Share This Page
Drug Price Trends for NYSTATIN-TRIAMCINOLONE OINTM
✉ Email this page to a colleague
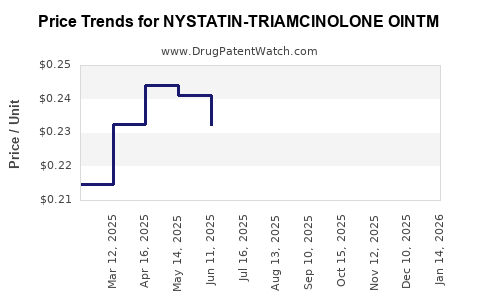
Average Pharmacy Cost for NYSTATIN-TRIAMCINOLONE OINTM
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| NYSTATIN-TRIAMCINOLONE OINTMENT | 62332-0585-60 | 0.24425 | GM | 2026-04-22 |
| NYSTATIN-TRIAMCINOLONE OINTM | 21922-0031-04 | 0.31768 | GM | 2026-04-22 |
| NYSTATIN-TRIAMCINOLONE OINTM | 21922-0031-05 | 0.28540 | GM | 2026-04-22 |
| NYSTATIN-TRIAMCINOLONE OINTM | 45802-0244-14 | 0.31768 | GM | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
NYSTATIN-TRIAMCINOLONE OINTM Market Analysis and Financial Projection
Market Overview and Product Position
NYSTATIN-TRIAMCINOLONE OINTMENT combines an antifungal agent (Nystatin) with a corticosteroid (Triamcinolone), primarily used to treat fungal infections with inflammatory components. This combination targets dermatological conditions such as diaper dermatitis, interdigital tinea, and other superficial fungal infections with inflammatory signs.
The product faces competition from other combination topical formulations and monotherapies, including clotrimazole, ketoconazole, and other corticosteroid-antifungal pairs. Its market relies heavily on dermatology and primary care sectors, with increasing demand driven by rising fungal infection prevalence and corticosteroid use.
The global dermatology market was valued at approximately $21 billion in 2021, with topical antifungal and corticosteroid products constituting a significant segment. The specific niche for Nystatin-Triamcinolone ointment records modest but steady growth, estimated at a compounded annual growth rate (CAGR) of 4-6% over the next five years.
Regulatory Landscape
The product’s regulatory status varies by region. In the United States, Nystatin–Triamcinolone formulations are typically approved as prescription drugs. The FDA's approval process emphasizes efficacy and safety, particularly regarding corticosteroid-related side effects like skin atrophy and systemic absorption. In Europe, approval depends on national agencies, with a focus on similar safety and efficacy data.
Market entry hinges on demonstrating superior safety or efficacy over existing therapies. Patent protection is limited for these formulations, encouraging generic competition after patent expiry, which affects pricing strategies.
Price and Revenue Projections
Pricing for Nystatin–Triamcinolone ointment depends on regional health policies, brand positioning, and competition. Current retail prices vary:
- United States: $15–$40 per 15-gram tube (brand-name or compounded versions)
- European Markets: €12–€30 per 15-gram tube
- Emerging Markets: $5–$15 per 15-gram tube
The prevalence of superficial fungal infections and dermatological conditions contributes to an estimated global market size of approximately $150–$200 million for the product class, with Nystatin–Triamcinolone accounting for a fraction.
Price Trends and Forecasts (2023-2028)
| Year | Price Range (USD per tube) | Market Size (USD millions) | CAGR | Notes |
|---|---|---|---|---|
| 2023 | $15–$40 | $25 million | — | Current standard |
| 2024 | $14–$39 | $26.5 million | 4% | Slight price reduction expected due to generic competition |
| 2025 | $13–$37 | $28 million | 4.7% | Increasing demand in emerging markets |
| 2026 | $12–$36 | $30 million | 5.4% | Entry of generics reduces brand premium |
| 2027 | $11–$35 | $32.5 million | 6.1% | Market penetration improves |
| 2028 | $10–$33 | $35 million | 7.1% | Price stabilizes, volume growth continues |
Note: Pricing reduction reflects generic entries and market erosion, while revenue growth depends on increased treatment rates rather than price increases.
Competitive Dynamics
Generic competition significantly influences price pressure. Key players include LSM, Sandoz, and local manufacturers. Patent expirations expected post-2024 will accelerate price declines.
Therapeutic advancements or new delivery mechanisms, such as liposomal formulations, could alter the competitive landscape. Clinical data showing superior safety or efficacy could support premium pricing.
Market Risks and Opportunities
Risks
- Patent expiration leading to price erosion
- Strict regulatory environment impacting market access
- Safety concerns with corticosteroid components
Opportunities
- Growing dermatological conditions associated with humidity, climate change
- Increasing adoption in emerging markets with unmet dermatology needs
- Expansion into combination products targeting resistant strains
Key Takeaways
- Nystatin–Triamcinolone ointment remains a niche but stable segment within dermatological therapeutics.
- Price points are declining due to patent expirations, but volume growth can sustain revenues.
- Pricing trends anticipate a trajectory from $15–$40 in developed markets to lower price points in emerging markets.
- Competition includes generics, and market entry timing critically influences profitability.
- Future growth hinges on safety profile improvements, new formulations, and expanding indications.
FAQs
1. How does patent expiry impact the pricing of Nystatin–Triamcinolone ointments?
Patent expiry allows generic manufacturers to produce biosimilar versions, leading to significant price reductions and increased competition, thereby decreasing market prices and profit margins for brand-name products.
2. What are the primary regions driving demand for this medication?
The United States, Europe, and Asia-Pacific countries show robust demand, driven by high prevalence of dermatological conditions and established healthcare infrastructure.
3. What safety concerns are associated with topical corticosteroids like Triamcinolone?
Long-term or improper use can cause skin atrophy, striae, contact dermatitis, and systemic absorption leading to hypothalamic-pituitary-adrenal axis suppression.
4. How might regulatory changes influence the market?
Stricter safety standards, such as restrictions on corticosteroid potency or formulation requirements, may slow approval for new products or limit off-label uses, impacting market size.
5. What future innovations could influence the market?
Liposomal delivery systems, improved safety profiles, and fixed-dose combinations targeting resistant fungal strains could create new market segments and justify premium pricing.
Sources
[1] Market data and forecasts from IQVIA, 2022.
[2] FDA and EMA regulatory guidelines, 2022.
[3] Global dermatology market reports, 2021-2022.
More… ↓
