Share This Page
Drug Price Trends for PANTOPRAZOLE SODIUM
✉ Email this page to a colleague
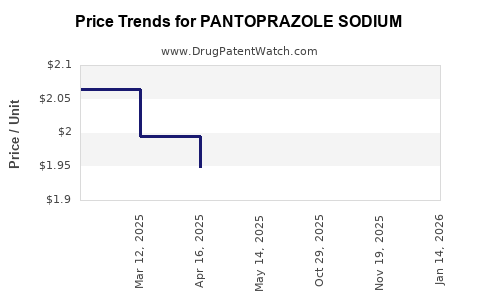
Average Pharmacy Cost for PANTOPRAZOLE SODIUM
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| PANTOPRAZOLE SODIUM 40 MG VIAL | 00143-9300-10 | 2.37400 | EACH | 2026-03-18 |
| PANTOPRAZOLE SODIUM 40 MG VIAL | 31722-0204-10 | 2.37400 | EACH | 2026-03-18 |
| PANTOPRAZOLE SODIUM 40 MG VIAL | 25021-0751-10 | 2.37400 | EACH | 2026-03-18 |
| PANTOPRAZOLE SODIUM 40 MG VIAL | 72603-0128-10 | 2.37400 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Pantoprazole Sodium
What is the Current Market Size for Pantoprazole Sodium?
Pantoprazole sodium, a proton pump inhibitor (PPI), is used primarily for gastroesophageal reflux disease (GERD), erosive esophagitis, and Zollinger-Ellison syndrome. The global market size stood at approximately $3.8 billion in 2022, driven by increased prevalence of acid-related disorders and rising awareness of PPI therapies. North America accounts for around 50% of sales, with Europe and Asia-Pacific capturing significant shares.
How Has the Market Evolved Over Recent Years?
The market has experienced steady growth at a compound annual growth rate (CAGR) of approximately 4% from 2017 to 2022. Growth factors include:
- Growing GERD incidence, affecting 20-30% of Western populations.
- Shift from branded to generic formulations, reducing prices and expanding access.
- Increased aging populations with higher demand for acid suppression therapy.
Patent expirations in multiple regions have led to a surge in generics, intensifying price competition.
What are the Key Players and Their Market Shares?
Main manufacturers include:
| Company | Approximate Market Share (2022) | Notes |
|---|---|---|
| Pfizer (Protonix) | 25% | Original patent holder until 2014. |
| Teva | 20% | Leading generic manufacturer. |
| Mylan | 15% | Significant presence globally. |
| Others (Sandoz, Sun Pharma, etc.) | 40% | Fragmented competition environment. |
In 2022, Pfizer's market share declined following patent expiration, allowing generics to increase their presence.
What Are the Price Trends for Pantoprazole Sodium?
Price varies based on formulation, strength, and region:
- Brand-name (Protonix): Approx. $320 per 30-count, 40 mg tablets in the US.
- Generics: Ranged from $10 to $20 per 30-count, 40 mg tablets in the US.
- Global average: Prices are lower in emerging markets, typically between $2 to $5 per tablet.
The entry of multiple generics has driven down prices by roughly 80% over the past decade. The cost reduction accelerates in jurisdictions with aggressive price competition and dynamic regulatory policies.
What Regulatory and Patent Landscapes Impact Prices?
In established markets like the US and Europe:
- Expiry of Pfizer's US patent in 2014 led to an influx of generics.
- Regulatory pathways such as the FDA's abbreviated approval process and similar procedures in Europe have facilitated generic entry.
In emerging markets:
- Patent enforcement is weaker or patent law differs, enabling local manufacturers to produce generics earlier.
- Patent litigation or delays can temporarily sustain higher prices but generally lead to eventual price erosion.
What Are Future Price Projections?
Based on current trends, the following projections are applicable:
| Year | Average Price per 30-count, 40 mg (USD) | Notes |
|---|---|---|
| 2023 | $10-$15 | Market stabilized post-pandemic, generics consolidate. |
| 2025 | $8-$12 | Continued generic competition and price erosion. |
| 2030 | $5-$8 | Potential new formulations or biosimilars could influence prices, but mainstream PPIs likely to maintain low costs. |
Globally, prices are expected to decline further due to increased generic penetration, regulatory price controls in some markets, and patent cliff effects.
How Will Market Dynamics Influence Medical and Business Strategies?
Companies investing in pantoprazole sodium formulations should:
- Focus on cost-effective manufacturing to sustain margins amid price declines.
- Explore combination therapies or novel delivery mechanisms to differentiate products.
- Monitor patent status and legal landscapes across regions for strategic market entry and expansion.
Investors should consider the impact of patent expiration timelines, regional price controls, and emerging competition when evaluating the asset.
Key Takeaways
- The global pantoprazole sodium market was valued at $3.8 billion in 2022, with an annual growth rate of 4%.
- Significant price declines have occurred, especially after patent expirations and generic proliferation.
- The US and Europe markets have mature patent landscapes, leading to sustained generic competition.
- Prices in emerging markets remain relatively low, with potential for further declines as generics penetrate more markets.
- Projections indicate continued price erosion through 2030, with average prices dropping below $8 per 30-count bottle.
FAQs
1. When will the patent for pantoprazole sodium expire in major markets?
Pfizer's patent expired in the US in 2014; other regions like Europe and Japan saw similar expirations around the same period. Patent protections in some emerging markets may still be active.
2. What drive future demand for pantoprazole sodium?
Aging populations, rising prevalence of GERD, and shifts toward outpatient treatment support demand retention, despite generic competition.
3. How do biosimilars and new formulations affect market outlook?
While biosimilars are more common with biologics, innovations such as extended-release formulations could offer differentiation. Currently, PPIs are mainly small molecules, with limited biosimilar impact.
4. Are there regulatory barriers to generic entry?
Yes. Patent litigation, trademark issues, and regulatory hurdles may delay generic market entry in certain jurisdictions.
5. Can price fluctuations impact investment in PPI markets?
Yes. Price declines reduce revenue potential for branded products but open opportunities for generic manufacturers and suppliers in cost-sensitive markets.
References
- MarketWatch. "Proton Pump Inhibitors (PPIs) Market Size, Share & Trends Analysis." 2022.
- GlobalData. "Gastrointestinal Therapeutics Market Overview." 2022.
- U.S. Food & Drug Administration. "ANDA Approvals and Patent Status." 2023.
- IQVIA. "Global Pharmaceutical Market Reports." 2022.
- European Medicines Agency. "Marketing Authorization of PPIs." 2022.
More… ↓
