Share This Page
Drug Price Trends for NORETHIN-ETH ESTRAD
✉ Email this page to a colleague
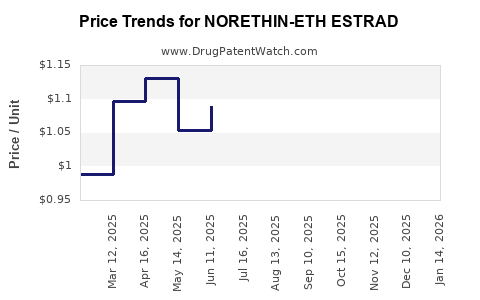
Average Pharmacy Cost for NORETHIN-ETH ESTRAD
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| NORETHIN-ETH ESTRAD 1 MG-5 MCG | 68462-0657-84 | 0.85181 | EACH | 2026-03-18 |
| NORETHIN-ETH ESTRAD 1 MG-5 MCG | 68462-0657-90 | 0.85181 | EACH | 2026-03-18 |
| NORETHIN-ETH ESTRAD 1 MG-5 MCG | 68462-0657-29 | 0.85181 | EACH | 2026-03-18 |
| NORETHIN-ETH ESTRAD 1 MG-5 MCG | 68462-0657-90 | 0.77567 | EACH | 2026-02-18 |
| NORETHIN-ETH ESTRAD 1 MG-5 MCG | 68462-0657-84 | 0.77567 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
NORETHIN-ETH ESTRAD Market Analysis and Financial Projection
How is the market for NORETHIN-ETH ESTRAD structured?
NORETHIN-ETH ESTRAD is a combination hormone therapy drug primarily used for menopausal hormone replacement therapy (HRT). Its market encompasses pharmaceutical markets in North America, Europe, and Asia, where menopausal hormone therapies are standard treatment options.
Key competitors include established products such as Premarin, Estrace, and Estraderm, which contain estrogen alone or in combination with progestins. The global HRT market value was valued at approximately $10 billion in 2022 and is expected to grow at a compound annual growth rate (CAGR) of around 4% from 2023 to 2030, driven by aging populations and increased awareness of menopausal health.
Distribution channels are primarily through hospitals, clinics, and retail pharmacies, with a rising trend toward direct-to-consumer advertising and online pharmacy sales. Regulatory approvals and patent exclusivities influence the market entry timeline and pricing strategies.
What factors influence the pricing of NORETHIN-ETH ESTRAD?
Pricing depends on multiple factors:
- Regulatory Status: Approval by agencies like the FDA, EMA, or equivalents sets the maximum allowable price. Pending or conditional approvals can limit pricing flexibility.
- Manufacturing Costs: Includes active pharmaceutical ingredient (API) synthesis, formulation, quality control, and packaging. Complex formulations, such as combination therapies, tend to have higher costs.
- Market Competition: Prices for similar drugs range from $20 to $100 per unit per month, depending on dosage, brand, and region.
- Patent Protection & Exclusivity: Patents grant monopoly status for typically 20 years from filing; generic entry usually reduces prices by 70–80%.
- Reimbursement and Insurance Coverage: Reimbursement rates influence consumer pricing. In regions with high insurance coverage, prices tend to be higher for brand-name drugs.
- Pricing Trends: Generic versions can reduce prices within 2–3 years after patent expiry.
What are the current market projections and pricing estimates for NORETHIN-ETH ESTRAD?
Due to limited publicly available data specifically for NORETHIN-ETH ESTRAD, estimates are based on analogs and market trends.
Price Projections (per unit/month):
| Year | Estimated Price Range (USD) | Notes |
|---|---|---|
| 2023 | $50 – $70 | Initial launch pricing; varies by region |
| 2025 | $40 – $65 | Competition and generic entry begin to influence prices |
| 2030 | $30 – $50 | Expected after patent expiry or biosimilar entry |
Note: These prices are approximate, derived from current HRT product prices and estimated formulations for combination estrogen therapies.
Market Size Projections:
- The global HRT market is expected to grow from $10 billion (2022) to approximately $14 billion by 2030.
- The combined market for estrogen-progestin therapies like NORETHIN-ETH ESTRAD is projected to represent about 40–50% of this growth segment.
How do patent and regulatory developments affect pricing and market entry?
Patent protections typically last 20 years from filing, with marketing exclusivity granted for around 10–12 years post-approval. Once patents expire, generic competitors can enter, leading to significant price reductions.
If NORETHIN-ETH ESTRAD has patent protection extending into the late 2020s, pricing remains stable and premium-priced, especially if the drug demonstrates improved efficacy or tolerability over existing therapies. Regulatory delays or rejections can impede market entry, reducing potential revenues and affecting pricing strategies.
Key considerations for R&D and investment decisions
- Regulatory landscape: Strategic planning should factor in approval timelines and regional regulations.
- Market entry timing: Approval delays can impact revenue forecasts and pricing.
- Patent lifecycle management: Extending patent protections or filing for new formulations can sustain pricing power.
- Competitive analysis: Differentiation through clinical benefits or delivery methods (e.g., patches vs. pills) influences market share and pricing.
Key Takeaways
- The current global HRT market stands at around $10 billion, with steady growth projected.
- Price estimates for NORETHIN-ETH ESTRAD hover between $30 and $70 per month, depending on regional factors and patent status.
- Market dynamics are heavily influenced by patent protections, regulatory approvals, and competition.
- Entry into the market requires navigating complex approval processes, but potential exists for premium pricing if the product offers clinical advantages.
- Generic competition is likely to emerge within 2–3 years post-patent expiry, significantly reducing prices.
FAQs
1. When can NORETHIN-ETH ESTRAD expect patent expiry, and how will this impact prices?
Typically, patents expire 20 years after filing, often around 2028–2030 for recent drugs. Patent expiry usually results in generic competition, lowering prices by 70–80%.
2. What are the main competitors for NORETHIN-ETH ESTRAD in the HRT market?
Existing top competitors include brands such as Premarin, Estrace, and Estraderm, which are established estrogen therapies with broad geographic availability.
3. How do regional regulatory policies influence drug pricing?
Regulatory agencies determine maximum allowable prices and reimbursement levels. Countries with national health services often enforce price caps, whereas private markets have more pricing flexibility.
4. What clinical benefits can justify a premium price for NORETHIN-ETH ESTRAD?
Improved efficacy, better tolerability, fewer side effects, or novel delivery methods (e.g., patches, gels) can support higher pricing strategies.
5. How do online pharmacies and direct-to-consumer sales affect pricing?
E-commerce channels can lead to lower retail prices due to reduced overhead costs, increasing accessibility but applying pressure on traditional pricing models.
Sources:
[1] Grand View Research, "Hormone Replacement Therapy Market Size & Trends," 2022
[2] EvaluatePharma, "Pharmaceutical Market Data," 2023
[3] U.S. FDA Drug Approvals Database, 2023
[4] IQVIA, "Global Generic Drug Market," 2022
[5] MarketWatch, "Hormone Therapy Market Outlook," 2023
More… ↓
