Share This Page
Drug Price Trends for OPZELURA
✉ Email this page to a colleague
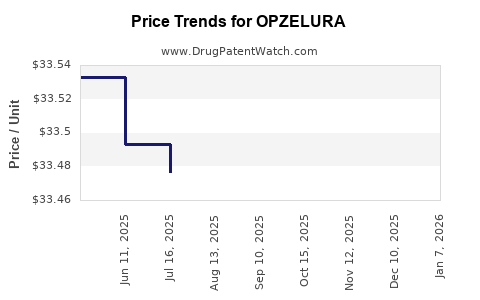
Average Pharmacy Cost for OPZELURA
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| OPZELURA 1.5% CREAM | 50881-0007-05 | 34.14330 | GM | 2026-01-01 |
| OPZELURA 1.5% CREAM | 50881-0007-05 | 33.47186 | GM | 2025-12-17 |
| OPZELURA 1.5% CREAM | 50881-0007-05 | 33.47252 | GM | 2025-11-19 |
| OPZELURA 1.5% CREAM | 50881-0007-05 | 33.48483 | GM | 2025-10-22 |
| OPZELURA 1.5% CREAM | 50881-0007-05 | 33.48477 | GM | 2025-09-17 |
| OPZELURA 1.5% CREAM | 50881-0007-05 | 33.48172 | GM | 2025-08-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Opzelura (Ruxolitinib Cream)
Overview
Opzelura (ruxolitinib cream) is a topical Janus kinase (JAK) inhibitor approved by the FDA in 2021 for treatment of mild to moderate atopic dermatitis. The drug is produced by Incyte Corporation, marketed through its partnership with Pfizer. As a first-in-class topical JAK inhibitor, Opzelura targets a significant dermatological market with unmet needs.
Market Size and Growth Drivers
The atopic dermatitis market globally was valued at approximately $4.2 billion in 2022. The U.S. represents a major share, estimated at $2.5 billion. The disease affects roughly 10-20% of children and 7-10% of adults worldwide, with increasing incidence in recent years.
Key drivers include:
- Growing prevalence of atopic dermatitis
- Limited efficacy of conventional topical corticosteroids and calcineurin inhibitors
- Rising patient demand for targeted, steroid-sparing therapies
- Approval of novel JAK inhibitors for dermatological conditions
Additional market segments involve other inflammatory skin diseases like vitiligo and alopecia areata, which are potential expansion areas for Opzelura.
Competitive Landscape
Opzelura faces competition from:
- Dupixent (dupilumab): injected biologic for moderate-to-severe atopic dermatitis; 2022 sales of ~$4.3 billion
- Eucrisa (crisaborole): topical phosphodiesterase-4 inhibitor; limited to mild cases
- Other JAK inhibitors in systemic form, such as Xeljanz (tofacitinib) and Eliquis (baricitinib), primarily for systemic use, which influence topical's market penetration
Despite competition, topical JAK inhibitors pose advantages due to non-invasiveness and potential for early intervention.
Pricing Analysis
Initial U.S. prescribing information set Opzelura's list price at approximately $1,250 to $1,350 per jar (60g supply). This pricing aligns with existing topical dermatological agents, but is higher than traditional corticosteroids, justified by its targeted mechanism and localized delivery.
Insurance negotiations and pharmacy benefit managers (PBMs) influence actual patient out-of-pocket costs, which may range between $50 and $150 monthly for insured patients. The high list price underscores focus on premium pricing strategies driven by clinical differentiation.
Price Trends and Projections
Sales began ramping in 2022, with Incyte reporting $132 million in U.S. net product revenue by Q2 2023. The market is expected to expand as the drug gains recommended positioning.
Projected price trajectories consider:
- Competitive pressure: Entry of biosimilars or generics for systemic JAK drugs could drive down prices of systemic alternatives but not affect topical pricing directly
- Market saturation: As prescription rates grow, economies of scale might allow slight reductions
- Payer negotiations: As formulary coverage improves, list prices may be moderated to improve access
Forecasting a 5% annual price decrease over the next five years is plausible, given typical competitive dynamics and inflation-adjusted pricing strategies in dermatology.
Market Penetration and Revenue Projections
Incyte targets capturing 15-20% of the atopic dermatitis topical segment within five years. Assuming a conservative annual growth rate of 10-15% in sales volume, revenue from Opzelura could reach approximately $600 million to $1 billion by 2027. Pricing remains a key variable, with potential adaptations based on payer contracts and market demand.
Regulatory and Policy Factors
Medicare and Medicaid rebate policies influence net price realizations. The Inflation Reduction Act of 2022 increases scrutiny on high drug prices, possibly leading to tighter negotiations and discounts.
Summary
Opzelura's market sits within a growing, competitive dermatology space with high-value clinical positioning. Its premium pricing strategy is supported by clinical benefits and specificity, with potential reductions driven by market competition, payer strategies, and scaling efficiencies.
Key Takeaways
- Opzelura addresses a high-growth segment in atopic dermatitis with a forecasted 2027 revenue reaching up to $1 billion.
- The initial list price is approximately $1,250–$1,350 per jar, with actual patient costs varying by insurance.
- Price decreases of around 5% annually are expected due to competitive and policy pressures.
- Market capture depends on formulary placement, prescriber adoption, and patient access initiatives.
- Systemic JAK inhibitors represent both a competitive threat and potential market expansion for topical JAK inhibitors.
FAQs
-
What factors influence Opzelura’s pricing strategy?
- Clinical differentiation, competitive landscape, insurance negotiations, manufacturing costs, and policy regulations.
-
How does Opzelure compare price-wise to injectable biologics like Dupixent?
- Opzelura’s annual outpatient cost is lower, around several hundred dollars monthly, compared to Dupixent’s high-cost injections exceeding $30,000 annually.
-
When might we see price reductions or discounts?
- Upon increased market penetration, payer negotiations, generic entries for systemic JAK drugs, and policy changes.
-
What is the potential for global market expansion?
- Regulatory approvals are pending in various regions, but pricing and reimbursement policies will significantly influence adoption.
-
What are the main revenue risks for Opzelura?
- Competition from systemic JAK inhibitors and biologics, insurance coverage limitations, and regulatory price controls.
Sources
[1] Incyte Corporation, 2023 Financial Reports
[2] Evaluate Pharma, 2022 Dermatology Market Data
[3] U.S. Food and Drug Administration, 2021 Approval Announcement
[4] IQVIA, 2023 Topical Dermatology Sales Data
[5] CMS and PBM policy documents, 2022-2023
More… ↓
