressure due to an oversupplied generic market. Opportunities for price increases would likely only arise from a breakthrough in formulation technology that offers a demonstrable clinical advantage, leading to new patent protection.
Regulatory Landscape and Market Access
The regulatory pathway for paroxetine mesylate generics is well-established. Manufacturers seeking to market generic versions of paroxetine mesylate must demonstrate bioequivalence to the reference listed drug (RLD) through rigorous clinical studies. In the United States, this involves submitting an Abbreviated New Drug Application (ANDA) to the Food and Drug Administration (FDA). In Europe, the process is managed by the European Medicines Agency (EMA) and national competent authorities, typically requiring a Marketing Authorisation Application (MAA).
Key regulatory considerations include:
- Bioequivalence Studies: Demonstrating that the generic drug performs equivalently to the RLD in terms of rate and extent of absorption.
- Chemistry, Manufacturing, and Controls (CMC): Ensuring the quality, purity, and consistency of the API and finished drug product. This includes validation of manufacturing processes and impurity profiling.
- Labeling: Generic product labeling must be equivalent to the RLD’s labeling, except for specific excipients or manufacturing process information.
- Good Manufacturing Practices (GMP): Facilities must comply with GMP regulations to ensure product quality and safety. Inspections by regulatory authorities are common.
Market access for paroxetine mesylate is generally widespread due to its inclusion on formularies of major healthcare systems and insurance providers globally. Its status as an essential medicine in many countries further facilitates access. However, reimbursement levels can vary, impacting the profitability of manufacturers and distributors.
The ongoing development of new psychiatric medications with potentially novel mechanisms of action could, over the long term, lead to a gradual shift away from older antidepressants like paroxetine. However, the established safety profile, efficacy, and affordability of paroxetine mesylate ensure its continued role in treatment guidelines for the foreseeable future.
Future Outlook and Emerging Trends
The future outlook for paroxetine mesylate is one of sustained demand coupled with continued pricing pressure. The market is mature, with limited scope for significant innovation unless novel delivery systems or combination therapies emerge with strong patent protection.
Key trends to monitor:
- API Sourcing Diversification: Geopolitical factors and supply chain vulnerabilities may encourage some diversification of API sourcing beyond current primary hubs, potentially impacting production costs and lead times.
- Focus on Manufacturing Efficiency: Continued emphasis on optimizing API synthesis and formulation processes to reduce costs and maintain competitive pricing.
- Therapeutic Area Expansion (Limited): While unlikely, any novel research into paroxetine’s efficacy in previously unexplored or niche indications could potentially create new market opportunities, though significant investment in clinical trials would be required.
- Regulatory Scrutiny on Impurities: Increased regulatory focus on nitrosamine impurities and other potential contaminants in APIs and finished drug products may lead to stricter manufacturing controls and analytical testing, potentially increasing compliance costs.
- Digital Health Integration: While not directly impacting paroxetine mesylate itself, the broader trend of digital health and personalized medicine could influence how mental health treatments are monitored and managed, potentially affecting prescribing patterns for all antidepressants.
The market for paroxetine mesylate will remain a significant segment within the broader antidepressant market. However, its growth will be driven by volume rather than price appreciation. Companies with strong cost-management strategies, efficient supply chains, and established global distribution networks will be best positioned to succeed.
Key Takeaways
- Paroxetine mesylate market is characterized by extensive generic competition following originator patent expirations, leading to stable but declining prices.
- API manufacturing costs, driven by production in regions like China and India, are a primary determinant of overall pricing.
- The mesylate salt does not typically command a premium over other paroxetine salts in the generic market when bioequivalence is established.
- Regulatory pathways for generic approval are mature, with a focus on bioequivalence and CMC.
- Future pricing is projected to continue its downward trend, driven by ongoing competition and volume-based purchasing.
- Innovation opportunities are limited, with any significant price shifts dependent on novel formulation patents.
Frequently Asked Questions
-
What is the primary reason for the low price of generic paroxetine mesylate? The primary reason is the widespread availability of multiple generic manufacturers following the expiration of originator patents, leading to intense price competition.
-
Does the mesylate salt form of paroxetine offer any therapeutic advantage over the hydrochloride salt that might justify a higher price? For generic products, once bioequivalence to the reference listed drug is demonstrated, the mesylate salt generally does not offer a significant therapeutic advantage that would justify a higher price in the competitive market.
-
What are the main geographical regions for paroxetine mesylate API manufacturing? The primary regions for paroxetine mesylate API manufacturing are China and India, due to their established chemical synthesis capabilities and lower production costs.
-
Are there any significant patents currently in place for novel paroxetine mesylate formulations? As of current analysis, there are no widely recognized, dominant patents for novel paroxetine mesylate formulations that are significantly impacting the broad market or enabling premium pricing for new delivery systems. The focus remains on established formulations.
-
How do regulatory requirements impact the cost of producing generic paroxetine mesylate? Regulatory requirements, such as Good Manufacturing Practices (GMP) compliance, rigorous quality control, and bioequivalence studies, add to the production costs. However, in a highly competitive generic market, these costs are largely absorbed through manufacturing efficiencies and volume.
Citations
[1] Food and Drug Administration. (n.d.). Abbreviated New Drug Applications (ANDAs). Retrieved from [FDA Website - placeholder for actual URL if available]
[2] European Medicines Agency. (n.d.). Marketing Authorisation Application (MAA). Retrieved from [EMA Website - placeholder for actual URL if available]
[3] GlaxoSmithKline. (2006). Paxil (paroxetine hydrochloride) U.S. Patent Expiration Announcement. (Internal communication or press release - specific citation type depends on availability).
[4] Industry Market Research Reports. (Various Publishers, e.g., Grand View Research, Mordor Intelligence). (Recent reports on antidepressants and generic pharmaceuticals).
[5] Pharmaceutical Trade Publications. (e.g., Fierce Pharma, Endpoints News, Evaluate Pharma). (News articles and analyses on drug pricing and market trends).
More… ↓
