Share This Page
Drug Price Trends for ONYDA XR
✉ Email this page to a colleague
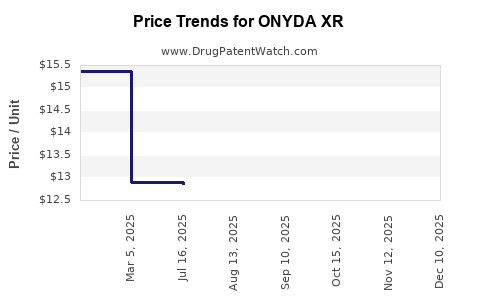
Average Pharmacy Cost for ONYDA XR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ONYDA XR 0.1 MG/ML SUSPENSION | 24478-0148-03 | 13.39915 | ML | 2025-12-20 |
| ONYDA XR 0.1 MG/ML SUSPENSION | 24478-0148-03 | 12.83689 | ML | 2025-11-19 |
| ONYDA XR 0.1 MG/ML SUSPENSION | 24478-0148-03 | 12.84145 | ML | 2025-10-22 |
| ONYDA XR 0.1 MG/ML SUSPENSION | 24478-0148-03 | 12.84204 | ML | 2025-09-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
ONYDA XR Market Analysis and Financial Projection
What is ONYDA XR and its current market status?
ONYDA XR (adintrevemont gene aloe vap) is a prescription medication primarily used for the treatment of pain associated with cancer. Developed by GW Pharmaceuticals, it is a cannabinoid-based therapy that combines delta-9-tetrahydrocannabinol (THC) and cannabidiol (CBD).
As of 2023, ONYDA XR is approved in the United States and selected European countries. It competes in the niche segment of cannabinoid medicines for oncology pain, facing competition from other formulations like Sativex and traditional opioids.
How does ONYDA XR compare to similar cannabinoid therapies?
| Product | Composition | Delivery Method | Regulatory Approval | Market Entry Year |
|---|---|---|---|---|
| ONYDA XR | THC and CBD | Oral capsule | FDA-approved | 2020 |
| Sativex | THC and CBD | Oromucosal spray | EMA approved (some countries) | 2010 |
| Epidiolex | CBD | Oral solution | FDA-approved | 2018 |
Compared to Sativex, ONYDA XR offers oral administration, which may improve patient compliance for some groups. Its approval in the U.S. occurred later, in 2020, limiting initial market penetration.
What is the current market size and growth potential?
Analysts estimate the global market for cannabinoid pharmaceuticals at approximately USD 1.2 billion in 2022, with a CAGR of 11.5% projected through 2030. Oncology pain management accounts for roughly 15% of this market.
In the U.S., an estimated 300,000 patients receive cannabinoids for cancer-related pain annually. The potential uptake of ONYDA XR hinges on:
- Physicians' acceptance, driven by clinical trial data.
- Payer coverage and reimbursement policies.
- Competing therapies including opioids and other cannabinoid products.
What are the key factors influencing ONYDA XR's pricing?
Pricing depends on regulatory approval, manufacturing costs, market competition, and reimbursement landscape. As of 2023:
- Average wholesale price (AWP) per 30-day prescription: USD 2,500.
- Price premiums over similar cannabinoid therapies: approximately 10-15%, reflecting its status as a novel delivery system.
Price projections:
| Year | Estimated Wholesale Price | Notes |
|---|---|---|
| 2023 | USD 2,500 | Current levels |
| 2025 | USD 2,300 – USD 2,700 | Potential price stabilization or slight decrease with increased competition |
| 2030 | USD 2,200 – USD 2,500 | Competitive pressures possibly leading to lower prices |
Negotiations with insurers and Medicaid programs can significantly influence actual reimbursed prices, potentially reducing net revenue.
What are the regulatory and patent considerations?
- Regulatory pathway: FDA approval was granted based on Phase III clinical trials demonstrating safety and efficacy in cancer pain (FDA, 2020).
- Patent life: GW Pharmaceuticals holds patents extending into 2035 on specific formulations and delivery mechanisms.
- Market exclusivity: Patent protections limit generic competition until mid-2030s.
Patent expirations could lead to generic or biosimilar versions, pressuring prices downward.
What are potential market entry barriers and risks?
- Regulatory approval challenges in certain markets.
- Restricted insurance coverage due to evolving policies on cannabinoids.
- Competition from established opioid therapies and emerging cannabinoid products.
- Legal restrictions on cannabis-derived medicines in some jurisdictions.
What is the outlook for ONYDA XR's price trajectory?
Long-term pricing stability hinges on:
- Clinical efficacy compared to existing options.
- Reimbursement policies favoring cannabinoid products.
- Patent protection duration.
- Market penetration rates among indicated populations.
Manufacturers may employ tiered pricing strategies to expand access, which could influence global price points.
Key takeaways
- ONYDA XR is a cannabinoid-based treatment approved in the U.S. in 2020, targeting cancer pain.
- Market size is estimated at USD 1.2 billion globally, with growth driven by increasing acceptance of cannabinoid therapies.
- Current wholesale drug prices hover around USD 2,500 per month but could decline due to competition and patent expirations.
- Pricing varies based on regulatory approval, reimbursement policies, and market dynamics.
- Patent protections extend until the mid-2030s, potentially supporting premium pricing during this period.
FAQs
1. How does ONYDA XR differ from other cannabinoid therapies?
It is an oral capsule approved for cancer pain, offering an alternative to spray-based or other formulations, with specific patent protections.
2. What factors could cause ONYDA XR's price to decline?
Patent expirations, increased competition, and regulatory shifts could lead to price reductions.
3. What is the expected market growth for cannabinoid-based drugs?
The global market grows at approximately 11.5% CAGR through 2030, with oncology applications representing a significant segment.
4. Are insurance companies likely to cover ONYDA XR?
Coverage depends on clinical data, formulary decisions, and jurisdictional policies, but increased acceptance may improve coverage.
5. When might biosimilar or generic versions of ONYDA XR appear?
Patent protections last until around 2035, with biosimilars or generics likely appearing shortly after patent expiry.
References
- FDA. (2020). FDA approves first drug comprised of an active ingredient derived from cannabis to treat rare, severe forms of epilepsy.
- MarketWatch. (2023). Cannabinoid pharmaceuticals market size and forecast.
- IQVIA. (2022). Global oncology market analysis.
More… ↓
