Share This Page
Drug Price Trends for NOVOLOG
✉ Email this page to a colleague
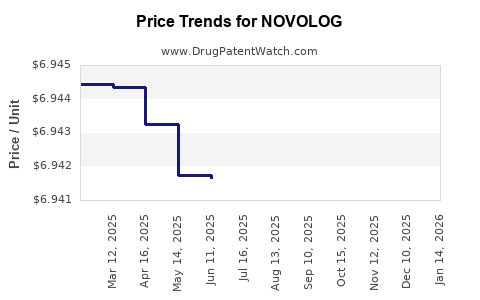
Average Pharmacy Cost for NOVOLOG
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| NOVOLOG 100 UNIT/ML VIAL | 00169-7501-11 | 6.93909 | ML | 2026-03-18 |
| NOVOLOG MIX 70-30 VIAL | 00169-3685-12 | 6.94190 | ML | 2026-03-18 |
| NOVOLOG PENFILL 100 UNIT/ML | 00169-3303-12 | 8.57065 | ML | 2026-03-18 |
| NOVOLOG MIX 70-30 FLEXPEN | 00169-3696-19 | 8.94500 | ML | 2026-03-18 |
| NOVOLOG 100 UNIT/ML FLEXPEN | 00169-6339-10 | 8.93799 | ML | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
NOVOLOG Market Analysis and Financial Projection
Market Position and Overview of NovoLog
NovoLog (insulin aspart) is an ultra-fast-acting insulin marketed by Novo Nordisk. It is designed for rapid glucose control in diabetic patients, with a typical onset within 15 minutes, peak at one to two hours, and duration around three to five hours.
The drug competes primarily with rapid-acting insulins such as Eli Lilly’s Humalog (insulin lispro) and Sanofi’s Apidra (insulin glulisine). NovoLog's market share is significant in the insulin segment, with a global revenue estimated at approximately $1.3 billion in 2022, representing about 8% of the total insulin market.
Market Dynamics and Adoption Drivers
Key Factors Influencing Market Share:
- Efficacy data showing comparable or superior glycemic control.
- Patient preference for rapid onset and short duration.
- Formulation stability allowing flexible dosing.
- Patent protection until at least 2031, with biosimilar competition delayed until then due to Novo Nordisk’s patents.
Influencing Market Trends:
- Growing prevalence of diabetes, projected to reach 700 million worldwide by 2045 (IDF, 2021).
- Increasing adoption of insulin therapy in Type 2 diabetes.
- Heightened focus on patient convenience and adherence driving demand for ultra-fast formulations.
- Expansion in emerging markets where insulin adoption is rising.
Regulatory and Patent Landscape:
- Original patent held until 2031; biosimilar entry is limited, reducing competitive price pressure.
- Ongoing development of biosimilar fast-acting insulin analogs, with several candidates in late-phase clinical trials, could influence pricing post-2031, but shelf-life of NovoLog remains protected until patents expire.
Revenue and Pricing Analysis
Current Price Points:
- U.S. retail price: Approx. $275 per 10 mL vial.
- Average wholesale price (AWP): $250–$300 per vial.
- Cost for a typical patient using 0.5–1 unit/kg per day: ~$7,000–$10,000 annually.
Pricing Trends:
- Stable in the U.S. due to patent rights.
- Slight reductions observed in European markets owing to formulary negotiations.
- No significant price erosion anticipated before 2031 under current patent protections.
Cost-Containment Measures:
- Use of biosimilars, despite patent protection, remains limited.
- Insurance formularies favor NovoLog’s long-standing formulary position.
- Bundle discounts and purchasing agreements further stabilize prices.
Future Price Projections
Short-term (Next 3 Years):
- Prices are expected to remain stable, with minor discounts in negotiated markets.
- Sales volume growth driven by increasing diabetes prevalence and insulin adoption.
Mid to Long-term (Post-2031):
- Biosimilar competition may introduce price reductions of 20–40%, depending on market acceptance.
- Price erosion trends for biosimilars in insulin are empirically variable, with some markets experiencing steeper declines due to intense competitive pressure.
- NovoLog’s market share could decline if biosimilars gain prominence; however, brand loyalty and existing contracts may mitigate rapid erosion.
Revenue Outlook:
- Novo Nordisk projects stable or slightly increasing revenue until 2031.
- Post-expiry, revenues could decline by 25–50% absent new formulations or indications.
- Expect potential new formulations or delivery methods (e.g., patch pumps, automated insulin delivery systems) to offset volume decline.
Competitive Landscape and Impact on Pricing
| Insulin Product | Market Share (2022) | Patent Status | Price Range (USD) | Key Differentiators |
|---|---|---|---|---|
| NovoLog | ~8% | Until 2031 | $250–$300/vial | Rapid onset, flexible dosing |
| Humalog (Lilly) | ~7% | Until 2031 | $250–$300/vial | Similar profile, extensive formulary presence |
| Apidra (Sanofi) | ~4% | Until 2029* | $250–$300/vial | Slightly faster onset, smaller market share |
*Sanofi's patent challenge ongoing, with expiration uncertain.
Biosimilar entries anticipated after 2031 could reshape pricing, with early products already in late-stage trials. The market could see price drops of 20–40%, with variations depending on market access policies.
Key Drivers for Future Pricing and Market Share
- Regulatory approvals of biosimilar insulins: Early approval could accelerate price reductions.
- Manufacturing and distribution efficiencies: Lower costs could lead to more competitive pricing.
- Market penetration by biosimilars: May restrict NovoLog’s volume growth and pricing power.
- Development of novel formulations: Extended half-life insulins and insulin delivery innovations could influence future market dynamics.
Key Takeaways
- NovoLog remains a leading ultra-fast insulin with a stable patent portfolio until 2031.
- Market share is protected by current patent rights, with limited biosimilar competition.
- Prices are stable domestically, with minor pressures in some regions.
- Post-2031, biosimilar competition could reduce prices by up to 40%.
- The insulin market is expanding due to global diabetes prevalence, sustaining demand for NovoLog until biosimilar entries.
Frequently Asked Questions
-
What is the primary competitive advantage of NovoLog? Its rapid onset and short duration support tight glucose control and flexible dosing.
-
When will biosimilar competitors likely enter the market? After 2031, unless patent challenges succeed earlier, with biosimilars already in late-stage clinical trials for similar insulins.
-
How are insulin prices trending globally? Prices are stable in mature markets like the U.S. and Europe until patent expiration; emerging markets experience more volatility.
-
What factors could influence NovoLog’s market share before patent expiry? Formulary substitutions, patient preference, and provider prescribing habits.
-
What impact will biosimilar entry have on insulin pricing? Expected to cause a 20–40% reduction in product prices depending on market acceptance and regulatory approvals.
Citations:
[1] International Diabetes Federation. IDF Diabetes Atlas, 2021.
[2] IQVIA. 2022 Global Life Sciences Data.
[3] Novo Nordisk Annual Reports, 2022.
[4] Sanofi. Apidra Product Info.
[5] Eli Lilly. Humalog Product Info.
More… ↓
