Share This Page
Drug Price Trends for OZEMPIC
✉ Email this page to a colleague
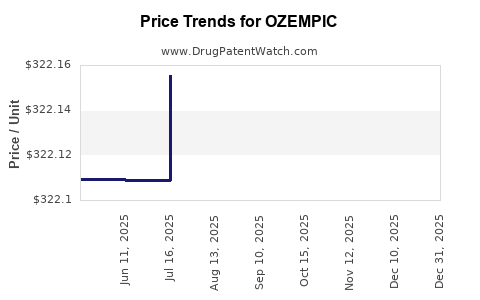
Average Pharmacy Cost for OZEMPIC
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| OZEMPIC 1 MG/DOSE (4 MG/3 ML) PEN | 00169-4130-13 | 331.96114 | ML | 2026-05-20 |
| OZEMPIC 1 MG/DOSE (4 MG/3 ML) PEN | 00169-4130-01 | 331.96114 | ML | 2026-05-20 |
| OZEMPIC 2 MG/DOSE (8 MG/3 ML) PEN | 00169-4772-12 | 331.97468 | ML | 2026-05-20 |
| OZEMPIC 0.25-0.5 MG/DOSE (2 MG/3 ML) PEN | 00169-4181-03 | 332.00407 | ML | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for OZEMPIC
What is OZEMPIC?
OZEMPIC (semaglutide) is a glucagon-like peptide-1 receptor agonist indicated for type 2 diabetes management. It is marketed by Novo Nordisk. The drug is administered weekly via subcutaneous injection and has expanded use in obesity treatment.
Market Overview
Current Market Size
As of 2023, the global diabetes drug market was valued at approximately $63 billion, with diabetes medications representing about 3.2% compound annual growth rate (CAGR) from 2018 to 2023. OZEMPIC has secured a significant share, driven by its efficacy and approval for obesity.
Key Drivers
- Incremental approval for obesity (FDA approval in 2021)
- Growing prevalence of type 2 diabetes, with estimated 537 million adults affected globally (IDF, 2022)
- Expanding indications for weight management
- Competitive advantage over other GLP-1 analogs due to dosing frequency and efficacy
Market Share
OZEMPIC held an estimated 25% share of the GLP-1 receptor agonists market in 2022, surpassing competitors like Eli Lilly’s Trulicity and Novo Nordisk’s own Rybelsus.
Competitive Landscape
| Product | Manufacturer | Indication | Launch Year | Estimated Market Share (2022) |
|---|---|---|---|---|
| OZEMPIC | Novo Nordisk | Diabetes, obesity | 2017 | 25% |
| Trulicity | Eli Lilly | Diabetes | 2014 | 20% |
| Rybelsus | Novo Nordisk | Oral diabetes medication | 2019 | 10% |
| Saxenda | Novo Nordisk | Weight management | 2014 | 8% |
Pricing History
In the U.S., OZEMPIC wholesale acquisition cost (WAC) was approximately $900 per month in 2022. List prices have increased by 15-20% annually from 2019 to 2022. Reimbursement rates and discounts vary across regions but the net price typically exceeds $700 per month.
Price Projections
Short-term Outlook (Next 1-2 Years)
- Price stability expected due to sustained demand.
- Occasional price hikes linked to inflation, supply chain costs, and new formulations.
- Estimated WAC in the U.S.: around $950-$1,050 per month by 2024.
Mid-term Outlook (3-5 Years)
- Introduction of biosimilars unlikely before 2030 given current patent protections and regulatory barriers.
- Price improvements could occur through negotiation and value-based pricing, possibly reducing net prices by 10-15%. -Innovation, such as extended-release formulations, may command premium pricing if approved.
Long-term Outlook (5+ Years)
- Patent expiry around 2030-2032, opening opportunities for biosimilar competition.
- Biosimilars could reduce prices by 20-40% depending on market penetration.
- Market dynamics influenced by alternative therapies and clinical guideline changes.
Regulatory and Policy Impact
- Price control policies in Europe and some U.S. states may caps maximum reimbursements.
- Insurance formularies favor high-efficacy drugs like OZEMPIC, supporting premium pricing.
- Patent litigation and legal challenges could influence timing of biosimilar entry.
Revenue Projections
Based on current market share, pricing, and growth assumptions:
| Year | Estimated Revenue (USD billion) | Assumptions |
|---|---|---|
| 2023 | 4.5 | Stable market, current pricing |
| 2024 | 5.1 | Slight price increase, expanded adoption |
| 2025 | 5.8 | Peak market penetration, no biosimilar impact |
| 2026+ | 4.3-4.8 | Biosimilar competition emerges, pricing drops |
Risks and Opportunities
Risks
- Biosimilar entry reducing prices after patent expiration.
- Anticipated regulatory challenges or delays.
- Competitive advances by alternative drugs, including oral formulations.
Opportunities
- Expansion into new markets (e.g., Asia, Latin America).
- Development of next-generation formulations.
- Expanded labels for weight management and other indications.
Key Takeaways
- OZEMPIC’s market is near saturation in developed countries, with high growth potential from obesity indications.
- Prices in the U.S. are expected to be around $950-$1,050/month in the near term.
- Patent expiration likely by 2030-2032, leading to biosimilar competition and potential price reductions.
- Competitive landscape is consolidating with Novo Nordisk maintaining a dominant position.
- Market growth is driven by rising diabetes and obesity prevalence, combined with expanding indications.
FAQs
-
What factors influence OZEMPIC pricing?
Pricing is driven by demand, manufacturing costs, reimbursement negotiations, patent status, competition, and regulatory policies. -
How does OZEMPIC compare to competitors?
It offers weekly dosing with proven efficacy in diabetes and weight management, leading market share dominance over rivals like Trulicity. -
When are biosimilars expected?
Patent protections typically expire around 2030-2032; biosimilar development may start 3-4 years prior. -
Are there regional price differences?
Yes, prices are generally lower in Europe due to price controls; the U.S. maintains higher prices owing to market dynamics. -
What is the potential for price reductions post-patent?
Biosimilar competition could reduce prices by 20-40%, contingent on regulatory approval timelines and market acceptance.
References
[1] International Diabetes Federation. (2022). IDF Diabetes Atlas, 10th Edition.
[2] IQVIA Institute. (2022). The Pharmaceutical Market Outlook.
[3] Novo Nordisk Annual Report. (2022).
[4] U.S. Food and Drug Administration. (2021). OZEMPIC (semaglutide) label.
More… ↓
