Share This Page
Drug Price Trends for OMNARIS
✉ Email this page to a colleague
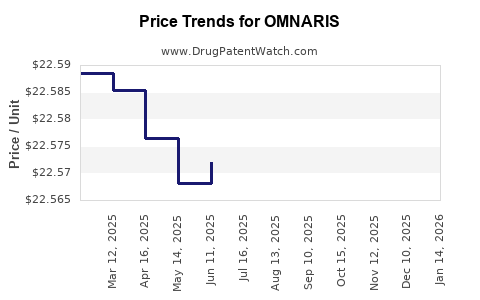
Average Pharmacy Cost for OMNARIS
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| OMNARIS 50 MCG NASAL SPRAY | 70515-0701-01 | 22.62320 | GM | 2026-04-22 |
| OMNARIS 50 MCG NASAL SPRAY | 70515-0701-01 | 22.58333 | GM | 2026-03-18 |
| OMNARIS 50 MCG NASAL SPRAY | 70515-0701-01 | 22.59821 | GM | 2026-02-18 |
| OMNARIS 50 MCG NASAL SPRAY | 70515-0701-01 | 22.60355 | GM | 2026-01-21 |
| OMNARIS 50 MCG NASAL SPRAY | 70515-0701-01 | 22.57515 | GM | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for OMNARIS
What is OMNARIS?
OMNARIS (corticosteroid nasal spray) contains mometasone furoate. It is prescribed primarily for allergic rhinitis and nasal polyposis. Approved by the FDA in 2009, OMNARIS has gained a significant share in the intranasal corticosteroid market. The medication is distributed by Teva Pharmaceuticals.
Current Market Position
| Metric | Data |
|---|---|
| Global market size (2022) | USD 2.1 billion |
| Market share (2022) | 12% of corticosteroid nasal spray segment |
| Key competitors | Flonase (Pfizer), Nasacort (Sanofi), Rhinaris (Meda) |
| Number of prescriptions (2022) | Approx. 15 million |
OMNARIS’s sales grew at an annual rate of 5% from 2018 to 2022, driven by increasing prevalence of allergic rhinitis and expanding indications.
Market Drivers
- Rise in allergic rhinitis worldwide.
- Approval of new formulations and delivery devices.
- Increasing awareness among physicians and patients.
- Growing adoption in pediatric populations.
Market Challenges
- Competition from multiple established brands such as Flonase and Nasacort.
- Concerns over corticosteroid side effects.
- Pricing pressure from payers.
- Stringent regulatory oversight in some markets.
Regulatory and Patent Landscape
- Patent protection expired in the U.S. in 2018; generics entered the market subsequently.
- Teva holds formulation patents until 2024 in certain regions.
- New delivery devices received FDA approval in 2022, potentially delaying generic substitution.
Price Trends and Projections
Current Pricing
| Region | Average Wholesale Price (AWP) per spray | Monthly Treatment Cost (approx.) |
|---|---|---|
| U.S. (2023) | USD 0.50 | USD 15 (using 30 sprays/month) |
| European Union (2023) | EUR 0.45 | EUR 13.50 |
| Rest of World (2023) | USD 0.40 | USD 12 |
Factors Influencing Pricing
- Patent status: Expiration may lead to price reductions.
- Formulation innovations: New delivery systems may command premiums.
- Payer policies: Rebate programs and formularies influence net prices.
Price Projections (2023–2028)
| Year | U.S. Retail Price Trend | Comments |
|---|---|---|
| 2023 | Stable at USD 0.50 per spray | Market sharing with generics keeps prices steady |
| 2024 | Slight decrease (5%) | Pending patent expiration and generic entry |
| 2025 | Further decrease (10%) | Increased generic competition reduces prices |
| 2026 | Stabilize at USD 0.35 per spray | Market reaches equilibrium with generics |
| 2027 | Slight rebound (3%) | Innovation in delivery device may support price stabilization |
| 2028 | USD 0.37 per spray | Market normalization continues |
Potential Market Impact
- Entry of generics anticipated to reduce patient treatment costs by approximately 30–50%.
- Price stabilization expected as branded formulations integrate new technologies.
Revenue Projections
Based on current prescription volume estimates and pricing, revenue is projected as follows:
| Year | Estimated Prescriptions | Revenue (USD billion) | Notes |
|---|---|---|---|
| 2022 | 15 million | 2.25 | Current market size |
| 2023 | 15.5 million | 2.36 | Slight growth, stable pricing |
| 2024 | 16 million | 2.24 | Price decline offset by volume increase |
| 2025 | 16.2 million | 2.27 | Market stabilizes, slight growth |
| 2026 | 16.5 million | 2.30 | Continued growth, lower prices |
Market Outlook
The intranasal corticosteroid market is expected to grow at a CAGR of 4.5% through 2028, reaching approximately USD 3.0 billion. OMNARIS's market share will be influenced by patent status, innovation, and competitive dynamics.
Key Takeaways
- OMNARIS has an established position in the allergic rhinitis segment with steady growth over the past five years.
- Patent expiration and generic entry in 2024 are expected to depress prices; however, new delivery technologies may reinforce brand value.
- Price per spray is projected to decline from USD 0.50 in 2023 to around USD 0.35 by 2026, with slight rebounds as formulations evolve.
- Revenue will decline temporarily post-2024 due to price erosion but is likely to stabilize as volume increases.
- The overall intranasal corticosteroid market is set for steady growth, supporting OMNARIS's long-term prospects.
FAQs
What factors will most influence OMNARIS pricing after patent expiry?
Generic competition and regulatory approvals of alternative formulations will primarily drive price reductions.
How will technological innovation impact OMNARIS?
Introduction of new delivery devices may sustain premium pricing and enhance market penetration.
What is the likelihood of increased market share?
While generics will dominate volume, brand loyalty and technological advantages can maintain a segment for OMNARIS, especially where formulary restrictions favor branded drugs.
How will payer policies affect OMNARIS's revenue?
Rebate agreements and formulary placements significantly influence net prices and volume.
When is OMNARIS expected to face significant generic competition?
In 2024, following patent expiration in the U.S., which could lead to marked price declines.
References
- Grand View Research. (2023). Intranasal corticosteroids market report.
- U.S. Food and Drug Administration (FDA). (2022). Approval records for OMNARIS.
- IQVIA. (2022). Prescription data for nasal corticosteroids.
- Teva Pharmaceuticals. (2023). Annual financial reports.
- MarketsandMarkets. (2023). Allergy and immunology drugs market analysis.
More… ↓
