Share This Page
Drug Price Trends for ORLISTAT
✉ Email this page to a colleague
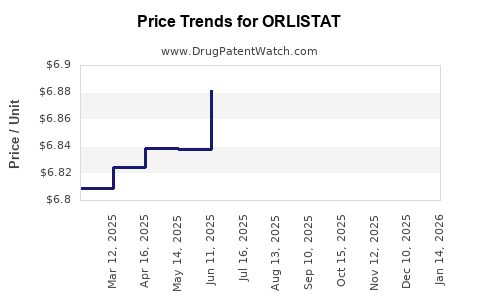
Average Pharmacy Cost for ORLISTAT
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ORLISTAT 120 MG CAPSULE | 61269-0565-90 | 6.85133 | EACH | 2026-04-22 |
| ORLISTAT 120 MG CAPSULE | 61269-0565-90 | 6.87372 | EACH | 2026-03-18 |
| ORLISTAT 120 MG CAPSULE | 61269-0565-90 | 6.90141 | EACH | 2026-02-18 |
| ORLISTAT 120 MG CAPSULE | 61269-0565-90 | 6.90511 | EACH | 2026-01-21 |
| ORLISTAT 120 MG CAPSULE | 61269-0565-90 | 6.90669 | EACH | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for ORLISTAT
Overview
Orlistat is a lipase inhibitor prescribed for weight management and obesity-related comorbidities. Market demand is driven by obesity prevalence, regulatory approvals, and healthcare reimbursement policies globally. The global weight-loss market, including prescription drugs like Orlistat, exceeded USD 20 billion in 2022, with a compound annual growth rate (CAGR) of approximately 4% expected through 2030.
Regulatory Status
Orlistat is approved in multiple regions under various brand names: Xenical (prescription) and Alli (over-the-counter). In the US, the FDA approved Orlistat in 1999. The European Medicines Agency (EMA) approved it in 1998. Regulatory agency decisions influence market access and pricing strategies, with recent shifts toward OTC availability impacting market size and sales volume.
Market Segmentation
- By Formulation: Prescription (Xenical, 120 mg), OTC (Alli, 60 mg)
- By Age Group: Adults, adolescents
- By Geography: North America, Europe, Asia-Pacific, Rest of World
Market Drivers
- Rising obesity rates: Global adult obesity prevalence reached approximately 13% in 2016, with projections surpassing 15% by 2030 (WHO).
- Growing awareness of weight management health risks.
- Healthcare reimbursement policies favoring obesity treatments.
Market Restraints
- Side effects such as gastrointestinal discomfort, malabsorption of fat-soluble vitamins.
- Availability of alternative weight management drugs.
- Consumer preference shifting toward combination therapies and surgical options.
Competitive Landscape
Orlistat faces competition from other pharmacological agents like phentermine, liraglutide, and bupropion-naltrexone combinations. Some generic formulations have entered the market, pressuring branded prices.
Price Trends and Projections
| Region | Current Average Price (USD) per Month | Notes |
|---|---|---|
| US (Prescription) | USD 300 – USD 400 | Branded, generic options vary in price. |
| US (OTC) | USD 25 – USD 30 (per pack of 60 capsules) | Lower cost, typical for OTC formulations. |
| Europe (Prescription) | EUR 250 – EUR 350 | Prices differ by country and reimbursement policies. |
| Asia-Pacific | USD 50 – USD 150 | Prices are lower; generic forms dominant. |
Note: Prices vary based on brand, generic availability, and health system reimbursement levels.
Price Projections (2023-2030)
- The market for Orlistat is expected to grow modestly, driven primarily by OTC sales expansion and increasing obesity rates.
- Unit prices for branded products are projected to decline by approximately 10-15% annually due to generic competition and increased market competition.
- OTC product prices are likely to decline as new generics enter the market, with average prices falling 5-10% annually.
- Overall, the global revenue for Orlistat is expected to increase at a CAGR of 2-3%, reaching approximately USD 1.2 billion by 2030, driven by volume growth rather than price increases.
Factors Impacting Future Prices
- Patent expiry: The original patent for Xenical expired in 2017 in most markets, increasing generic availability.
- Regulatory changes: Potential reclassification of prescription Orlistat as OTC in more markets could lower prices but increase volume sales.
- Market entry: New formulations or combination therapies may shift demand and influence prices.
- Healthcare policies: Reimbursement adjustments can constrain or boost retail prices.
Key Industry Players
- Roche (Xenical) – original patent holder, now generic versions dominate.
- GlaxoSmithKline and Teva – leading producers of OTC brands.
- Mylan, Sandoz – generic manufacturers.
Conclusion
The Orlistat market exhibits modest growth with declining unit prices owing to generic competition. Volume increases partly offset declining prices, sustaining overall revenue growth. Price sensitivity varies between high-income countries with reimbursement systems supporting branded products and emerging markets where generics prevail.
Key Takeaways
- The global market for Orlistat is growing slowly amid intensified generic competition.
- Prices are forecasted to decline 5-15% annually, depending on region and formulation type.
- Volumes are expected to increase from obesity prevalence and OTC availability.
- Patent expiries and regulatory shifts will significantly influence pricing strategies.
- The market size could reach USD 1.2 billion by 2030, with volume-driven growth compensating for price drops.
FAQs
1. How does patent expiration affect Orlistat prices?
Patent expiry enables generic manufacturers to produce lower-cost versions, increasing competition and reducing prices for both prescription and OTC formulations.
2. What factors could increase Orlistat’s market share?
Increased obesity prevalence, wider OTC availability, and favorable healthcare policies could raise overall sales volumes despite declining unit prices.
3. Are there significant risks to Orlistat’s market growth?
Yes. Market risks include regulatory reclassification as OTC, competing drugs with better efficacy or fewer side effects, and consumer preference shifts.
4. How do regional differences impact pricing?
In wealthier markets with strict reimbursement policies, prices are higher; in emerging markets with limited reimbursement, prices are lower, driven primarily by generics.
5. What is the outlook for novel formulations or combination therapies?
Innovations combining Orlistat with other weight management agents could either replace or supplement existing products, influencing price and market dynamics.
Sources
- World Health Organization (WHO). Obesity and overweight statistics.
- MarketWatch. Weight-loss market size and forecasts.
- European Medicines Agency (EMA). Regulatory approvals and decisions.
- Pharma intelligence reports on generic drug pricing trends.
- Company filings and press releases from Roche, GSK, and Teva.
More… ↓
