Share This Page
Drug Price Trends for PAIN RELIEVING
✉ Email this page to a colleague
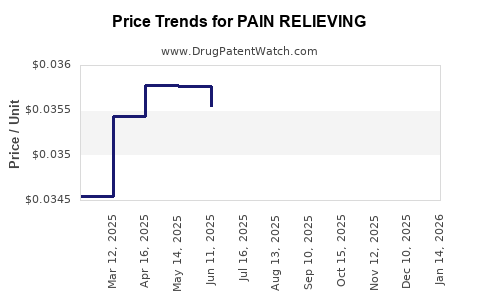
Average Pharmacy Cost for PAIN RELIEVING
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| PAIN RELIEVING 3.1-6-10% PATCH | 70000-0368-01 | 0.62007 | EACH | 2026-04-22 |
| PAIN RELIEVING 1%-15% CREAM | 00536-1101-45 | 0.04236 | GM | 2026-04-22 |
| PAIN RELIEVING 3.1-6-10% PATCH | 70000-0368-01 | 0.62115 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
PAIN RELIEVING Market Analysis and Financial Projection
What Is the Current Market Size for Pain-Relieving Drugs?
The global pain management market was valued at approximately USD 64 billion in 2021. It is projected to grow at a compound annual growth rate (CAGR) of 4.2% from 2022 to 2030. The demand is driven by increasing prevalence of chronic pain conditions, aging populations, and rising awareness of pain management therapies.
What Are the Key Segments in the Pain-Relieving Market?
Pain-relieving drugs are categorized into several segments:
- Opioids: Approximately 60% of the market, valued at USD 38 billion in 2021. This segment includes drugs like oxycodone and fentanyl.
- Non-opioid pain relievers: About 30%, valued at USD 19 billion. Includes NSAIDs like ibuprofen and acetaminophen.
- Adjuvant therapies: Roughly 10%, including antidepressants, anticonvulsants, and corticosteroids.
How Does the Market Distribution Vary Geographically?
North America dominates with roughly 45% of the global market share in 2021, driven by high treatment needs and favorable reimbursement policies. Europe accounts for around 25%, while Asia-Pacific shows the fastest growth at 5.0% CAGR due to increasing healthcare infrastructure and awareness.
What Are the Drivers and Barriers to Market Growth?
Drivers:
- Increasing prevalence of chronic pain conditions, including back pain and neuropathic pain.
- Aging populations, particularly in North America and Europe.
- Advances in drug formulations and delivery systems, such as transdermal patches and long-acting formulations.
- Growing adoption of non-opioid therapies owing to regulatory restrictions on opioids.
Barriers:
- Stringent regulations governing opioid prescriptions due to abuse potential.
- Concerns over side effects and dependency associated with opioids.
- High drug development costs and lengthy approval processes.
- Competition from non-pharmacological therapies such as physiotherapy and nerve stimulation.
What Are the Leading Drugs and Companies?
Key drugs:
- Opioids: OxyContin (purdue Pharma), Fentanyl patches (Johnson & Johnson)
- NSAIDs: Advil (Johnson & Johnson), Aleve (Bayer)
- Adjuvant therapies: Lyrica (Pfizer), Neurontin (Pfizer)
Leading companies:
- Johnson & Johnson
- Pfizer
- Teva Pharmaceutical Industries
- Novartis
- AbbVie
What Are the Price Projections for Pain-Relief Drugs?
Current Pricing Trends:
- Opioid therapies: USD 0.10–0.50 per dose. For example, fentanyl patches cost approximately USD 5–10 per patch.
- NSAIDs: USD 0.05–0.20 per tablet.
- Adjuvant therapies: USD 1–5 per capsule or tablet.
Future Price Trends (2023–2030):
Market analysts expect prices for branded opioids to decline by approximately 3% annually due to patent expirations and generic competition. Generic versions of drugs like oxycodone and fentanyl patches are expected to reduce prices significantly, lowering costs by up to 50% over the next five years.
Prices for non-opioid and adjuvant drugs may stabilize or increase slightly, driven by innovations in drug delivery and formulation technology. The introduction of biosimilars and off-patent drugs could further impact pricing.
Impact of Regulatory and Market Factors:
- Restrictions on opioid prescribing are expected to pressure prices downward.
- Increased use of non-opioid therapies may alter demand, affecting pricing strategies.
- Price decreases may be offset by shifts towards more expensive, advanced formulations, such as implantable devices and long-acting injectables.
What Are the Entry Barriers and Opportunities?
Entry barriers:
- High R&D costs and lengthy regulatory approval process.
- Established market dominance by large pharmaceutical firms.
- Stringent regulations, especially for opioids.
- Pre-existing patent protections on key drugs until mid-2020s.
Opportunities:
- Development of non-addictive pain therapies.
- Adoption of digital health and remote monitoring for pain management.
- Formulations that improve patient compliance and reduce side effects.
- Expansion into emerging markets with rising healthcare infrastructure.
Key Takeaways
- The global pain management market was valued at USD 64 billion in 2021, with a CAGR of 4.2%.
- North America dominates, but Asia-Pacific shows the highest growth potential.
- Opioids constitute the largest segment, though their market faces regulatory challenges.
- Prices for pain-relieving drugs are projected to decline for opioids due to generic competition, while non-opioid therapies may see incremental price increases.
- Market entry is challenging due to high development costs and regulatory barriers, but opportunities exist in non-addictive therapies and digital health solutions.
FAQs
1. How will regulatory changes affect opioid markets?
Regulatory restrictions on opioid prescriptions are expected to reduce demand for branded opioids and favor generic alternatives, leading to price declines.
2. What is the outlook for non-opioid pain relievers?
Growth is driven by increasing awareness of opioid risks and innovations in delivery technology. Prices are likely to stabilize, with potential slight increases for advanced formulations.
3. Are biosimilars impacting the pain management market?
Yes. Biosimilars, especially for adjuvant therapies like biologic-based treatments, are reducing treatment costs and increasing accessibility.
4. What emerging therapies are disrupting the market?
Non-pharmacological interventions, including neuromodulation devices and digital pain management tools, are gaining acceptance and market share.
5. How significant is the Asian market for future growth?
Very significant; Asia-Pacific’s CAGR is forecasted at 5.0%, driven by expanding healthcare infrastructure and rising chronic pain burden.
References
- Grand View Research. Pain Management Market Size, Share & Trends Analysis Report, 2022–2030.
- MarketWatch. Global Pain Management Drugs Market Revenue Forecast, 2022.
- WHO. Chronic Pain: Strategies for Relief, 2020.
- IQVIA. Pharmaceuticals Market Analysis, 2022.
- U.S. Food & Drug Administration. Opioid Regulations and Impact, 2021.
More… ↓
