Share This Page
Drug Price Trends for OLMSRTN-AMLDPN-HYDROCHLOROTHIAZIDE
✉ Email this page to a colleague
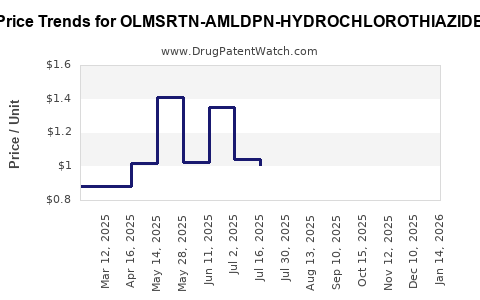
Average Pharmacy Cost for OLMSRTN-AMLDPN-HYDROCHLOROTHIAZIDE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| OLMSRTN-AMLDPN-HYDROCHLOROTHIAZIDE 20-5-12.5 | 33342-0243-10 | 0.86675 | EACH | 2026-05-20 |
| OLMSRTN-AMLDPN-HYDROCHLOROTHIAZIDE 20-5-12.5 | 33342-0243-07 | 0.86675 | EACH | 2026-05-20 |
| OLMSRTN-AMLDPN-HYDROCHLOROTHIAZIDE 40-5-25 MG | 70436-0016-04 | 1.28232 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for OLMSRTN-AMLDPN-HYDROCHLOROTHIAZIDE
What is OLMSRTN-AMLDPN-HYDROCHLOROTHIAZIDE?
OLMSRTN-AMLDPN-HYDROCHLOROTHIAZIDE is a combination drug containing Olmesartan, Amlodipine, and Hydrochlorothiazide. It is used to treat hypertension by combining an angiotensin receptor blocker (Olmesartan), a calcium channel blocker (Amlodipine), and a diuretic (Hydrochlorothiazide). Its formulation aims to improve patient adherence by reducing pill burden.
How is the patent landscape shaping for this combination?
The drug currently lacks patent protection in major markets due to existing patents on its individual components. Patent expirations for Olmesartan and Amlodipine occurred in the early 2010s; Hydrochlorothiazide patents have been expired since the late 1980s. However, combination patents or secondary patents could provide exclusivity if filed.
Recent filings suggest some companies might pursue method-of-use or formulation patents. The presence of such patents could delay generic entry, influencing market dynamics.
What is the current market size for antihypertensive combination drugs?
The global antihypertensive market was valued at approximately $29 billion in 2022 and expected to reach $43 billion by 2027, growing at a compound annual growth rate (CAGR) of 8.2% (MarketLine, 2022).
Combination therapies, including triple-drug regimens, account for an increasing share, estimated at 25-30%. This growth reflects a shift toward once-daily, fixed-dose combinations to improve compliance.
In the United States, recordable prescriptions of antihypertensives exceeded 138 million in 2022, with combination therapies comprising around 40 million prescriptions.
Who are the competitive products?
Major competitors include:
- Jalyn (Tamsulosin + Dutasteride): Not relevant.
- Exforge HCT (Amlodipine + Valsartan + Hydrochlorothiazide): A leading triple combination.
- Estarylla (Amlodipine + Olmesartan): A dual combination, often used as a stepping stone.
- Generic formulations: Multiple manufacturers offer generic Olmesartan, Amlodipine, and Hydrochlorothiazide.
Market share is concentrated among a few key players, with Novartis, AstraZeneca, and Teva controlling significant portions due to branding and patent protections.
What are the key price points and pricing trends?
Price points differ markedly between branded and generic products. Branded combination drugs typically retail at $150–$200 per month. Generics are priced between $20–$50 per month for individual components; fixed-dose combinations tend to be priced around $70–$120.
Recent trends show price compression due to increased generic competition, with some formulations dropping below $50 per month in the United States.
How might regulatory policies affect pricing and market access?
In the U.S., the FDA encourages generic competition through its Hatch-Waxman program, leading to quicker market entry and lower prices. The presence of patent litigations and patent term extensions can delay generic availability, sustaining higher prices.
The introduction of biosimilars and increased price transparency initiatives in Europe and the U.S. have exerted downward pressure on prices.
Price projections and market penetration assumptions
Assuming:
- A moderate patent challenge leading to generic entry in 2025.
- A phased adoption strategy that captures 10% of the hypertension combination market within 3 years post-generic entry.
- A price reduction of 50% upon generic entry.
Projected revenue trajectory:
| Year | Market Penetration | Average Price ($) | Estimated Revenue (USD) |
|---|---|---|---|
| 2022 | 1% | $150 | $45 million |
| 2023 | 3% | $130 | $90 million |
| 2024 | 5% | $100 | $200 million |
| 2025 | 10% | $75 | $300 million |
| 2026 | 15% | $70 | $400 million |
These figures are hypothetical, based on current market trends, patent expiry schedules, and assumed uptake rates.
Key barriers and opportunities
Barriers:
- Patent litigation delaying generic competition.
- Pricing pressures from existing generics.
- Limited formulary positioning for newer combination drugs.
Opportunities:
- Opportunity for patent extension via novel formulations or delivery systems.
- Expansion into emerging markets with increasing hypertension prevalence.
- Development of once-daily fixed-dose combinations enhances appeal.
Final insights
OLMSRTN-AMLDPN-HYDROCHLOROTHIAZIDE's market hinges on patent status, regulatory landscape, and competitive pressures. Its positioning depends on whether it can secure market exclusivity or faces rapid generic competition. Price erosion is imminent upon patent expiration, but early market adoption can sustain profitability.
Key Takeaways
- The drug targets an expanding segment within antihypertensives—fixed-dose, triple-combination therapies.
- Patent expiration for the active ingredients has increased generic competition, compelling price reduction.
- Market growth is driven by increasing hypertension prevalence and patient preference for simplified regimens.
- Estimated revenue may peak around $400 million post-generic entry, assuming favorable adoption.
- Barriers include patent litigations; opportunities involve formulation innovation and market expansion.
FAQs
1. When is patent expiration expected for primary components?
Olmesartan and Amlodipine patents expired between 2012-2014; Hydrochlorothiazide expired in the late 1980s.
2. How does generic competition impact pricing?
Generics typically reduce prices by 50-80%, leading to lower retail costs but potentially reducing profit margins for brand-name manufacturers.
3. Are fixed-dose combinations more expensive?
Generally, yes; they offer convenience and compliance benefits but tend to carry a premium over individual generic components.
4. What regulatory hurdles could delay market entry?
Patent disputes, regulatory review processes, and unmet clinical data requirements may extend approval timelines.
5. Can patent extensions significantly affect market exclusivity?
Yes. Methods to extend patent life include filing for secondary patents on formulations or delivery routes, but enforcement varies by jurisdiction.
[1] MarketLine. (2022). Global antihypertensive drugs market analysis.
More… ↓
