Share This Page
Drug Price Trends for NURTEC ODT
✉ Email this page to a colleague
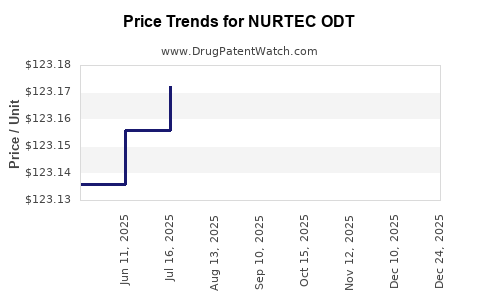
Average Pharmacy Cost for NURTEC ODT
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| NURTEC ODT 75 MG TABLET | 72618-3000-02 | 128.74606 | EACH | 2026-01-01 |
| NURTEC ODT 75 MG TABLET | 72618-3000-02 | 123.20197 | EACH | 2025-12-17 |
| NURTEC ODT 75 MG TABLET | 72618-3000-02 | 123.18761 | EACH | 2025-11-19 |
| NURTEC ODT 75 MG TABLET | 72618-3000-02 | 123.18493 | EACH | 2025-10-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Nurtec ODT Market Analysis and Price Projections
Nurtec ODT (rimegepant) is an orally disintegrating tablet for the acute treatment of migraine with or without aura. Developed by Biohaven Pharmaceuticals, now owned by Pfizer, Nurtec ODT represents a novel CGRP receptor antagonist therapy. Its market penetration and pricing are influenced by its clinical profile, competitive landscape, and payer reimbursement policies.
What is the Current Market Position of Nurtec ODT?
Nurtec ODT has established a significant position in the acute migraine treatment market. Its key differentiators include rapid onset of action and a non-oral tablet formulation, offering convenience for patients. The drug competes with other acute migraine treatments, including triptans, other CGRP antagonists, and gepants.
Key Market Metrics:
- Net Sales: Biohaven reported net sales of $721.3 million for Nurtec ODT in 2022. Pfizer's acquisition is expected to drive further growth.
- Market Share: While specific market share data is proprietary, Nurtec ODT has captured a notable segment of the acute migraine market, particularly among patients seeking alternatives to triptans or those who prefer the ODT formulation.
- Prescription Volume: Prescription data from IQVIA and other market research firms indicate consistent growth in new and total prescriptions since its launch.
What is the Competitive Landscape for Nurtec ODT?
The acute migraine market is dynamic, with multiple therapeutic classes and emerging novel agents. Nurtec ODT competes on several fronts:
1. Other Gepants:
- Ubrelvy (ubrogepant) - AbbVie: Ubrelvy is another oral CGRP antagonist for acute migraine. It has a similar mechanism of action but differs in formulation and pharmacokinetic profile.
- Qulipta (atogepant) - AbbVie: While Qulipta is indicated for migraine prevention, its existence within AbbVie’s migraine portfolio signifies the broader competitive presence of CGRP-targeting therapies.
2. Triptans:
- Sumatriptan (various manufacturers), Rizatriptan (Maxalt), Zolmitriptan (Zomig), etc.: Triptans remain a first-line treatment for many migraine sufferers. They are widely available as generics, offering a lower cost point but are associated with vasoconstrictive side effects, making them unsuitable for certain patient populations.
3. Other Acute Migraine Treatments:
- NSAIDs (e.g., ibuprofen, naproxen): Over-the-counter and prescription NSAIDs are common initial treatments.
- Ditans (e.g., Reyvow - Eli Lilly): Reyvow is a serotonin 5-HT1F receptor agonist offering a non-vasoconstrictive mechanism but has shown a lower response rate compared to some other agents.
Comparison Table: Nurtec ODT vs. Key Competitors
| Feature | Nurtec ODT (Rimegepant) | Ubrelvy (Ubrogepant) | Sumatriptan (Oral) |
|---|---|---|---|
| Mechanism of Action | CGRP receptor antagonist | CGRP receptor antagonist | Serotonin 5-HT1B/1D receptor agonist |
| Indication | Acute treatment of migraine with or without aura | Acute treatment of migraine with or without aura | Acute treatment of migraine with or without aura |
| Formulation | Orally disintegrating tablet (ODT) | Oral tablet | Oral tablet, nasal spray, injection |
| Onset of Action | Rapid (within 30-60 minutes) | Rapid (within 30-60 minutes) | Varies by formulation; generally 30-60 minutes for oral |
| Dosage Frequency | Max 2 doses in 24 hours | Max 2 doses in 24 hours | Max 2 doses in 24 hours (for oral) |
| Key Side Effects | Nausea, fatigue, dry mouth | Nausea, fatigue, somnolence | Dizziness, tingling, flushing, chest pain, nausea |
| Contraindications | Hypersensitivity to rimegepant; severe hepatic/renal impairment | Hypersensitivity to ubrogepant; severe hepatic/renal impairment | Cardiovascular disease, uncontrolled hypertension, stroke history |
| Pricing (List) | High (e.g., ~$1,000 for 8 tablets) | High (e.g., ~$1,000 for 8 tablets) | Low (generic availability) |
| Developer/Owner | Biohaven (Pfizer) | AbbVie | Multiple generic manufacturers |
Note: Pricing is indicative and subject to change based on formulary placement, patient assistance programs, and negotiation.
What are the Pricing Strategies and Reimbursement Dynamics for Nurtec ODT?
Nurtec ODT is positioned as a premium therapy. Its pricing reflects its novel mechanism of action, ODT formulation, and clinical benefits in a significant patient population.
Pricing Structure:
- Wholesale Acquisition Cost (WAC): The list price for a 30-day supply (typically 8 tablets) of Nurtec ODT is approximately $1,000.
- Patient Assistance Programs: Pfizer offers co-pay assistance programs and savings cards to eligible commercially insured patients, reducing out-of-pocket expenses.
- Medicare and Medicaid Reimbursement: Coverage and co-pays vary significantly based on individual plan formularies and Medicare/Medicaid coverage rules.
Reimbursement Challenges and Opportunities:
- Payer Scrutiny: As a high-cost specialty drug, Nurtec ODT faces scrutiny from payers. Prior authorization requirements, step-therapy protocols (requiring trial of less expensive agents first), and site-of-care restrictions are common.
- Value-Based Agreements: While less common in acute migraine treatment currently, discussions around value-based contracting or outcomes-based agreements could emerge to demonstrate the drug's long-term economic value (e.g., reduced ER visits, improved productivity).
- Formulary Placement: Securing favorable formulary placement on commercial and government plans is critical for market access. This is often achieved through rebates and discounts negotiated by Pfizer.
- Physician and Patient Preference: Physician preference for a rapidly acting, convenient formulation and patient demand for effective migraine relief contribute to payer willingness to cover the drug.
What are the Projected Market Size and Growth for Nurtec ODT?
The market for acute migraine treatments is substantial and is expected to grow due to increased diagnosis rates, improved treatment awareness, and the introduction of novel therapies like Nurtec ODT.
Market Drivers:
- Increasing Migraine Prevalence: Migraine affects an estimated 15% of the global population, with significant unmet needs.
- Shift to Novel Therapies: A growing number of patients are moving away from older treatments like triptans due to side effects or lack of efficacy, favoring newer options such as CGRP antagonists.
- Pfizer's Commercial Strength: The acquisition by Pfizer brings significant commercialization resources, sales force expertise, and marketing power, which are expected to accelerate Nurtec ODT's market penetration.
- Potential for New Indications: While currently approved for acute treatment, any future approvals for preventive therapy or other migraine subtypes would expand its market.
Market Size Projections:
- Acute Migraine Market: The global acute migraine market is projected to reach tens of billions of dollars in the coming years.
- Gepant Market Segment: The gepant class, including Nurtec ODT and Ubrelvy, is anticipated to capture a significant share of this market, potentially exceeding $5-10 billion annually within the next five years.
- Nurtec ODT Specific Projections: Analysts project Nurtec ODT's annual sales to reach or exceed $1.5-2 billion within the next 3-5 years, driven by expanded access, strong clinical performance, and Pfizer's commercial strategy. These projections are contingent on continued favorable reimbursement and competitive positioning.
Table: Projected Nurtec ODT Net Sales (USD Billions)
| Year | Projected Net Sales | Growth Rate (%) |
|---|---|---|
| 2023 | 0.8 - 1.0 | N/A |
| 2024 | 1.1 - 1.3 | 15-30% |
| 2025 | 1.3 - 1.6 | 10-25% |
| 2026 | 1.5 - 2.0 | 10-25% |
| 2027 | 1.7 - 2.3 | 10-20% |
Source: Analyst estimates based on market trends, company guidance, and competitive dynamics.
What are the Key Risks and Opportunities for Nurtec ODT?
Risks:
- Intensifying Competition: The rapid development of new acute migraine treatments, including other gepants and potentially novel mechanisms, could erode market share.
- Payer Restrictions: Continued or increased payer restrictions (e.g., higher co-pays, stricter prior authorizations) could limit patient access and impact sales.
- Generic Competition: While a long-term risk for many branded drugs, the current patent protection for Nurtec ODT is robust, but the eventual emergence of generics will significantly impact its market.
- Adverse Event Profile: While generally well-tolerated, any emerging safety concerns could impact its perception and uptake.
- Pfizer Integration Challenges: Successful integration of Biohaven's assets and commercial teams within Pfizer's broader structure is critical for sustained growth.
Opportunities:
- Expanding Patient Access: Continued efforts to secure broad payer coverage and reduce patient out-of-pocket costs will be key.
- Label Expansions: While not currently indicated for prevention, any future expansion of Nurtec ODT's label to include preventive therapy would dramatically increase its market potential.
- Geographic Expansion: Leveraging Pfizer's global infrastructure to expand Nurtec ODT's availability in international markets.
- Combination Therapies: Potential for development of combination products or co-packaging with preventive migraine therapies.
- Leveraging ODT Formulation: Continued emphasis on the convenience and rapid onset of the ODT formulation as a key differentiator.
Key Takeaways
Nurtec ODT (rimegepant) is a leading acute migraine treatment with significant market penetration and strong growth potential, bolstered by Pfizer's acquisition. Its novel CGRP antagonism and convenient ODT formulation provide a competitive edge against triptans and other gepants. While pricing is premium, patient assistance programs aim to mitigate out-of-pocket costs. Market growth is driven by increasing diagnosis rates and a shift towards novel therapies. However, competition and payer restrictions pose ongoing challenges. Opportunities lie in expanding access, potential label expansions, and international market penetration.
FAQs
-
What is the primary mechanism of action for Nurtec ODT? Nurtec ODT is a calcitonin gene-related peptide (CGRP) receptor antagonist. It works by blocking the activity of CGRP, a molecule involved in migraine pathophysiology.
-
How does Nurtec ODT compare to triptans for acute migraine treatment? Nurtec ODT offers a non-vasoconstrictive mechanism, making it suitable for patients who cannot tolerate triptans or have cardiovascular contraindications. It is also formulated as an orally disintegrating tablet for rapid administration and absorption. Triptans are a first-line therapy but can cause vasoconstriction.
-
What is the typical out-of-pocket cost for a commercially insured patient using a Nurtec ODT savings card? Commercially insured patients may be eligible for savings cards that reduce their out-of-pocket cost to as low as $0 for a 30-day supply, subject to program terms and conditions.
-
Has Nurtec ODT been approved for migraine prevention? As of its current approval status, Nurtec ODT is indicated for the acute treatment of migraine with or without aura in adults. It is not approved for migraine prevention.
-
What is the predicted timeline for generic entry of Nurtec ODT? Based on current patent filings and typical patent lifecycles for novel small molecules, generic competition for Nurtec ODT is not anticipated for several years, likely beyond 2030. Specific patent expiry dates and any potential legal challenges will determine the exact timeline.
Citations
[1] Biohaven Pharmaceuticals. (2023). Form 10-K for the fiscal year ended December 31, 2022. U.S. Securities and Exchange Commission. [2] Pfizer Inc. (2023). Investor Relations Presentations and Earnings Calls. (Specific details will vary by presentation date and quarterly report). [3] IQVIA Institute for Human Data Science. (Various reports). The Global Use of Medicines. [4] Market Research Reports from various industry analysis firms (e.g., Grand View Research, Allied Market Research, EvaluatePharma). (Specific report titles and publication dates will vary). [5] U.S. Food and Drug Administration. (Accessed 2023). Drug Approvals Database.
More… ↓
