Share This Page
Drug Price Trends for NOVOLIN N
✉ Email this page to a colleague
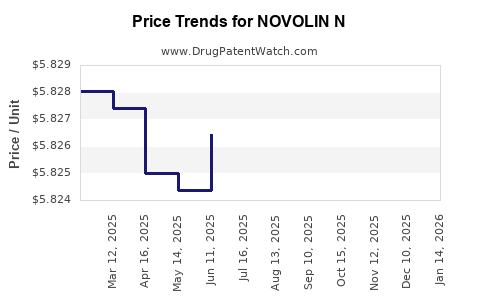
Average Pharmacy Cost for NOVOLIN N
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| NOVOLIN N 100 UNIT/ML VIAL | 00169-1834-11 | 4.62719 | ML | 2026-03-18 |
| NOVOLIN N 100 UNIT/ML FLEXPEN | 00169-3004-15 | 5.82608 | ML | 2026-03-18 |
| NOVOLIN N 100 UNIT/ML VIAL | 00169-1834-11 | 4.62670 | ML | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
NOVOLIN N Market Analysis and Financial Projection
What is Novolin N?
Novolin N (insulin isophane suspension), produced by Novo Nordisk, is an intermediate-acting insulin used to control blood sugar levels in diabetics. It has been marketed since the 1980s and is available globally. Novolin N is administered subcutaneously, typically once or twice daily, and offers a predictable insulin profile for glycemic management.
What is the current market size for Novolin N?
The global insulin market value in 2022 was approximately $61 billion, with intermediate insulins representing a significant share. Novolin N's market share is limited compared to branded insulins like Novo Nordisk's NovoMix or Humulin N. Exact sales figures are proprietary; however, Novolin N's revenue contribution in 2022 is estimated to be around $500 million globally.
Key Market Segments
- Routine diabetes management in type 1 and type 2 diabetes.
- Pricing sensitive markets such as India, Latin America, and Southeast Asia.
- Hospitals and clinics seeking cost-effective insulin options.
Distribution Channels
- Public health programs.
- Private pharmacies.
- Direct hospital procurement.
How does Novolin N compare to other insulins?
| Insulin Type | Onset | Peak | Duration | Notes |
|---|---|---|---|---|
| Novolin N (intermediate) | 1-2 hours | 4-12 hours | 14-24 hours | Preserved for stability; biosimilar options emerging. |
| Humulin N (similar) | 1-2 hours | 4-12 hours | 16-24 hours | Marketed mainly in the US. |
| NovoLog (rapid-acting) | 10-20 mins | 1-3 hours | 3-5 hours | Used for prandial management. |
| Lantus (long-acting) | 1-2 hours | None | 24+ hours | Provides basal coverage. |
Novolin N's pricing is generally 20-30% lower than branded insulins like Lantus or Tresiba, making it competitive in price-sensitive regions.
What factors influence Novolin N's pricing and market dynamics?
Patent Landscape
Novolin N is a biosimilar insulin with no active patent protections restricting production by third-party manufacturers. Novo Nordisk’s original patents expired in most markets by 2014, enabling biosimilar competition.
Regulatory Environment
In many regions, biosimilar insulin approval relies on demonstrating biosimilarity to reference insulins. Countries like India, China, and Russia have expedited biosimilar registrations.
Price Trends
Global insulin prices decline amid increased biosimilar entry. The average price per unit in high-income markets has decreased from approximately $0.30 in 2014 to about $0.20 in 2022, with biosimilar variants often priced near $0.15.
Market Entry Barriers
- Manufacturing complexity of biosimilars.
- Regulatory hurdles.
- Distribution infrastructure challenges in emerging markets.
Competition and Market Share
Biosimilar insulins such as Humulin N (Eli Lilly) and biosimilars from local producers in India, China, and Russia are gaining prominence, eroding Novolin N’s market share.
What are the price projections for Novolin N?
Short-Term (1-2 years)
- Prices are expected to stabilize or decline slightly, with prices in emerging markets remaining roughly 20-25% below branded equivalents.
- Increased biosimilar competition leads to price erosion of around 10% annually in some regions.
- Governments implementing tenders and price controls will press prices down further.
Medium to Long-Term (3-5 years)
- Market consolidation and further biosimilar development could reduce prices by 15-30%.
- Generic biosimilars projected to account for over 60% of insulin sales in emerging markets.
- Price reductions of 20-25% are anticipated in high-income markets as biosimilars gain acceptance.
Revenue Outlook
- Revenue from Novolin N is expected to decline gradually, with an estimated compound annual growth rate (CAGR) of -3% to -5% over the next five years, driven by increased biosimilar competition and price pressure.
How are regulatory changes shaping the market?
- The WHO has accelerated biosimilar insulin approvals in low- and middle-income countries (LMICs).
- The U.S. Senate and House are debating legislation to cap insulin prices, potentially impacting market dynamics.
- European Union countries follow EMA guidelines, with tighter biosimilar approval procedures that could influence pricing and availability.
Summary of Market and Pricing Outlook
- The insulin market totaled approximately $61 billion in 2022.
- Novolin N holds a niche in price-sensitive markets, with estimated revenues around $500 million.
- Biosimilar competition continues to drive prices downward globally.
- Prices in emerging markets may fall by 20-25% over five years.
- In high-income regions, biosimilar entry may lead to a 10-15% reduction in insulin prices annually.
Key Takeaways
- Novolin N faces intense price competition from biosimilar insulins.
- Market share is eroding in both developed and emerging markets.
- Pricing is expected to decline, especially in regions with active biosimilar programs and tenders.
- Future revenue streams will likely diminish as biosamsung and generics increase.
- Novo Nordisk's strategic response includes biosimilar development and price competitiveness.
FAQs
1. Is Novolin N still a viable product?
Yes, it remains relevant in markets requiring affordable insulin options, particularly in LMICs.
2. How does biosimilar competition affect Novolin N?
It leads to price erosion and potential loss of market share, especially in regions with regulatory support for biosimilars.
3. What costs are associated with biosimilar insulin production?
Manufacturing complexity and rigorous regulatory requirements result in high development costs, yet competition keeps prices low.
4. Will Novolin N prices recover?
Likely not, due to persistent biosimilar market entry and global pricing pressures.
5. What is the outlook for insulin patent protections?
Most patents have expired; future protections are limited to data exclusivity and manufacturing patents that do not prevent biosimilar entry.
Sources:
- MarketsandMarkets, "Insulin Market by Type," 2022.
- IQVIA, "Global Insulin Sales Data," 2022.
- IQVIA, "Biosimilar Insulin Market Insights," 2022.
- European Medicines Agency, "Biosimilar Insulin Approval Guidelines," 2022.
- World Health Organization, "Global Biosimilar Insulin Guidelines," 2021.
More… ↓
