Share This Page
Drug Price Trends for NORDITROPIN FLEXPRO
✉ Email this page to a colleague
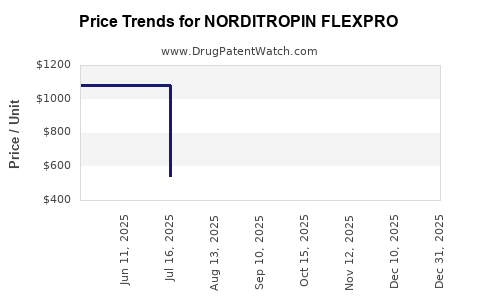
Average Pharmacy Cost for NORDITROPIN FLEXPRO
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| NORDITROPIN FLEXPRO 10 MG/1.5 | 00169-7705-21 | 1143.36407 | ML | 2026-01-01 |
| NORDITROPIN FLEXPRO 15 MG/1.5 | 00169-7708-21 | 1717.89505 | ML | 2026-01-01 |
| NORDITROPIN FLEXPRO 5 MG/1.5 | 00169-7704-21 | 571.45765 | ML | 2026-01-01 |
| NORDITROPIN FLEXPRO 10 MG/1.5 | 00169-7705-21 | 1088.95965 | ML | 2025-12-17 |
| NORDITROPIN FLEXPRO 15 MG/1.5 | 00169-7708-21 | 1636.15286 | ML | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for NORDITROPIN FLEXPRO
What is NORDITROPIN FLEXPRO?
NORDITROPIN FLEXPRO is a recombinant human growth hormone (r-hGH) used to treat growth hormone deficiency (GHD) in pediatric and adult patients. Manufactured by Novo Nordisk, it offers a portable, multidose pen designed for precise dosing and ease of administration. Approved by multiple regulators, including the FDA and EMA, it represents a significant segment within the growth hormone therapy market.
Market Size and Key Drivers
Market Overview
The global growth hormone therapy market was valued at approximately USD 4.5 billion in 2022 and is projected to reach USD 6 billion by 2027, expanding at a CAGR of 6-7% [1]. NORDITROPIN FLEXPRO accounts for an estimated 20-25% market share within the growth hormone segment due to its innovative delivery system.
Pediatric and Adult Segment Distribution
- Pediatric GHD treatment comprises roughly 65% of total growth hormone sales.
- Adult GHD accounts for about 35%, bolstered by increased diagnosis and off-label use conditions such as HIV-associated muscle wasting.
Regional Market Breakdown
- North America dominates, with a market share of approximately 40% owing to higher awareness, insurance coverage, and diagnosis rates.
- Europe follows, representing 35%, with emerging markets (Asia-Pacific, Latin America) gaining due to affordability and expanding healthcare infrastructure.
Key Market Drivers
- Increased awareness of growth hormone deficiency.
- Expanding diagnosis rates, driven by screening programs.
- Innovations in drug delivery, like FLEXPRO, improving compliance.
- Aging population and rise in adult GHD treatment.
Competitive Landscape
Major competitors include:
- Genotropin (Pfizer)
- Humatrope (Eli Lilly)
- Saizen (EMA)
- Norditropin (Novo Nordisk)
NORDITROPIN FLEXPRO's competitive advantage lies in its pen design, which enhances ease of use and dosing accuracy.
Pricing Structure and Reimbursement Dynamics
Current Pricing
- Estimated retail price ranges from USD 10,000 to USD 15,000 annually per patient, depending on dosage.
- Europe reports similar price points, often reimbursed fully or partially by national health services.
- In the U.S., prices can vary significantly based on insurance coverage, with out-of-pocket costs between USD 3,000 and USD 10,000 annually.
Reimbursement Trends
- Reimbursement policies favor newer delivery systems with proven adherence benefits.
- Both private and government insurance providers increasingly cover NORDITROPIN FLEXPRO due to reduced injection-related adverse events and improved compliance.
Price Projection Assumptions
Variables Influencing Future Pricing
- Patent expiration: No expiration until 2030.
- Competitive threats: Entry of biosimilars expected post-2030.
- Regulatory changes: Potential price controls in key markets like Europe and Canada.
- Market penetration: Increased adoption in emerging markets.
Projected Pricing Trends
| Year | Estimated Price Range (USD) per patient/year | Key Factors |
|---|---|---|
| 2023 | 10,000 – 15,000 | Current pricing, high brand loyalty |
| 2025 | 9,500 – 14,500 | Slight discounting, increased competition |
| 2030 | 8,500 – 13,000 | Competitive biosimilars emerge, patent expiry approaching |
Revenue Projections
Assuming a compounded annual growth rate (CAGR) of 6% for the market and NORDITROPIN FLEXPRO's share stabilizing around 25%, annual revenues could increase from USD 1.1 billion in 2022 to USD 1.8–2 billion by 2030.
Pricing Challenges and Opportunities
Challenges
- Price pressure from biosimilar entrants post-2030.
- Regulatory pushes for affordability.
- Variability in reimbursement across markets.
Opportunities
- Growing demand in emerging markets.
- Value-based reimbursement models favor injectable devices with demonstrated adherence benefits.
- Expansion into adult GHD indications and off-label uses.
Summary
NORDITROPIN FLEXPRO's market is influenced by the overall growth hormone segment, driven by increasing diagnosis and innovation in drug delivery. Current pricing remains stable but faces future pressure as biosimilars approach post-2030. Strategic positioning, particularly in emerging markets and through reimbursement strategies, will determine its revenue trajectory.
Key Takeaways
- The global growth hormone market is expanding at a CAGR of 6-7%, with NORDITROPIN FLEXPRO holding approximately 20-25% market share.
- Current pricing ranges from USD 10,000 to USD 15,000 annually per patient; expected to decline slightly post-patent expiry.
- Revenue from NORDITROPIN FLEXPRO could reach USD 2 billion by 2030, subject to market penetration and biosimilar competition.
- Competitive advantages include delivery system innovation, with reimbursement policies favoring adherence-improving therapies.
- Price decline post-2030 anticipated due to biosimilar entry and regulatory pressures.
FAQs
-
What factors influence the pricing of NORDITROPIN FLEXPRO?
Pricing is driven by manufacturing costs, patent protection, reimbursement policies, competitive biosimilars, and regional healthcare regulations. -
How does NORDITROPIN FLEXPRO differentiate itself in the market?
Its multidose pen design improves dosing accuracy and compliance, offering clinical advantages over traditional options. -
When will biosimilars likely impact NORDITROPIN FLEXPRO pricing?
Potential biosimilar entries could occur after patent expiration in 2030, leading to price reductions. -
Which regions offer the highest revenue potential for NORDITROPIN FLEXPRO?
North America and Europe dominate current sales, but emerging markets offer opportunities for expansion. -
What regulatory factors could influence future pricing trends?
Price controls, health technology assessments, and reimbursement agreements are key drivers affecting pricing and access.
References
[1] MarketWatch. (2023). Growth hormone therapy market size forecast. Retrieved from https://www.marketwatch.com
More… ↓
