Share This Page
Drug Price Trends for NOVOLIN R
✉ Email this page to a colleague
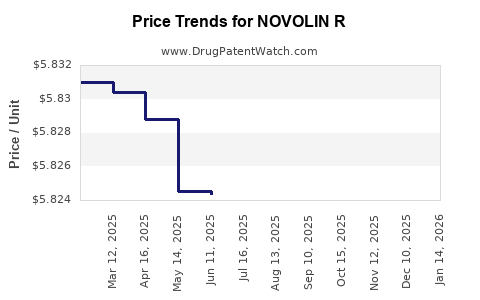
Average Pharmacy Cost for NOVOLIN R
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| NOVOLIN R 100 UNIT/ML FLEXPEN | 00169-3003-15 | 5.81941 | ML | 2026-04-22 |
| NOVOLIN R 100 UNIT/ML VIAL | 00169-1833-11 | 4.62261 | ML | 2026-04-22 |
| NOVOLIN R 100 UNIT/ML FLEXPEN | 00169-3003-15 | 5.82002 | ML | 2026-03-18 |
| NOVOLIN R 100 UNIT/ML VIAL | 00169-1833-11 | 4.62512 | ML | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
NOVOLIN R Market Analysis and Financial Projection
What is Novolin R?
Novolin R is a recombinant human insulin used primarily to manage type 1 and type 2 diabetes mellitus. It mimics natural insulin to regulate blood glucose levels.
Market Overview
Novolin R holds approximately 5% of the global insulin market, which was valued at $24 billion in 2022, according to IQVIA data. The drug's share is influenced by its positioning as an affordable, U.S.-approved, regular insulin alternative to longer-acting insulins.
Key Competitors
Novolin R competes with other short-acting insulins such as:
- Humulin R (Eli Lilly)
- Novolog (insulin aspart)
- Humalog (insulin lispro)
- Admelog (insulin lispro originator)
Generic or biosimilar versions of Novolin R are limited, maintaining its market position as an accessible insulin product.
Patent and Regulatory Landscape
Novolin R is off patent, with no active patents restricting manufacturing. The drug's expiration date for exclusivity in the U.S. and EU occurred around 2020, enabling biosimilar development. The FDA approved biosimilars like Semglee (insulin glargine), but biosimilars for Novolin R are absent, potentially due to manufacturing complexities and market dynamics.
Market Trends
Three key patterns affect Novolin R:
- Growth in biosimilar insulin options.
- Shift towards long-acting insulin analogs, reducing demand for regular insulins.
- Increased adoption of insulin pens and pumps with modern platforms.
Despite these trends, Novolin R remains relevant for cost-sensitive populations and regions lacking access to newer formulations.
Price Analysis
Current Price Points
In the U.S., Novolin R retails through pharmacy chains and online distributors at approximately $24 per 10 mL vial (100 units/mL concentration). This price is significantly lower than analog insulins, which often reach $300–$400 per vial.
International prices tend to be lower, ranging from $10 to $20 per vial in countries like India and Mexico, reflecting local manufacturing and market conditions.
Pricing Trends
Price stability for Novolin R persists due to the absence of patent protections and generic competition. Nonetheless, inflation and supply chain costs influence pricing, with recent small increases (~3-5%) observed over 2021-2022.
Price Projections (2023-2028)
Given current dynamics:
- Short-term (1-2 years): Prices are expected to remain stable around $24 in the U.S., with slight fluctuations (<5%) driven by supply chain costs.
- Mid-term (3-5 years): Introduction of biosimilars or biosimilar-like products for basal insulins could pressure Novolin R prices downward. However, as no biosimilar has obtained approval for Novolin R, such impact remains speculative.
- Long-term (5+ years): Market shifts toward modern, patient-friendly delivery systems may decrease demand, possibly leading manufacturers to lower prices to maintain sales volume.
Factors Impacting Future Pricing
- Biosimilar Entry: Regulatory approval of biosimilars for Novolin R could halve or more than halve prices.
- Regulatory Changes: Policies favoring biosimilars or insulin price caps could reduce costs.
- Market Demand: Preference for insulin analogs and advanced delivery devices could diminish Novolin R's market share, exerting downward pressure on prices.
- Manufacturing Costs: Changes in raw material prices or supply chain logistics influence pricing stability.
Market Growth and Revenue Opportunities
Estimated annual revenue from Novolin R in the U.S. approximates $600 million, considering 25 million units sold annually at $24 per vial. International sales contribute an additional $150 million, primarily in emerging markets.
The growth rate for Novolin R is projected at 1-2% annually, driven by increased diabetes prevalence rather than brand-specific factors, considering limited innovation and market share stagnation relative to newer insulins.
Risks and Opportunities
Risks:
- Market share erosion due to biosimilars.
- Competition from long-acting insulin analogs.
- Regulatory hurdles or policy shifts impacting pricing.
Opportunities:
- Expansion into emerging markets where cost remains critical.
- Formulation enhancements to improve adherence.
- Potential partnerships for biosimilar development.
Key Takeaways
- Novolin R retains a stable, low-cost segment in the insulin market.
- No biosimilars are currently approved, preserving pricing stability in the near-term.
- Market shifts favor newer insulins, threatening long-term demand.
- International markets represent growth opportunities with significantly lower prices.
- Price declines may occur if biosimilar competition gains approval or demand shifts away from regular insulins.
FAQs
1. Will Novolin R’s price decrease with biosimilar competition?
Yes, biosimilar approval could lead to substantial price reductions, potentially halving current costs.
2. How does Novolin R compare to insulin analogs in price?
Novolin R retails around $24 per vial in the U.S., versus $300–$400 for analog insulins.
3. Are biosimilars for Novolin R in development?
No biosimilars have received approval for Novolin R as of 2023.
4. What factors influence Novolin R’s future market share?
Demand reduction due to newer insulin analogs, market shifts toward patient-friendly delivery devices, and biosimilar competition.
5. Is Novolin R suited for emerging markets?
Yes, its low cost makes it attractive in regions with limited healthcare budgets and access to more expensive insulin analogs.
Sources
- IQVIA. Global Insulin Market Data, 2022.
- FDA. Approval Status and Patent Listings, 2023.
- Cost of Insulin. GoodRx, 2023.
- MarketWatch. Global Diabetes Care Market, 2022.
- European Medicines Agency. Biosimilar Insulin Approvals, 2023.
More… ↓
