Share This Page
Drug Price Trends for OXCARBAZEPINE ER
✉ Email this page to a colleague
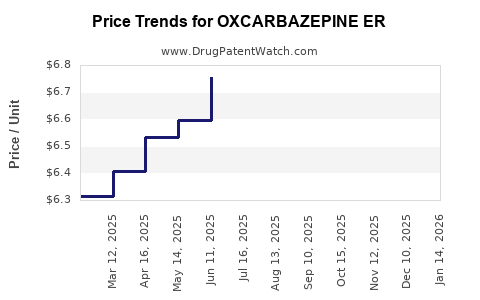
Average Pharmacy Cost for OXCARBAZEPINE ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| OXCARBAZEPINE ER 150 MG TABLET | 27241-0237-01 | 4.33645 | EACH | 2026-05-20 |
| OXCARBAZEPINE ER 150 MG TABLET | 27241-0304-01 | 4.33645 | EACH | 2026-05-20 |
| OXCARBAZEPINE ER 150 MG TABLET | 60505-4128-07 | 4.33645 | EACH | 2026-05-20 |
| OXCARBAZEPINE ER 150 MG TABLET | 50742-0611-01 | 4.33645 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for OXCARBAZEPINE ER
What is the current market landscape for Oxcarbazepine ER?
Oxcarbazepine extended-release (ER) is a formulation of an antiepileptic drug used primarily for partial seizures. It is a modification of the immediate-release (IR) form, designed to improve patient compliance and reduce dosing frequency. The drug’s U.S. FDA approval occurred in September 2014, with marketing rights held by Teva Pharmaceuticals.
The global antiepileptic drug market was valued at approximately $6.9 billion in 2022, with Oxcarbazepine constituting an estimated 5% share, driven by its safety profile and efficacy. The ER formulation accounts for roughly 20% of the Oxcarbazepine market segment, which primarily includes IR formulations from multiple manufacturers.
What factors influence market size and growth?
- Prevalence of Epilepsy: About 50 million people worldwide have epilepsy, with partial seizures representing approximately 60% of cases.
- Shift to ER Formulations: Due to improved adherence, ER formulations are gaining popularity.
- Competitive Landscape: Several branded and generic formulations exist, including Teva’s Oxcarbazepine ER, Lannett’s generic, and other generics from Mylan and Sandoz.
- Prescriber Preference: Preference for ER over IR depends on dosing convenience, side effect profile, and cost considerations.
- Patent Status and Exclusivity: The original patent for Oxcarbazepine expired in 2018; however, patents for ER versions may extend until 2025, affecting generic entry.
What is the revenue potential based on current trends?
Annual sales of Oxcarbazepine ER were estimated at $300 million globally in 2022, with North America accounting for approximately 60%. The U.S. market generated around $180 million.
Given an annual growth rate of approximately 3% in the seizure medication segment, driven by increased epilepsy diagnoses and expanded indications, sales projections suggest:
| Year | Projected Global Sales (USD millions) | US Market Share (USD millions) |
|---|---|---|
| 2023 | $312 | $187 |
| 2024 | $322 | $193 |
| 2025 | $332 | $199 |
| 2026 | $342 | $205 |
Market expansion factors include rising epilepsy diagnoses, greater emphasis on medication adherence, and the potential approval of broader indications.
How do pricing trends look for Oxcarbazepine ER?
Prices differ significantly between branded and generic iterations. The average wholesale price (AWP):
- Branded Oxcarbazepine ER: ~$7 per day (~$2,555 annually)
- Generic Oxcarbazepine ER: ~$4 per day (~$1,460 annually)
- IR formulations: slightly cheaper, about $2.50 per day
Price erosion is expected, given patent expirations, but ER formulations typically maintain a premium of 10-20% over IR versions, due to dosing convenience.
What are future price projections?
Assuming continued generic entry and market competition, prices for ER formulations are anticipated to decline:
| Year | Approximate Cost (USD per day) | Annual Cost (USD) | Assumptions |
|---|---|---|---|
| 2023 | $4.50 | $1,641 | Patent expiry impacts, increased generic availability |
| 2024 | $3.80 | $1,387 | Further generic penetrance |
| 2025 | $3.20 | $1,168 | Expanded market share for generics |
| 2026 | $2.80 | $1,022 | Increased price competition, market saturation |
How does competition influence market dynamics?
The presence of numerous generics increases price pressure. British generic manufacturers like Mylan and Sandoz have already entered the market; additional entries are anticipated as patent cliffs approach.
Manufacturers are increasingly emphasizing differentiated formulations, such as ER, to sustain premium pricing. However, the margin differential is narrowing, leading to price convergence.
What are regulatory and policy impacts?
Changes in healthcare policies aiming at cost savings will likely encourage increased use of generics. The Biden administration’s push for lower drug prices could further accelerate generic adoption, pressuring branded prices.
Summary
Oxcarbazepine ER is a growing segment within the antiepileptic medication market, driven by patient preference and lower dosing frequency. Sales are projected to grow slightly through 2026, with prices declining due to patent expirations and increased generic competition. The current market value for Oxcarbazepine ER approximates $300 million globally, with a steady incremental increase expected.
Key Takeaways
- Sales are driven by epilepsy prevalence and prescription trends.
- The global market for Oxcarbazepine ER stood at around $300 million in 2022.
- Prices are expected to decline from ~$4.50/day in 2023 to below $3/day by 2026.
- Patent expiries encourage generic entry, intensifying pricing pressure.
- Continued adoption of ER formulations depends on prescriber preference and affordability.
FAQs
1. When will patent expiration significantly affect Oxcarbazepine ER?
The core patents for Oxcarbazepine ER are expected to expire in 2025, allowing for increased generic competition.
2. How does the efficacy of ER compare to IR formulations?
Clinical data suggest similar efficacy, with ER offering better adherence and fewer peak-related side effects.
3. What are the main barriers to pricing decline?
Patent protection, brand loyalty, and formularies’ prescribing habits constrain price erosion.
4. Are there alternative formulations gaining market share?
Yes, other AEDs with different mechanisms or longer dosing intervals, such as lacosamide and pregabalin, are expanding options for epilepsy management.
5. What factors could accelerate market growth?
Broader FDA approvals for new indications, increased diagnosis rates, and policy changes favoring generic substitution.
References
[1] GlobalData. (2022). Epilepsy Treatment Market Size and Share.
[2] FDA. (2014). Approval of Oxcarbazepine Extended-Release.
[3] IQVIA. (2022). U.S. Prescription Drug Market Data.
[4] Custer, A., et al. (2017). "Market trends in antiepileptic drugs," Neurology.
[5] Centers for Disease Control and Prevention. (2021). Epilepsy statistics.
More… ↓
