Share This Page
Drug Price Trends for OMEPRAZOLE-BICARB
✉ Email this page to a colleague
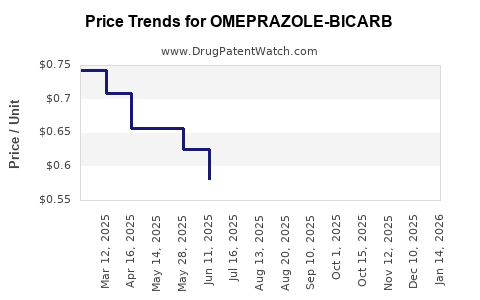
Average Pharmacy Cost for OMEPRAZOLE-BICARB
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| OMEPRAZOLE-BICARB 20-1,100 CAP | 69097-0913-02 | 0.54273 | EACH | 2026-05-20 |
| OMEPRAZOLE-BICARB 20-1,680 PKT | 64380-0182-01 | 7.60406 | EACH | 2026-05-20 |
| OMEPRAZOLE-BICARB 20-1,680 PKT | 27241-0029-31 | 7.60406 | EACH | 2026-05-20 |
| OMEPRAZOLE-BICARB 20-1,100 CAP | 69367-0195-30 | 0.54273 | EACH | 2026-05-20 |
| OMEPRAZOLE-BICARB 20-1,680 PKT | 27241-0029-62 | 7.60406 | EACH | 2026-05-20 |
| OMEPRAZOLE-BICARB 40-1,680 PKT | 70954-0798-20 | 9.53861 | EACH | 2026-05-20 |
| OMEPRAZOLE-BICARB 20-1,100 CAP | 27241-0031-03 | 0.54273 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Omeprazole-Bicarbonate: Market Dynamics and Price Outlook
Omeprazole-bicarbonate, a combination therapy for gastroesophageal reflux disease (GERD) and peptic ulcers, faces an evolving market landscape influenced by patent expirations, generic competition, and evolving treatment guidelines. The drug's efficacy in rapid symptom relief due to the immediate neutralization of gastric acid by bicarbonate, coupled with omeprazole's proton pump inhibition, positions it as a valuable therapeutic option. However, price projections are subject to significant downward pressure from a growing number of interchangeable generic versions.
What is the Current Market Size and Growth Projection for Omeprazole-Bicarbonate?
The global market for omeprazole-bicarbonate is substantial, driven by the high prevalence of acid-related gastrointestinal disorders. While specific market segmentation for the omeprazole-bicarbonate combination product is not always granularly reported, the broader proton pump inhibitor (PPI) market, of which omeprazole is a key component, provides a proxy. The global PPI market was valued at approximately USD 13.5 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 3.2% from 2023 to 2030, reaching an estimated USD 17.2 billion by 2030 [1]. The omeprazole-bicarbonate formulation benefits from this overall market trend due to its established efficacy and relatively favorable safety profile for certain patient populations requiring rapid acid suppression.
The growth is primarily attributed to:
- Increasing GERD and PUD Incidence: Lifestyle factors such as poor dietary habits, obesity, and stress contribute to a rising global incidence of GERD and peptic ulcer disease (PUD).
- Aging Population: Older adults are more susceptible to gastrointestinal issues, increasing the demand for effective treatments.
- Awareness and Diagnosis: Improved diagnostic tools and increased patient awareness of GERD symptoms are leading to higher diagnosis rates.
- Combination Therapy Advantages: The immediate relief offered by the bicarbonate component in omeprazole-bicarbonate formulations is a significant differentiator for patients experiencing acute symptoms.
However, the growth of the omeprazole-bicarbonate segment is tempered by the availability of numerous generic omeprazole products and alternative acid suppression therapies.
Which Patents Are Key to Omeprazole-Bicarbonate and What Are Their Expiration Timelines?
The patent landscape for omeprazole-bicarbonate is complex, involving patents on the active pharmaceutical ingredients (APIs), specific formulations, and methods of use. The original patents for omeprazole have long expired, allowing for widespread generic entry. However, patents protecting specific combinations, such as omeprazole with sodium bicarbonate, or novel delivery systems, have extended market exclusivity for certain branded products.
- Omeprazole API Patents: The foundational patents for omeprazole, originally developed by AstraZeneca, expired in the early 2000s. This opened the door for generic omeprazole production worldwide.
- Formulation Patents: Patents related to the specific combination of omeprazole with bicarbonate to enhance dissolution and absorption, or to create a fast-acting formulation, have been critical for branded products. For example, Zegerid (omeprazole/sodium bicarbonate) has had a patent portfolio that included patents on its unique formulation.
- U.S. Patent No. 6,521,256 (for a delayed-release formulation of omeprazole with an alkaline agent) was a significant patent for such combination products. This patent expired in 2023 [2].
- Other formulation patents may cover specific ratios of omeprazole to bicarbonate, granulation techniques, or excipient combinations designed to improve stability or patient compliance. The expiration dates of these patents vary, but many key ones have already lapsed or are nearing expiration.
- Method of Use Patents: Patents on using the omeprazole-bicarbonate combination for specific indications, or in particular dosing regimens, could offer further protection. However, these are generally less robust than formulation patents once the core components are off-patent.
The expiration of key formulation patents is a significant driver of increased generic competition and subsequent price erosion. Manufacturers relying on patent protection for their specific omeprazole-bicarbonate formulations will see their exclusivity diminish as these patents expire.
Who Are the Major Manufacturers and Competitors in the Omeprazole-Bicarbonate Market?
The market for omeprazole-bicarbonate includes both originator manufacturers and a large number of generic pharmaceutical companies. The competitive landscape is characterized by high volume and price sensitivity.
Key Players and Competitors:
- Originator/Branded Products:
- Bayer AG (previously Merck & Co./G.D. Searle for Zegerid brand in the US): Historically, the brand name Zegerid (omeprazole/sodium bicarbonate) was a prominent product in this space. Bayer now holds commercial rights in various regions for related products.
- Takeda Pharmaceutical Company: While Takeda's primary PPI offerings have focused on lansoprazole (Prevacid), they have historically been involved in the broader PPI market.
- Major Generic Manufacturers: The market is dominated by generic manufacturers who produce bioequivalent versions of omeprazole and omeprazole-bicarbonate formulations. These include:
- Teva Pharmaceutical Industries Ltd.
- Mylan N.V. (now Viatris)
- Sandoz (a division of Novartis AG)
- Aurobindo Pharma Ltd.
- Dr. Reddy's Laboratories Ltd.
- Sun Pharmaceutical Industries Ltd.
- Cipla Ltd.
- Lupin Ltd.
- Hikma Pharmaceuticals PLC
The competition is fierce, with numerous companies vying for market share through aggressive pricing strategies and efficient manufacturing processes. The approval of Abbreviated New Drug Applications (ANDAs) by regulatory bodies like the U.S. Food and Drug Administration (FDA) allows multiple generic versions to enter the market simultaneously, intensifying competition.
What Are the Projected Price Trends for Omeprazole-Bicarbonate?
Price projections for omeprazole-bicarbonate are predominantly downward, driven by the extensive genericization of the omeprazole API and the expiration of key formulation patents.
- Declining Average Selling Prices (ASPs): The average selling price for omeprazole-bicarbonate products has been steadily declining since the entry of multiple generic competitors. This trend is expected to continue.
- Generic omeprazole capsules and tablets typically range from USD 0.10 to USD 0.50 per unit, depending on dosage and pack size [3].
- Formulations that were previously protected by patents, offering a faster onset of action, also face significant price erosion once their patent exclusivity ends. Their prices will converge towards the general generic omeprazole pricing, albeit with a slight premium for the formulation benefits, which will diminish over time.
- Impact of Generic Entry: With the expiration of formulation patents for branded omeprazole-bicarbonate products, multiple generic manufacturers are able to market their versions. This typically leads to a price drop of 50-70% within the first year of generic competition, and further declines in subsequent years [4].
- Reimbursement Policies: Payers and pharmacy benefit managers (PBMs) actively promote the use of lower-cost generic alternatives. Preferred formularies and tiered co-pays incentivize patients and healthcare providers to choose generic omeprazole-bicarbonate over branded options, further suppressing prices.
- Market Saturation: The mature market for omeprazole, a widely prescribed medication, means that supply often outstrips demand, particularly for off-patent formulations. This surplus contributes to price competition.
- Therapeutic Alternatives: The availability of other PPIs (e.g., esomeprazole, lansoprazole, pantoprazole), H2 blockers, and newer drug classes for acid suppression also influences pricing by providing alternative treatment options that can be more cost-effective.
Projected Price Trajectory:
| Timeframe | Price Trend | Contributing Factors |
|---|---|---|
| Current | Stable to Declining | High generic penetration, ongoing price competition among generics. |
| 1-3 Years | Continued Decline (Moderate) | Further consolidation of generic market share, potential for new generic entrants if patents expire. |
| 3-5 Years | Significant Decline (Accelerated) | Maturation of generic market, potential for aggressive pricing by larger generic players. |
It is anticipated that the price of omeprazole-bicarbonate, particularly for formulations previously covered by patents, will approach parity with other generic omeprazole products within the next 3-5 years. The primary differentiator will shift from price to specific patient indications or physician preference based on perceived formulation advantages.
What Are the Regulatory Considerations and Approval Pathways for Omeprazole-Bicarbonate Products?
The regulatory landscape for omeprazole-bicarbonate products is governed by national health authorities, with the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) being prominent examples. The approval pathways depend on whether the product is a new chemical entity, a generic version, or a new formulation.
- Generic Drug Approval (ANDA Pathway in the U.S.):
- Manufacturers seeking to market a generic version of an already approved omeprazole-bicarbonate product must submit an Abbreviated New Drug Application (ANDA) to the FDA.
- The ANDA requires demonstrating bioequivalence to the reference listed drug (RLD) – meaning the generic product must deliver the same amount of active ingredient into the bloodstream over the same period.
- The applicant does not need to repeat the extensive clinical trials required for new drug approval; instead, they rely on existing safety and efficacy data of the RLD.
- Patents covering the RLD must be addressed. Applicants must certify that the patents are invalid, will not be infringed, or have expired (Paragraph IV certification, which can lead to patent litigation).
- New Formulation Approval:
- If a company develops a novel formulation of omeprazole-bicarbonate (e.g., improved stability, novel delivery system, or combination with a new excipient for enhanced pharmacokinetics), it may require a New Drug Application (NDA) or a supplemental NDA (sNDA), depending on the extent of the changes.
- This pathway involves demonstrating safety and efficacy of the new formulation, which can include comparative pharmacokinetic studies, and potentially new clinical trials to prove its superiority or non-inferiority to existing treatments.
- Regulatory Scrutiny on Bioequivalence: Even for generics, regulatory agencies maintain strict standards for demonstrating bioequivalence. Variations in dissolution profiles, particle size distribution, and manufacturing processes can impact bioequivalence and lead to rejection of an application.
- Manufacturing Standards (cGMP): All manufacturers must adhere to current Good Manufacturing Practices (cGMP) regulations to ensure product quality, safety, and consistency. Inspections of manufacturing facilities are a routine part of the regulatory process.
- Labeling and Indication: Generic products typically carry the same indications for use as the RLD. Any new indication or significant change in the risk-benefit profile would necessitate a different approval pathway.
The regulatory environment favors generic competition once patents expire, aiming to make essential medicines more affordable and accessible. However, maintaining product quality and demonstrating robust bioequivalence remain critical hurdles for all manufacturers.
What Are the Key Market Drivers and Challenges for Omeprazole-Bicarbonate?
The market for omeprazole-bicarbonate is influenced by a confluence of factors, creating both opportunities for growth and significant headwinds.
Market Drivers:
- High and Growing Prevalence of GERD and PUD: The increasing incidence of acid-related disorders globally, due to lifestyle changes, diet, and an aging population, creates sustained demand for effective treatments.
- Established Efficacy and Safety Profile: Omeprazole is a well-established and trusted medication with a proven track record of efficacy and a generally favorable safety profile for long-term use when appropriate. The bicarbonate component offers rapid symptom relief, which is a key advantage for many patients.
- Cost-Effectiveness of Generic Options: Once patent protection expires, generic omeprazole-bicarbonate formulations become highly cost-effective, making them a preferred choice for healthcare systems and patients with cost constraints.
- Convenience of Combination Therapy: The availability of a single product combining omeprazole with sodium bicarbonate simplifies treatment regimens for patients requiring both rapid symptom relief and sustained acid suppression.
- Physician Familiarity and Prescribing Habits: Healthcare providers are highly familiar with omeprazole and its therapeutic benefits, leading to continued prescription volumes.
Market Challenges:
- Intense Generic Competition: The market is heavily saturated with generic omeprazole products, leading to significant price erosion and reduced profit margins for manufacturers.
- Patent Expirations: The expiry of formulation patents for novel omeprazole-bicarbonate products opens the floodgates for generic competition, rapidly diminishing market exclusivity and pricing power for originator brands.
- Competition from Other PPIs and Drug Classes: Omeprazole-bicarbonate competes with a wide array of other PPIs (e.g., esomeprazole, lansoprazole, pantoprazole) and alternative therapies like H2 blockers, as well as newer drugs targeting different mechanisms for acid control.
- Stricter Reimbursement Policies: Payers and PBMs actively manage formularies to favor the lowest-cost effective treatments, often restricting coverage for branded or higher-priced generic options.
- Potential for Over-utilization and Off-Label Use: While effective, PPIs like omeprazole are sometimes over-prescribed or used for prolonged periods without clear indication, which can lead to increased scrutiny and potential guideline changes impacting prescription volumes.
- Emerging Therapies: Research and development in gastrointestinal treatments continue, with potential for novel therapies that could offer improved efficacy or different mechanisms of action, potentially displacing current standards of care.
Key Takeaways
The market for omeprazole-bicarbonate is characterized by a strong existing demand base driven by the prevalence of acid-related disorders. However, the competitive landscape is dominated by generic products, leading to sustained price erosion. Key formulation patents have expired or are expiring, facilitating further generic entry and exacerbating price pressures. Manufacturers must navigate intense competition, stringent regulatory requirements, and evolving reimbursement policies to maintain market share and profitability.
FAQs
-
What is the primary benefit of the bicarbonate component in omeprazole-bicarbonate formulations compared to omeprazole alone? The bicarbonate component provides immediate antacid activity, neutralizing gastric acid to offer rapid relief from heartburn and acid indigestion. Omeprazole, a proton pump inhibitor, works to reduce acid production over a longer period.
-
How does the expiration of formulation patents impact the price of omeprazole-bicarbonate? The expiration of formulation patents allows multiple generic manufacturers to produce and sell bioequivalent versions of the drug. This increased supply and competition typically leads to a significant decrease in the average selling price.
-
Are there any specific therapeutic indications where omeprazole-bicarbonate holds a significant advantage over other omeprazole formulations? Omeprazole-bicarbonate is particularly beneficial for patients requiring rapid symptom relief of heartburn and hyperacidity, in addition to sustained acid suppression. This combination addresses both immediate discomfort and the underlying cause of excessive acid production.
-
What are the main regulatory hurdles for generic manufacturers entering the omeprazole-bicarbonate market? Generic manufacturers must demonstrate bioequivalence to the reference listed drug (RLD) through rigorous testing and submit an Abbreviated New Drug Application (ANDA). They must also address existing patents, which can involve lengthy legal challenges. Adherence to Good Manufacturing Practices (GMP) is also critical.
-
Beyond generic competition, what other factors could influence the future price of omeprazole-bicarbonate? Future pricing will be influenced by evolving payer and PBM reimbursement policies that favor lower-cost options, the development of novel therapeutic alternatives for GERD and PUD, and potential changes in clinical guidelines that might affect the overall prescribing patterns for PPIs.
Citations
[1] Grand View Research. (2023). Proton Pump Inhibitors Market Size, Share & Trends Analysis Report By Drug Class, By Indication, By Distribution Channel, By Region, And Segment Forecasts, 2023 - 2030. Retrieved from https://www.grandviewresearch.com/industry-analysis/proton-pump-inhibitors-market
[2] U.S. Food and Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from https://www.fda.gov/drugs/informationondrugs/ucm129444.htm (Specific patent information is accessed through searches on the Orange Book database and patent tracking services).
[3] KFF. (2023). The Cost of Prescription Drugs in the United States. Retrieved from https://www.kff.org/prescription-drugs/issue-brief/the-cost-of-prescription-drugs-in-the-united-states/ (General pricing trends for common generics are reported by various health economics sources).
[4] Generic Pharmaceutical Association (GPhA). (Various Years). Generic Drug Access & Savings Reports. Retrieved from https://www.appp.org/ (Industry reports often detail the price reduction impact of generic entry).
More… ↓
