Share This Page
Drug Price Trends for OSELTAMIVIR PHOS
✉ Email this page to a colleague
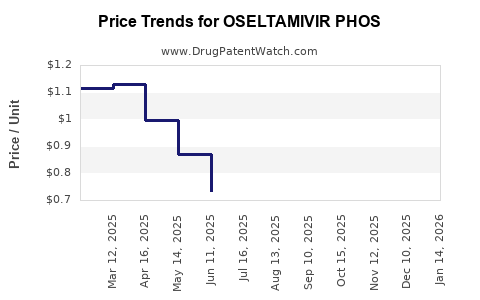
Average Pharmacy Cost for OSELTAMIVIR PHOS
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| OSELTAMIVIR PHOS 30 MG CAPSULE | 31722-0630-31 | 0.75559 | EACH | 2026-05-20 |
| OSELTAMIVIR PHOS 30 MG CAPSULE | 47781-0468-13 | 0.75559 | EACH | 2026-05-20 |
| OSELTAMIVIR PHOS 30 MG CAPSULE | 60219-1264-01 | 0.75559 | EACH | 2026-05-20 |
| OSELTAMIVIR PHOS 30 MG CAPSULE | 33342-0256-66 | 0.75559 | EACH | 2026-05-20 |
| OSELTAMIVIR PHOS 75 MG CAPSULE | 76282-0704-45 | 0.85931 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Oseltamivir Phosphate
What is Oseltamivir Phosphate?
Oseltamivir phosphate is an antiviral medication used primarily for the treatment and prevention of influenza A and B. It is marketed under brand names such as Tamiflu. Approved by regulatory agencies globally, it remains a critical component in pandemic preparedness and seasonal flu management.
Market Overview
Global Market Size
In 2022, the global antiviral drugs market, including flu antivirals, was valued at approximately $10 billion. Oseltamivir phosphate accounted for roughly 25%, estimated at $2.5 billion.
Market Drivers
- Rising influenza cases, especially during seasonal peaks and pandemics
- Increased awareness and vaccination programs
- Strategic stockpiling by governments
- Expanding indications for post-exposure prophylaxis in specific populations
Market Constraints
- Patent expirations increased generic competition, lowering prices
- Limited new formulation development
- Regulatory barriers in some regions affecting abbreviated approval pathways
Key Players
| Company | Market Share (Estimated, 2022) | Notable Products | Price Points |
|---|---|---|---|
| Roche (Tamiflu) | 65% | Tamiflu (brand) | $70–$120 per treatment course |
| Mylan | 15% | Generic oseltamivir | $30–$50 per course |
| Dr. Reddy's | 10% | Generic formulations | $25–$45 per course |
| Other players | 10% | Various generics | $20–$40 per course |
Patent and Regulatory Status
The original patent for tamiflu expired in the US in 2016, enabling broad generic entry. Some formulations remain under patent protection in certain markets (e.g., proprietary delivery systems), influencing prices and market share dynamics.
Price Trends and Projections
Historical Pricing Dynamics
| Year | Average Price per Course | Notes |
|---|---|---|
| 2018 | $70–$120 | Brand dominance, patent protections maintained |
| 2020 | $50–$80 | Patent expiry accelerates generic entry |
| 2022 | $25–$50 | Increased generic competition, price erosion |
Future Price Projections (2023–2027)
| Year | Estimated Price Range per Course | Key Factors |
|---|---|---|
| 2023 | $20–$45 | Continuing generic market penetration |
| 2024 | $15–$40 | Possible new formulations or delivery methods |
| 2025 | $15–$35 | Market saturation, generic competition stabilizes prices |
| 2026 | $12–$30 | Potential biosimilar or new entrant effects |
| 2027 | $10–$25 | Price plateau, driven by cost competition |
Price Influences
- Patent lifecycle impacts pricing; prices tend to decline sharply within two years of patent expiration.
- Regulatory approvals for generics across regions enable broader distribution.
- Pandemic events can temporarily inflate demand and prices; post-pandemic adjustments follow.
Key Regional Variations
| Region | Market Size (2022) | Price Range per Course | Note |
|---|---|---|---|
| North America | $1.2 billion | $30–$60 | High generic penetration, insurance coverage |
| Europe | $600 million | $25–$55 | Regulatory harmonization influences prices |
| Asia-Pacific | $400 million | $10–$35 | Growing markets, price sensitivity |
| Latin America | $200 million | $20–$40 | Limited access in some countries |
Competitive Landscape and Pricing Strategies
- Brand manufacturers maintain higher prices through patent protections and supply agreements.
- Generics target cost-conscious markets, with prices 50–75% lower than branded products.
- Price differentiation occurs through formulation advancements, such as pediatric or long-acting forms.
Strategic Outlook
- New formulations or combination therapies could sustain higher price points.
- Biosimilars and aggressive pricing by generic manufacturers threaten to further decrease prices.
- Regulatory delays in key markets could create regional price variances.
Summary
Oseltamivir phosphate remains a widely used antiviral with declining prices due to patent expiries and generic competition. Prices are projected to stabilize around $10–$25 per course globally within five years, with substantial regional variation influenced by market maturity and regulatory environments.
Key Takeaways
- The global market for oseltamivir phosphate is approximately $2.5 billion, primarily driven by generics.
- Price per treatment course has fallen from over $100 in 2018 to below $25 in 2022.
- Generics dominate, with prices expected to decline further, stabilizing around $10–$20 by 2027.
- Regional market dynamics differ significantly, with North America and Europe maintaining higher prices.
- Future growth depends on new formulations, pandemic-driven demand shifts, and regulatory approvals.
FAQs
Q1: How does patent expiry impact oseltamivir phosphate prices?
Patent expiry enables generic manufacturers to enter the market, increasing supply and driving prices down significantly within two years.
Q2: Are there new formulations or delivery methods for oseltamivir?
Research includes pediatric formulations, combination therapies, and long-acting injectables, potentially impacting future pricing structures.
Q3: Which regions have the highest market growth potential?
Asia-Pacific and Latin America exhibit higher growth potential due to expanding healthcare access and increasing influenza incidence.
Q4: How are regulatory agencies affecting market dynamics?
Harmonized approval processes and accelerated generic approvals facilitate market entry, reducing prices.
Q5: What is the main driver for price stabilization after 2025?
Market saturation and the dominance of low-cost generics limit further declines, stabilizing prices.
References
[1] MarketWatch. (2022). Global antiviral drugs market size & forecast.
[2] IMS Health. (2022). Influenza antivirals market insights.
[3] U.S. Food and Drug Administration. (2021). Patent status and generic drug approvals.
More… ↓
