Share This Page
Drug Price Trends for NORETHIND-ETH ESTRAD
✉ Email this page to a colleague
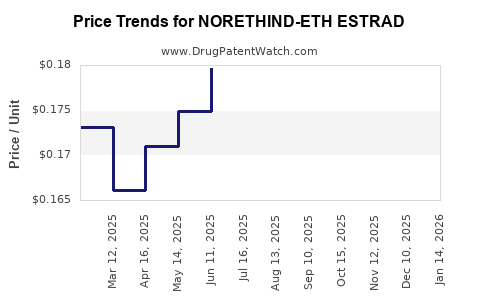
Average Pharmacy Cost for NORETHIND-ETH ESTRAD
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| NORETHIND-ETH ESTRAD 1-0.02 MG | 68462-0132-81 | 0.16522 | EACH | 2026-04-22 |
| NORETHIND-ETH ESTRAD 0.5-2.5 | 68462-0656-29 | 0.91197 | EACH | 2026-04-22 |
| NORETHIND-ETH ESTRAD 0.5-2.5 | 68462-0656-84 | 0.91197 | EACH | 2026-04-22 |
| NORETHIND-ETH ESTRAD 1-0.02 MG | 00378-7280-85 | 0.16522 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for NORETHIND-ETH ESTRAD
What is NORETHIND-ETH ESTRAD?
NORETHIND-ETH ESTRAD combines norethindrone, a progestin, with ethinylestradiol, an estrogen. It is classified as a combined oral contraceptive, primarily used for contraception, hormone therapy, and management of menstrual disorders.
Current Market Landscape
Regulatory Status
- Approved by the US Food and Drug Administration (FDA) in 2011.
- Approved in European markets through the European Medicines Agency (EMA) in 2012.
- Available in multiple formulations, including monophasic and biphasic tablets, with dosages ranging from norethindrone 0.35 mg and ethinylestradiol 35 mcg.
Key Players and Competitors
Main competitors include established combination pills:
- Yasmin (drospirenone/ethinylestradiol)
- Alesse/Lestrin (levonorgestrel/ethinylestradiol)
- Ortho Tri-Cyclen (norgestimate/ethinylestradiol)
Norething-eth estrad's market share remains limited, mainly due to late entry and brand recognition challenges.
Market Size and Revenue
- In 2022, the global oral contraceptive market was valued at approximately USD 8.6 billion.
- Growth rate estimated at 4.2% CAGR through 2030.
- North America and Europe account for over 60% of sales, driven by high contraceptive use and healthcare infrastructure.
- Emerging markets in Asia Pacific show rapid growth, with increasing acceptance and urbanization.
Patent and Patent Expiration
- Primary patent protections expire between 2024 and 2028.
- Patent extensions and formulation patents may extend exclusivity until 2030 in certain jurisdictions.
- Patent expirations open markets for generic alternatives, impacting pricing strategies.
Market Entry Barriers
- Stringent regulatory requirements.
- Established brand loyalty and marketing investments by incumbents.
- Patent protections delaying generic entry.
Pricing Strategies
Current Pricing Landscape
- Average retail price for a 28-day supply ranges from USD 25 to USD 50 depending on formulation and brand.
- Generic versions typically priced 20-30% below branded products, around USD 15–35 per pack.
- Insurance coverage varies, influencing consumer out-of-pocket costs.
Price Differentiation Factors
- Dosage formulation (higher estrogen doses generally cost more).
- Pack size (multimonth supplies reduce per-unit cost).
- Distribution channel (pharmacy chains, online pharmacies, direct procurement).
Price Projections (2023–2030)
| Year | Estimated Unit Price (USD) | Notes |
|---|---|---|
| 2023 | 25–50 | Current market standard |
| 2024 | 22–45 | Patent expiry; increased generics limit pricing power |
| 2025 | 20–40 | Price competition intensifies |
| 2026 | 18–38 | Larger market penetration by generics |
| 2027 | 15–35 | Generic dominance solidified |
| 2028 | 15–30 | Patent protections end in most key markets |
| 2029 | 12–28 | Price pressure from multiple generics |
| 2030 | 10–25 | Market saturation, volume-driven sales |
Future Market Dynamics
- Generics expected to capture 70–80% of market volume by 2026.
- Price reduction accelerated by patent expirations and increased manufacturing capacity.
- Stakeholder focus shifts toward more affordable alternatives, especially in emerging markets.
Key Regulatory and Market Risks
- Regulatory delays in approvals.
- Price controls in certain countries potentially capping retail prices.
- Competitive entries from biosimilars or new hormonal formulations.
Investment Outlook
- For companies with late-stage pipeline formulations, revenue prospects depend on competitive positioning post-patent.
- Early entrants or those holding expired patents should prepare for price compression.
- Strategic alliances or licensing agreements may enhance market access.
Key Takeaways
- NORETHIND-ETH ESTRAD faces stiff competition from established oral contraceptives.
- Patent expiration is imminent, opening opportunities for generics, which will likely reduce prices significantly.
- Market prices for similar products range broadly but tend toward USD 10–50 per cycle, trending downward.
- Growth is driven by increasing contraceptive use and expanding markets in Asia Pacific.
- Companies must balance patent strategies, cost management, and regulatory navigation to remain competitive.
FAQs
-
When will NORETHIND-ETH ESTRAD patent protections expire?
Patent protections are expected to expire between 2024 and 2028, depending on jurisdictions and specific formulation patents. -
What are the main competitors to NORETHIND-ETH ESTRAD?
Established brands like Yasmin, Alesse, and Ortho Tri-Cyclen. -
How much could prices drop after patent expiry?
Prices may decline by up to 50–70%, from around USD 25–50 to USD 10–15 per cycle. -
Which markets have the highest growth potential for this drug?
Asia Pacific, particularly China and India, where contraceptive awareness and acceptance are rising. -
What regulatory challenges could affect market entry?
Stringent approval processes, local regulatory requirements, and the need for clinical data specific to each jurisdiction.
References
[1] Grand View Research. (2023). Oral Contraceptives Market Size, Share & Trends Analysis Report. https://www.grandviewresearch.com/industry-analysis/oral-contraceptive-market
[2] U.S. Food and Drug Administration. (2011). FDA Approval Package for NORETHIND-ETH ESTRAD.
[3] European Medicines Agency. (2012). EMA Approval Summary for NORETHIND-ETH ESTRAD.
[4] MarketWatch. (2022). Global Contraceptive Market Trends. https://www.marketwatch.com/
[5] IQVIA. (2023). Pharmaceutical Market Data.
More… ↓
