Share This Page
Drug Price Trends for ONDANSETRON ODT
✉ Email this page to a colleague
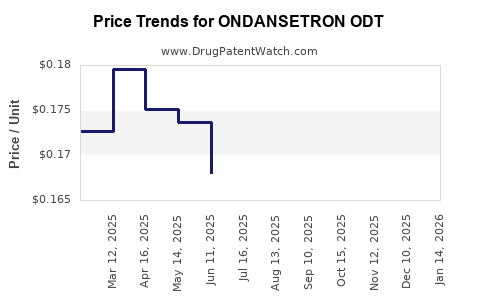
Average Pharmacy Cost for ONDANSETRON ODT
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| ONDANSETRON ODT 4 MG TABLET | 16714-0200-30 | 0.13346 | EACH | 2026-05-20 |
| ONDANSETRON ODT 4 MG TABLET | 57237-0077-30 | 0.13346 | EACH | 2026-05-20 |
| ONDANSETRON ODT 4 MG TABLET | 57237-0077-50 | 0.13346 | EACH | 2026-05-20 |
| ONDANSETRON ODT 4 MG TABLET | 57237-0077-10 | 0.13346 | EACH | 2026-05-20 |
| ONDANSETRON ODT 8 MG TABLET | 83980-0013-13 | 0.17517 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
ONDANSETRON ODT: Market Dynamics and Price Forecast
Ondansetron orally disintegrating tablet (ODT) formulations offer improved patient compliance and convenience, particularly for individuals with difficulty swallowing. This analysis projects market growth driven by increasing prevalence of chemotherapy-induced nausea and vomiting (CINV) and radiotherapy-induced nausea and vomiting (RINV), alongside a stable generic competitive landscape.
What is the current market size and projected growth for Ondansetron ODT?
The global market for Ondansetron ODT is estimated to be approximately $250 million in 2023, with a projected Compound Annual Growth Rate (CAGR) of 4.2% over the next five years, reaching an estimated $308 million by 2028. This growth is primarily influenced by an expanding patient pool requiring antiemetic therapy.
Key market drivers include:
- Oncology Treatments: The increasing incidence of cancer diagnoses globally necessitates more aggressive chemotherapy and radiotherapy regimens, directly correlating with higher demand for effective antiemetics like Ondansetron ODT. The World Health Organization (WHO) reported an estimated 19.3 million new cancer cases in 2020, a figure projected to rise.
- Gastrointestinal Disorders: While not the primary indication, Ondansetron is also utilized off-label for nausea and vomiting associated with various gastrointestinal conditions, contributing to a broader market base.
- Convenience of ODT Formulation: The ODT format eliminates the need for water and is ideal for patients experiencing severe nausea or vomiting who cannot tolerate oral liquids or solid tablets. This patient-centric advantage underpins its sustained adoption.
- Generic Competition: Ondansetron is a well-established generic drug. The ODT formulation represents a value-added generic, allowing manufacturers to command a premium over standard oral tablets while still maintaining affordability compared to branded alternatives.
What are the key therapeutic areas and patient populations driving demand?
The primary therapeutic areas driving demand for Ondansetron ODT are oncology and, to a lesser extent, general medical applications involving nausea and vomiting.
Oncology Applications
- Chemotherapy-Induced Nausea and Vomiting (CINV): Ondansetron is a first-line agent for preventing and treating CINV. The intensity and unpredictability of CINV significantly impact patient quality of life and treatment adherence. ODT formulations are particularly beneficial for patients undergoing highly emetogenic chemotherapy. The American Cancer Society estimates that over 1.8 million new cancer cases will be diagnosed in the United States in 2023.
- Radiotherapy-Induced Nausea and Vomiting (RINV): Similar to CINV, RINV can be debilitating and affect treatment tolerance. Ondansetron ODT offers a convenient option for patients receiving radiation therapy, especially for head and neck or abdominal cancers.
Other Medical Applications
- Postoperative Nausea and Vomiting (PONV): While other antiemetics are frequently used post-surgery, Ondansetron ODT can be an option for patients requiring rapid relief and who have difficulty swallowing.
- Gastroenteritis and Migraine-Associated Nausea: Off-label use for severe nausea associated with viral gastroenteritis or migraines contributes a smaller but consistent segment of demand.
The patient population includes individuals of all ages undergoing cancer treatment, post-surgical patients, and individuals experiencing acute episodes of severe nausea. The elderly population, who may have dysphagia, particularly benefit from the ODT formulation.
What is the competitive landscape for Ondansetron ODT?
The competitive landscape for Ondansetron ODT is characterized by robust generic competition, with several established pharmaceutical manufacturers offering the product. Differentiation primarily occurs through formulation technology, packaging, and market access strategies rather than novel patent protections.
Key Manufacturers and Market Share
Major generic manufacturers offering Ondansetron ODT include:
- Teva Pharmaceutical Industries Ltd.
- Sun Pharmaceutical Industries Ltd.
- Dr. Reddy's Laboratories Ltd.
- Aurobindo Pharma Ltd. (now Eugia Pharma)
- Mylan N.V. (now part of Viatris Inc.)
- Hikma Pharmaceuticals PLC
These companies compete on price, product availability, and supply chain reliability. Market share is fragmented, with no single entity dominating due to the generic nature of the product.
Formulation Technology and Intellectual Property
While the active pharmaceutical ingredient (API) Ondansetron is off-patent, specific ODT formulations may have process patents or patents related to excipients that enhance disintegration or taste masking. However, these patents are generally nearing expiry or have expired, allowing for broader generic entry.
- Orally Disintegrating Tablet Technology: This involves specialized excipients and manufacturing processes to ensure rapid dissolution in the mouth without water. Manufacturers invest in optimizing these technologies for better patient acceptance (e.g., taste, texture).
- Exclusivity: Generic manufacturers may benefit from periods of market exclusivity upon patent expiry of branded ODT products, though this is typically limited to 180 days in the U.S. and subject to litigation.
Pricing Strategies
Pricing is highly competitive, driven by tender processes in institutional settings (hospitals, clinics) and by pharmacy benefit manager (PBM) formulary negotiations in the retail sector. Manufacturers aim to achieve cost-effective production to remain competitive.
What are the projected price trends for Ondansetron ODT?
The price trends for Ondansetron ODT are expected to remain stable with a slight downward pressure due to ongoing generic competition and formulary management by healthcare payers.
Historical Pricing
Since the patent expiry of the originator ODT product, prices have steadily declined. The average selling price (ASP) for Ondansetron ODT 4mg, in a 10-count bottle, has fallen from an initial premium over standard tablets to approximately $15-$25, depending on the manufacturer and channel.
Projected Price Trends (2024-2028)
- Year-over-Year Decline: Expect a modest annual price decrease of 1-2% driven by competitive pressures and volume-based purchasing by large healthcare systems.
- Formulation Premium: Ondansetron ODT will continue to command a premium of 15-25% over standard oral Ondansetron tablets due to the added value of the ODT technology. This premium is expected to remain relatively consistent as the technology is established and widely adopted by multiple generic players.
- Supply Chain and Raw Material Costs: Fluctuations in API manufacturing costs and global supply chain disruptions could introduce short-term price volatility. However, the availability of multiple API suppliers for Ondansetron mitigates significant long-term price increases.
- Payer Influence: Pharmacy benefit managers and government payers will continue to exert pressure on pricing through preferred drug lists and reimbursement policies, encouraging the use of the lowest-cost, therapeutically equivalent options.
Example Price Comparison (Hypothetical 2025 ASP)
- Ondansetron ODT (4mg, 10-count): $20-$23
- Ondansetron Standard Tablet (4mg, 10-count): $16-$19
This price differential reflects the manufacturing complexity and patient benefit of the ODT formulation.
What are the regulatory and patent considerations impacting the Ondansetron ODT market?
The regulatory landscape for Ondansetron ODT is relatively mature, with established pathways for generic approval. Patent considerations are primarily historical, focusing on the expiry of the active ingredient patent and any secondary patents related to formulation technologies.
Regulatory Approvals
- U.S. Food and Drug Administration (FDA): Generic Ondansetron ODT products must receive Abbreviated New Drug Application (ANDA) approval from the FDA. This requires demonstrating bioequivalence to the reference listed drug (RLD) and meeting quality standards.
- European Medicines Agency (EMA): Similar approval processes exist in European Union member states through the EMA or national competent authorities.
- Other Global Regulatory Bodies: Approvals are required from regulatory agencies in other key markets, such as Health Canada, Australia's Therapeutic Goods Administration (TGA), and Japan's Pharmaceuticals and Medical Devices Agency (PMDA).
Patent Expiry and Litigation
- API Patent: The fundamental patent for Ondansetron expired decades ago, opening the market to generic competition for standard oral dosage forms.
- Formulation Patents: Patents covering specific ODT technologies, such as those involving unique disintegrants, binders, or manufacturing processes, have been a source of market exclusivity for some time. However, many of these foundational ODT patents have expired or are nearing expiry.
- Paragraph IV Certifications: Generic manufacturers often challenge existing formulation patents through Paragraph IV certifications under the Hatch-Waxman Act, leading to potential litigation and earlier market entry. This has been a common strategy for Ondansetron ODT.
- Exclusivity Periods: Upon approval of an ANDA for a bioequivalent generic drug, a 180-day period of market exclusivity may be granted, preventing other generics from entering. This is a crucial factor for early market entrants.
Current Patent Landscape
As of 2023-2024, the primary patents related to the original Ondansetron ODT formulations are largely expired or nearing expiry, leading to a highly competitive generic market. New patents in this space are unlikely to significantly alter the market structure for Ondansetron ODT, as the focus has shifted to newer antiemetic agents with different mechanisms of action.
Key Takeaways
- The Ondansetron ODT market is projected to grow at a CAGR of 4.2% through 2028, driven by oncology demand and the ODT formulation's patient advantages.
- Key manufacturers are established generic pharmaceutical companies, leading to a fragmented market share and price competition.
- Pricing is expected to exhibit a slight downward trend, with Ondansetron ODT maintaining a premium over standard tablets due to its formulation.
- The regulatory and patent landscape is mature, with most fundamental patents expired, allowing for robust generic entry and competition.
FAQs
What is the difference in efficacy between Ondansetron ODT and standard Ondansetron tablets?
Efficacy is considered bioequivalent when comparing Ondansetron ODT to standard Ondansetron tablets, provided both are approved generic products. The primary difference lies in the route and speed of administration and patient convenience, not in the pharmacological effect or therapeutic outcome.
Are there any emerging trends or novel formulations for Ondansetron?
While Ondansetron is a mature drug, research continues into optimized ODT formulations focusing on improved taste masking, faster disintegration times, and more stable oral films. However, significant novel formulation breakthroughs are unlikely to disrupt the established generic market for ODTs in the near term.
How does the price of Ondansetron ODT compare to newer antiemetic drugs?
Ondansetron ODT is significantly less expensive than newer antiemetic drugs, such as NK1 receptor antagonists (e.g., aprepitant) or serotonin-dopamine antagonists. These newer agents often have broader spectrum efficacy for highly emetogenic chemotherapy and are used in combination regimens, commanding higher price points.
What is the typical duration of treatment with Ondansetron ODT for CINV?
Treatment duration varies based on the chemotherapy regimen's emetogenic potential and the patient's response. It can range from a few days to the entire duration of chemotherapy treatment, and sometimes extends into the recovery period for persistent nausea.
Which markets are expected to see the highest growth for Ondansetron ODT?
North America and Europe currently represent the largest markets due to advanced healthcare infrastructure and high cancer treatment rates. Emerging markets in Asia-Pacific and Latin America are expected to exhibit higher CAGRs due to increasing access to cancer care and adoption of improved drug formulations.
Citations
[1] World Health Organization. (2020). Globocan 2020: Estimated incidence, mortality and prevalence worldwide in 2020. International Agency for Research on Cancer.
[2] American Cancer Society. (2023). Cancer Facts & Figures 2023.
[3] U.S. Food and Drug Administration. (n.d.). Abbreviated New Drug Applications (ANDAs). Retrieved from [FDA website]
More… ↓
