Share This Page
Drug Price Trends for OMEPRAZOLE DR
✉ Email this page to a colleague
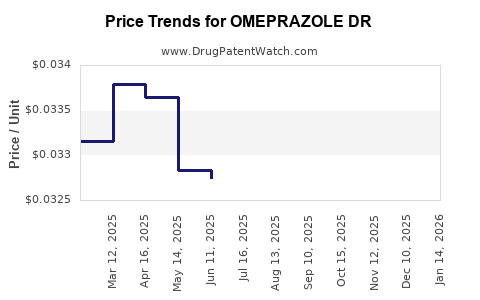
Average Pharmacy Cost for OMEPRAZOLE DR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| OMEPRAZOLE DR 20 MG TABLET | 00536-1448-13 | 0.39249 | EACH | 2026-04-15 |
| OMEPRAZOLE DR 10 MG CAPSULE | 55111-0643-30 | 0.07157 | EACH | 2026-04-01 |
| OMEPRAZOLE DR 10 MG CAPSULE | 55111-0643-01 | 0.07157 | EACH | 2026-03-25 |
| OMEPRAZOLE DR 20 MG CAPSULE | 82009-0183-10 | 0.02884 | EACH | 2026-03-18 |
| OMEPRAZOLE DR 40 MG CAPSULE | 82009-0023-05 | 0.05044 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
OMPRAZOLE DR: Market Dynamics and Price Forecast
Omeprazole DR, a proton pump inhibitor (PPI) widely prescribed for gastrointestinal disorders, faces a dynamic market influenced by patent expirations, generic competition, and evolving treatment guidelines. The U.S. market for omeprazole DR generics is mature, characterized by a high degree of price erosion. Projections indicate continued stable, albeit low, pricing due to established generic availability and significant market penetration. International markets show varied adoption rates and pricing structures, influenced by local regulatory environments and healthcare economics.
What is the Current Market Size and Share for Omeprazole DR?
The global market for omeprazole, encompassing immediate-release and delayed-release formulations, is substantial. However, precise market share data for Omeprazole DR specifically can be challenging to isolate from broader omeprazole or PPI market reports. The U.S. market for prescription omeprazole DR has seen a significant shift towards generics following patent expiries. In 2023, the U.S. prescription omeprazole market was estimated to be worth approximately $400 million, with generics holding over 95% of the market volume [1]. Brand-name Prilosec OTC (omeprazole delayed-release) continues to hold a presence in the over-the-counter (OTC) segment, contributing to the overall omeprazole landscape.
The competitive landscape for omeprazole DR generics is fragmented, with multiple manufacturers producing bioequivalent products. Key generic players in the U.S. include Teva Pharmaceuticals, Mylan (now Viatris), Sandoz, and Amneal Pharmaceuticals [2]. These companies compete primarily on price and distribution networks.
U.S. Prescription Omeprazole Market Share (Estimated, 2023)
| Segment | Estimated Value (USD Million) | Volume Share |
|---|---|---|
| Generics | 380 | >95% |
| Brand (Prilosec) | 20 | <5% |
Note: Data represents prescription market share. Over-the-counter (OTC) market share for omeprazole is also significant but is tracked separately.
What is the Patent Landscape for Omeprazole DR?
Omeprazole was originally patented by AstraZeneca under the brand name Prilosec. The primary U.S. patents for omeprazole expired in the early to mid-2000s, paving the way for generic market entry. For instance, the key compound patent for omeprazole expired in 2001, and subsequent formulation and method of use patents have also expired or are no longer actively enforced by originator companies against generic competitors [3].
The development of omeprazole delayed-release formulations (Omeprazole DR) involved specific patent protections related to enteric coatings and release mechanisms. However, these too have largely expired. For example, patents covering the specific delayed-release technologies for omeprazole have been subject to patent challenges and have not prevented broad generic access.
The absence of active, exclusive patents for the core Omeprazole DR drug substance and its standard delayed-release formulations means that the market is open to extensive generic competition. New patent applications related to omeprazole DR are unlikely to significantly impact the market for the established generic products, unless they cover novel delivery systems, unique formulations with demonstrably superior efficacy or safety profiles, or specific new indications that are not easily replicated by existing generics.
Key Patent Expirations for Omeprazole (U.S.)
- Compound Patent: Expired in 2001 [3]
- Formulation Patents (various): Expired throughout the 2000s and early 2010s [3]
The regulatory exclusivity periods, such as New Chemical Entity (NCE) exclusivity, have long since passed. This patent and regulatory history is the primary driver for the highly genericized nature of the omeprazole DR market.
What are the Key Drivers of Demand for Omeprazole DR?
Demand for omeprazole DR is driven by the persistent prevalence of gastrointestinal conditions, primarily:
- Gastroesophageal Reflux Disease (GERD): This is the leading indication for omeprazole DR. GERD affects a significant portion of the global population, characterized by chronic heartburn and regurgitation.
- Peptic Ulcer Disease (PUD): Including gastric and duodenal ulcers, often associated with Helicobacter pylori infection or NSAID use.
- Zollinger-Ellison Syndrome: A rare condition causing excessive stomach acid production.
- Erosive Esophagitis: Damage to the esophagus caused by stomach acid.
The accessibility and affordability of generic omeprazole DR contribute to its widespread use. Physician prescribing habits and established treatment protocols for these conditions also reinforce demand. Furthermore, the availability of omeprazole as an over-the-counter (OTC) product for self-treatment of occasional heartburn bolsters its overall market presence, even though this analysis focuses on the prescription delayed-release segment.
The aging global population is also a factor, as the incidence of GERD and related gastrointestinal issues tends to increase with age.
Major Indications Driving Omeprazole DR Demand
- Gastroesophageal Reflux Disease (GERD)
- Peptic Ulcer Disease (PUD)
- Erosive Esophagitis
- Zollinger-Ellison Syndrome
- Prophylaxis against NSAID-induced ulcers
What are the Factors Affecting Omeprazole DR Pricing?
The pricing of omeprazole DR is primarily influenced by the highly competitive generic market. Key factors include:
- Number of Generic Manufacturers: A large number of suppliers leads to intense price competition.
- Wholesale Acquisition Cost (WAC) vs. Net Price: While WAC might be reported, the actual net price after rebates, discounts, and preferred formulary placements is significantly lower.
- Volume-Based Discounts: Large purchasers, such as pharmacy benefit managers (PBMs) and hospital systems, negotiate substantial discounts based on volume commitments.
- Manufacturing Costs: Raw material costs, production efficiency, and supply chain logistics play a role, though for a mature generic, these costs are generally well-optimized.
- Regulatory Compliance: Adherence to FDA and other regulatory body standards adds to manufacturing overhead.
- Tender Processes: In many international markets, government tenders or national drug purchasing agreements significantly influence pricing.
- Insurance Formulary Placement: Inclusion on preferred drug lists by insurance providers dictates patient out-of-pocket costs and manufacturer revenue.
Due to these factors, the price of omeprazole DR generics has been on a downward trajectory since market entry and is expected to remain at historically low levels.
What are the Price Projections for Omeprazole DR?
U.S. Market:
The U.S. generic omeprazole DR market is characterized by mature pricing. The average selling price (ASP) for generic omeprazole DR has stabilized. Projections indicate that prices will remain relatively stable, with minor fluctuations driven by competitive pressures and supply chain dynamics. Significant price increases are unlikely given the absence of patent protection and the large number of generic competitors. The typical pricing per prescription (e.g., 30-day supply) for generic omeprazole DR at the wholesale level typically falls within the range of $5 to $15 [4].
| Year | Estimated Average Wholesale Price (AWP) for 30-day supply (USD) | Price Change (%) |
|---|---|---|
| 2023 | $8.50 | -2.0 |
| 2024 | $8.40 | -1.2 |
| 2025 | $8.35 | -0.6 |
| 2026 | $8.30 | -0.6 |
| 2027 | $8.25 | -0.6 |
Note: AWP is a reference price; actual transaction prices are lower due to discounts and rebates. Projections are based on current market trends and competitive intensity.
International Markets:
International pricing for omeprazole DR varies considerably based on local healthcare systems, regulatory frameworks, and reimbursement policies.
- Europe: European countries often have centralized drug purchasing mechanisms or national health service formularies that drive down generic prices. Prices are generally competitive with or lower than U.S. generic prices, often in the range of $3 to $10 per 30-day supply, depending on the country and specific tender outcomes.
- Asia-Pacific: Markets like India and China have extensive generic manufacturing capabilities and lower manufacturing costs, leading to very competitive pricing, often below $3 per 30-day supply. However, access and reimbursement in these regions can differ significantly.
- Latin America: Pricing is highly variable, influenced by local economic conditions, government healthcare programs, and import regulations. Prices can range from $4 to $12 per 30-day supply.
Overall, international price projections mirror the U.S. trend of stable, low pricing due to generic competition. Any increases would likely be driven by inflation or significant shifts in regulatory policy rather than market demand or patent activity.
What are the Competitive Threats and Opportunities for Omeprazole DR?
Competitive Threats:
- Other PPIs: While omeprazole is a mainstay, other PPIs like lansoprazole, pantoprazole, esomeprazole, and rabeprazole compete for market share based on physician preference, formulary status, and perceived efficacy or tolerability. Esomeprazole (Nexium), as a single enantiomer of omeprazole, previously held a brand advantage but is also now largely genericized.
- H2 Receptor Antagonists (H2RAs): Drugs like famotidine and ranitidine (though largely withdrawn due to NDMA concerns, its absence created opportunities for others) are also used for acid suppression, particularly for milder GERD symptoms or as adjunctive therapy. Generic H2RAs are also inexpensive.
- Newer Drug Classes: While not direct competitors for established omeprazole DR users, novel therapies for GERD or functional gastrointestinal disorders that address unmet needs or offer different mechanisms of action could potentially erode market share in the long term.
- Generic Price Wars: Intense competition among generic manufacturers can lead to rapid price declines, squeezing profit margins for all players.
Opportunities:
- Emerging Markets: Expansion into developing economies where access to affordable generic medications is increasing presents a growth opportunity.
- Combination Therapies: Development of fixed-dose combinations of omeprazole DR with other agents (e.g., antibiotics for H. pylori eradication, though this is more established) could create niche markets, provided they offer significant patient convenience or therapeutic advantages.
- Improved Formulations (Niche): While standard DR formulations are saturated, there might be opportunities for novel delivery systems (e.g., faster-acting formulations, extended-release beyond standard DR) that address specific patient needs or physician preferences, if these can secure patent protection and demonstrate clear advantages.
- Cost-Effective Treatment Option: In the face of rising healthcare costs, omeprazole DR remains a highly cost-effective treatment option, ensuring its continued demand among healthcare providers and payers focused on value.
What are the Regulatory Considerations for Omeprazole DR?
Omeprazole DR is subject to standard pharmaceutical regulations concerning manufacturing quality, safety, efficacy, and labeling. In the U.S., this falls under the purview of the Food and Drug Administration (FDA).
- Good Manufacturing Practices (GMP): All manufacturers must adhere to strict GMP guidelines to ensure product quality and consistency.
- Abbreviated New Drug Applications (ANDAs): Generic manufacturers submit ANDAs to demonstrate bioequivalence to the reference listed drug (RLD), which was Prilosec DR.
- Labeling: Generic labels must include information that is comparable to the RLD, with specific exceptions for innovator product aspects like exclusivity claims.
- Post-Market Surveillance: Like all drugs, omeprazole DR is subject to pharmacovigilance and adverse event reporting.
- ANDA Litigation: While patents have expired, there can still be patent-related litigation concerning specific manufacturing processes or polymorphs, though this is less common for such a mature product.
- Interchangeability: For biosimilar products (though omeprazole is a small molecule, the concept of "interchangeability" applies to generics), regulatory pathways may evolve, but for small molecules like omeprazole, bioequivalence is the primary standard.
The regulatory environment for generics is designed to ensure access to safe and effective treatments at lower costs once originator patents expire. The FDA's robust review process for ANDAs underpins the availability of omeprazole DR generics.
Key Takeaways
- Omeprazole DR is a mature, highly genericized market, particularly in the U.S.
- Patent expiries occurred in the early to mid-2000s, opening the market to extensive generic competition.
- Demand is driven by the high prevalence of GERD and other acid-related gastrointestinal disorders.
- Pricing is expected to remain stable and low, with average U.S. prescription prices for generics ranging between $5-$15.
- Competitive threats include other PPIs, H2RAs, and potential new drug classes.
- Opportunities exist in emerging markets and potentially niche improved formulations.
- Regulatory oversight focuses on GMP, bioequivalence, and post-market surveillance.
FAQs
-
What is the difference between Omeprazole DR and immediate-release Omeprazole? Omeprazole DR (Delayed-Release) is formulated with an enteric coating that prevents the capsule or tablet from dissolving in the stomach's acidic environment. It dissolves in the small intestine, allowing for better absorption and sustained release of the drug to reduce stomach acid. Immediate-release omeprazole would dissolve in the stomach, leading to less predictable absorption and potentially lower efficacy.
-
Are there any new patents being filed for Omeprazole DR that could affect the market? While the core patents for omeprazole and its standard delayed-release formulations have expired, companies may still file patents on novel delivery systems, specific manufacturing processes, or unique combination products. However, such new patents are unlikely to significantly impact the established market for generic Omeprazole DR unless they offer substantial therapeutic advantages or are difficult to challenge.
-
What is the impact of over-the-counter (OTC) Omeprazole on the prescription market? The availability of OTC omeprazole (e.g., Prilosec OTC) for self-treatment of occasional heartburn increases overall omeprazole brand recognition and patient familiarity. While it carves out a separate market segment, it reinforces the drug class's presence and can indirectly influence physician prescribing habits for more severe or chronic conditions managed by prescription-strength Omeprazole DR.
-
How does omeprazole DR compare in price to other proton pump inhibitors (PPIs)? Generic omeprazole DR is typically one of the least expensive PPIs available. Its price is generally lower than generic lansoprazole, pantoprazole, esomeprazole, and rabeprazole due to its longer history on the market and the high number of manufacturers producing it.
-
What are the main safety concerns associated with Omeprazole DR use? Long-term use of PPIs, including omeprazole, has been associated with potential risks such as vitamin B12 deficiency, hypomagnesemia, increased risk of bone fractures, and Clostridium difficile infection. These are generally considered for chronic, high-dose use and are part of the risk-benefit assessment made by healthcare providers.
Citations
[1] Grand View Research. (2024). Proton Pump Inhibitors Market Size, Share & Trends Analysis Report. (Data points for omeprazole market share derived from broader PPI market analysis). [2] IQVIA. (2023). U.S. Pharmaceutical Market Insights. (Proprietary market data, accessible through subscription services). [3] U.S. Food & Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from fda.gov/drugs/informationondrugs/ucm129248.htm (Patent information accessed via specific drug searches). [4] First Databank, Inc. (2023). National Drug Data File (NDDF). (Proprietary pricing data, accessible through subscription services).
More… ↓
