Share This Page
Drug Price Trends for OXYMORPHONE HCL ER
✉ Email this page to a colleague
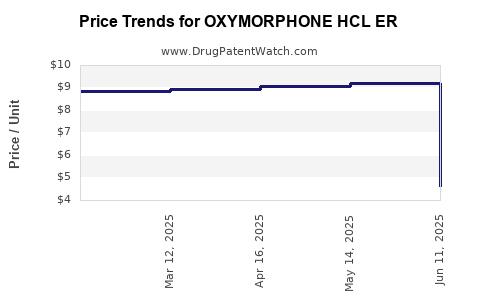
Average Pharmacy Cost for OXYMORPHONE HCL ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| OXYMORPHONE HCL ER 10 MG TAB | 64896-0697-01 | 9.27273 | EACH | 2025-06-18 |
| OXYMORPHONE HCL ER 15 MG TAB | 64896-0698-01 | 12.60921 | EACH | 2025-06-18 |
| OXYMORPHONE HCL ER 10 MG TAB | 64896-0697-13 | 9.27273 | EACH | 2025-06-18 |
| OXYMORPHONE HCL ER 15 MG TAB | 64896-0698-13 | 12.60921 | EACH | 2025-06-18 |
| OXYMORPHONE HCL ER 40 MG TAB | 64896-0701-13 | 29.50935 | EACH | 2025-05-21 |
| OXYMORPHONE HCL ER 10 MG TAB | 64896-0697-01 | 9.19210 | EACH | 2025-05-21 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
OXYMORPHONE HCL ER Market Analysis and Financial Projection
Market Analysis and Price Projections for Oxymorphone HCl ER
Market Overview
Oxycodone Hydrochloride Extended-Release (ER) is a Schedule II opioid analgesic used mainly for managing severe pain requiring around-the-clock opioid therapy. Its market is driven by the broader needs for chronic pain management, regulatory controls, patent statuses, and manufacturing practices.
Market Drivers
-
Prevalence of Chronic Pain: An estimated 50 million adults in the U.S. suffer from chronic pain, creating consistent demand for long-acting opioids. According to the CDC[1], opioid prescriptions, including ER formulations, are common in pain management protocols.
-
Regulatory Environment: Stricter regulations on opioid prescribing, including prescription caps and enhanced monitoring, influence market dynamics. Such policies may reduce overall volume but increase the importance of branded, monitored, and compliant products.
-
Generic Competition: Oxycodone ER was initially marketed by Purdue Pharma under the brand name OxyContin. Patents have expired or are near expiration, leading to many generic manufacturers entering the market, which impacts pricing.
-
Alternative Therapies: The rise of non-opioid pain management drugs, including cannabinoids and nerve modulators, could cap the growth potential for Oxycodone ER in some markets.
Market Size and Trends
-
The global opioids market was valued at approximately $8.5 billion in 2022[2], with ER formulations constituting roughly 45% of this segment in the U.S.
-
The U.S. market represents over 80% of demand for oxycodone products, driven by widespread use in pain management protocols.
-
Market growth has been relatively flat since 2021, with a compound annual growth rate (CAGR) of approximately 2%, attributable to regulatory tightening and changing prescribing habits.
Key Market Players
| Company | Product Name | Patent Expiry (U.S.) | Market Share (Estimated) | Notes |
|---|---|---|---|---|
| Purdue Pharma | OxyContin | 2023 (patent) | 20% | Leading brand; patent expired, now generic available |
| Teva Pharmaceuticals | Oxycodone ER | N/A | 15% | Major generic manufacturer, competitive pricing |
| Mallinckrodt | Oxycodone ER | 2024 (patent) | 10% | Market share gradually declining due to patent expiration |
| Others | Various | Varies | 55% | Fragmented generic landscape |
Price Trends and Projections
Historical Pricing
-
Brand-name OxyContin: In 2019, the wholesale cost per 30mg pill was approximately $10[3].
-
Generics: Prices declined rapidly post patent expiry, with average wholesale prices (AWP) for 30mg oxycodone ER dropping to $4-$6 per pill by 2020.
Current Pricing (2023)
-
Brand-name OxyContin: Approximate AWP of $9-$11 per 30mg pill due to limited supply and sustained demand.
-
Generics: Range from $3-$5 per 30mg pill, with variability depending on manufacturer and distribution channels.
Forecast (Next 3-5 Years)
-
Price Stabilization: Prices for both branded and generic oxycodone ER are expected to stabilize as market saturation occurs and supply-chain logistics normalize.
-
Price Increase Factors:
-
Regulatory restrictions disfavor higher dosages and prescription volumes, which could exert downward pressure.
-
Growing concerns about abuse potential may reduce legal prescribing, impacting demand.
-
Manufacturing costs for controlled substances might rise due to tighter security and compliance measures.
-
-
Projected Price Range (2028)
| Formulation | 30mg Pill Price Range | Comment |
|---|---|---|
| Brand OxyContin | $10-$12 | Scarce supply due to limitations on new marketing |
| Generic | $3.50-$5 | Dominant form, volume-driven pricing |
Regional Variations
-
U.S.: Dominant market with established supply chains and pricing norms.
-
Europe/Asia: Prices tend to be lower due to different regulatory environments, with price points roughly 20-30% below U.S. levels.
-
Emerging Markets: Pricing is highly variable but often significantly lower because of weaker regulatory enforcement and lower demand.
Risks and Market Constraints
-
Regulatory Risks: Increasing restrictions on opioid prescriptions could reduce market size or limit new market entry.
-
Legal Challenges: Litigation related to opioid addiction crises influences prescribing practices and product availability.
-
Diversification of Pain Management: Development of non-opioid therapeutics threatens long-term demand.
-
Public Policy Trends: Governments emphasizing opioid reduction initiatives could suppress growth and pricing.
Key Takeaways
-
Oxycodone HCl ER is supported by steady, though modest, demand driven by chronic pain management.
-
Patent expiries drive market share shifts to generics, pushing prices downward.
-
Pricing has stabilized post-generic entry, with minimal growth expected.
-
Market growth is constrained by tightening regulations and the proliferation of alternative pain therapies.
-
Regional variations significantly impact pricing strategies and market sizes.
FAQs
1. How will patent expiries affect the Future Pricing of Oxycodone ER?
Patent expiries enable generic manufacturers to enter the market, increasing competition and reducing prices. Prices for branded products typically decline sharply after patent expiration, stabilizing at competitive levels. Future pricing will depend on market volume, regulatory environment, and manufacturing costs.
2. What regulatory developments could impact the Oxycodone ER market?
Enhanced prescribing restrictions, prescription monitoring programs, and potential legislation aimed at reducing opioid abuse could decrease demand. Conversely, regulatory approval of abuse-deterrent formulations might impact the competitive landscape.
3. Are there emerging competitors to Oxycodone ER?
Yes. Non-opioid pain management drugs, including nerve growth factor inhibitors and cannabinoids, are expanding and could reduce reliance on opioid therapies, affecting long-term demand for oxycodone ER.
4. How significant are regional differences in pricing?
Regional regulatory environments significantly influence pricing. U.S. prices consistently outperform those in Europe and Asia. Variations depend on local policies, healthcare systems, and market maturity.
5. What is the outlook for generic manufacturers?
Generic manufacturers continue to compete aggressively on price. Margins are compact due to price erosion, and revenue depends heavily on market volume and regulatory stability.
References
[1] CDC. "2022 National Pain Strategy," Centers for Disease Control and Prevention, 2022.
[2] MarketWatch. "Global Opioids Market Size, Share, Growth," 2022.
[3] IMS Health. "Pharmacy & Therapeutics Data," 2019.
More… ↓
