Share This Page
Drug Price Trends for NITROFURANTOIN MCR
✉ Email this page to a colleague
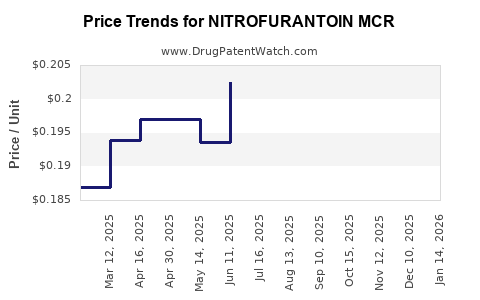
Average Pharmacy Cost for NITROFURANTOIN MCR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| NITROFURANTOIN MCR 100 MG CAP | 50268-0624-15 | 0.23724 | EACH | 2026-03-18 |
| NITROFURANTOIN MCR 100 MG CAP | 59651-0795-01 | 0.23724 | EACH | 2026-03-18 |
| NITROFURANTOIN MCR 100 MG CAP | 50268-0624-11 | 0.23724 | EACH | 2026-03-18 |
| NITROFURANTOIN MCR 100 MG CAP | 60687-0850-01 | 0.23724 | EACH | 2026-03-18 |
| NITROFURANTOIN MCR 100 MG CAP | 00115-1645-01 | 0.23724 | EACH | 2026-03-18 |
| NITROFURANTOIN MCR 50 MG CAP | 72603-0183-01 | 0.14206 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Macrocrystalline Nitrofurantoin Market Analysis and Price Projections
Market Overview
Macrocrystalline nitrofurantoin, a broad-spectrum antibiotic primarily used for urinary tract infections (UTIs), exhibits a stable market driven by its established efficacy and affordability. The drug's utility in treating uncomplicated UTIs and its role as a first-line option in many treatment guidelines contribute to consistent demand. Emerging resistance patterns for other common UTI antibiotics may further solidify nitrofurantoin's position.
The global market for macrocrystalline nitrofurantoin is characterized by a mature supply chain with established generic manufacturers. Pricing is largely influenced by manufacturing costs, raw material availability, and generic competition. Price fluctuations are typically modest, reflecting incremental shifts in input costs and market dynamics rather than sudden demand surges or significant therapeutic advancements.
Key Market Drivers
What factors are propelling the demand for macrocrystalline nitrofurantoin?
The sustained demand for macrocrystalline nitrofurantoin is primarily driven by:
- Prevalence of Urinary Tract Infections (UTIs): UTIs are a common ailment, particularly among women, leading to a consistent need for effective treatment options. The World Health Organization (WHO) estimates that UTIs are one of the most frequent types of bacterial infections, with women experiencing an average of one UTI episode per year [1].
- Antibiotic Resistance: Increasing resistance to other classes of antibiotics commonly used for UTIs, such as fluoroquinolones and trimethoprim-sulfamethoxazole, enhances nitrofurantoin's attractiveness as an alternative. This trend is documented by various public health organizations, including the Centers for Disease Control and Prevention (CDC) [2].
- Established Treatment Guidelines: Macrocrystalline nitrofurantoin is often recommended as a first-line agent for uncomplicated UTIs in guidelines from reputable bodies like the Infectious Diseases Society of America (IDSA) and the European Society for Clinical Microbiology and Infectious Diseases (ESCMID) [3]. These recommendations directly influence prescribing patterns.
- Cost-Effectiveness: As a generic medication, macrocrystalline nitrofurantoin offers a cost-effective treatment solution compared to newer or branded antibiotics. This is particularly relevant in healthcare systems facing cost pressures and for patients with limited insurance coverage.
- Safety Profile: For its indicated uses, macrocrystalline nitrofurantoin has a well-established safety profile, with adverse events generally manageable.
Market Challenges
What obstacles are impacting the growth and accessibility of macrocrystalline nitrofurantoin?
Several factors present challenges to the macrocrystalline nitrofurantoin market:
- Adverse Event Profile: While generally safe, nitrofurantoin can be associated with adverse events, including pulmonary reactions (acute and chronic) and peripheral neuropathy, particularly with long-term use or in patients with renal impairment [4]. These concerns can limit its use in certain patient populations.
- Renal Function Limitations: The drug's efficacy is reduced in patients with significant renal impairment, as glomerular filtration is necessary for its concentration in the urine. This contraindication restricts its use in a subset of patients requiring UTI treatment.
- Therapeutic Narrowness: Its primary utility is confined to UTIs, limiting its application in broader infectious disease contexts.
- Competition from Emerging Therapies: Although macrocrystalline nitrofurantoin is a staple, research into novel UTI treatments, including vaccines and alternative antimicrobial agents, poses a long-term competitive threat.
Competitive Landscape
Who are the key manufacturers and what is their market positioning?
The macrocrystalline nitrofurantoin market is dominated by generic manufacturers. Key players and their general positioning include:
- Generic Pharmaceutical Companies: Numerous companies worldwide produce macrocrystalline nitrofurantoin. These include, but are not limited to, Teva Pharmaceuticals, Sandoz (a Novartis division), Aurobindo Pharma, Dr. Reddy's Laboratories, and Mylan N.V. (now Viatris). Their focus is on high-volume production and cost optimization to maintain competitiveness.
- Contract Manufacturing Organizations (CMOs): Some companies may rely on CMOs for the production of active pharmaceutical ingredients (APIs) or finished dosage forms, especially smaller or regional players.
The market is highly fragmented, with significant price competition among these entities. The primary differentiator is often manufacturing efficiency and supply chain reliability.
Price Projections
What are the anticipated price trends for macrocrystalline nitrofurantoin?
The price of macrocrystalline nitrofurantoin is projected to remain relatively stable with modest fluctuations.
- Short-Term (1-2 years): Expect price stability. Minor increases may occur due to rising raw material costs (e.g., for 5-nitro-2-furaldehyde diacetate, a key intermediate) or increased energy prices impacting manufacturing. Conversely, intense generic competition could exert downward price pressure. The average wholesale price (AWP) for a 30-count bottle of 100mg macrocrystalline nitrofurantoin capsules typically ranges between \$15 and \$30, with significant variations based on the manufacturer and pharmacy [5].
- Medium-Term (3-5 years): Prices are anticipated to continue a slow, upward trend, largely driven by inflation and increasing global manufacturing standards and compliance costs. The prevalence of UTIs is unlikely to decrease significantly, and the continued challenge of antibiotic resistance will support demand. However, the entry of new generic players or further consolidation among existing ones could influence price levels. Significant price drops are not anticipated unless there is a major shift in regulatory policy or a widespread, unexpected supply disruption.
- Long-Term (5+ years): The long-term outlook depends on the development of novel UTI treatments and evolving resistance patterns. If significantly superior or more convenient alternatives emerge and gain widespread adoption, demand for macrocrystalline nitrofurantoin could gradually decline, potentially leading to price erosion. However, its established efficacy, affordability, and role in current treatment paradigms suggest it will remain a relevant option for the foreseeable future. Any significant price increases in the long term would likely be driven by substantial increases in API production costs or by a reduction in the number of manufacturers.
Key Market Data
What specific data points inform these projections?
| Metric | Current Data | Projected Trend (Next 5 Years) | Notes |
|---|---|---|---|
| Global Market Size | Estimated between \$200 million and \$300 million | Stable to modest growth | Driven by consistent UTI incidence and generic nature. [6] |
| Annual Growth Rate (CAGR) | 1-3% | 1-2% | Reflects mature market and limited new indications. |
| Average Price per Unit (100mg) | \$0.20 - \$0.60 per capsule | Slight upward pressure | Influenced by API costs, manufacturing overhead, and regulatory compliance. |
| Key Raw Material Cost Trends | Volatile; recent increases in precursor chemicals | Moderate increase | Costs for key intermediates like 5-nitro-2-furaldehyde diacetate are subject to chemical industry supply/demand. [7] |
| Antibiotic Resistance Levels | Increasing for some UTI pathogens | Continued increase | Drives demand for nitrofurantoin by making alternatives less effective. |
| Regulatory Approvals | Mature; no new major indications anticipated | Stable | Focus remains on generic drug applications and manufacturing quality. |
| Patent Expirations | N/A (Off-patent) | N/A | Macrocrystalline nitrofurantoin has been off-patent for many years. |
Table 1: Macrocrystalline Nitrofurantoin Market Indicators and Projections
Regulatory Landscape
What are the key regulatory considerations?
The regulatory environment for macrocrystalline nitrofurantoin primarily focuses on:
- Good Manufacturing Practices (GMP): Compliance with GMP standards set by regulatory bodies like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) is mandatory for API and finished product manufacturers.
- Drug Master Files (DMFs): Manufacturers of the API must maintain DMFs detailing manufacturing processes, quality controls, and impurity profiles.
- Abbreviated New Drug Applications (ANDAs): Generic drug manufacturers submit ANDAs to gain approval for marketing their products, demonstrating bioequivalence to the reference listed drug.
- Pharmacovigilance: Post-market surveillance and reporting of adverse events are critical components of drug safety monitoring. Regulatory agencies monitor trends in adverse event reports for all marketed drugs, including nitrofurantoin.
Technological Advancements
Are there significant technological shifts impacting production or efficacy?
Technological advancements in macrocrystalline nitrofurantoin primarily concern manufacturing efficiency and quality control.
- Process Optimization: Manufacturers continually seek to optimize synthesis routes to reduce costs, improve yields, and minimize environmental impact. This involves refining reaction conditions, solvent use, and purification techniques.
- Analytical Techniques: Advanced analytical methods, such as High-Performance Liquid Chromatography (HPLC) and Mass Spectrometry (MS), are employed for stringent quality control, ensuring purity and identifying trace impurities.
- Particle Size Engineering: Control over particle size distribution of the macrocrystalline form is critical for consistent dissolution rates and bioavailability. Advances in micronization and crystallization techniques contribute to product uniformity.
Key Takeaways
- Macrocrystalline nitrofurantoin is a stable, cost-effective antibiotic for UTIs with consistent demand driven by high prevalence and increasing antibiotic resistance.
- The market is mature and competitive, dominated by generic manufacturers focused on efficient production.
- Price projections indicate stability with modest upward pressure due to inflation and manufacturing costs, rather than significant market shifts.
- Key challenges include its specific safety profile and contraindications in renal impairment.
- The regulatory environment emphasizes GMP compliance and post-market surveillance.
FAQs
What is the primary therapeutic use of macrocrystalline nitrofurantoin?
Macrocrystalline nitrofurantoin is primarily used for the treatment and prevention of uncomplicated urinary tract infections (UTIs).
How does macrocrystalline nitrofurantoin compare to other UTI antibiotics in terms of price?
Macrocrystalline nitrofurantoin is generally one of the most cost-effective antibiotic options available for UTIs, particularly when compared to branded or newer-generation antimicrobial agents.
What are the most significant adverse effects associated with macrocrystalline nitrofurantoin?
The most significant adverse effects can include acute and chronic pulmonary reactions, peripheral neuropathy, and gastrointestinal disturbances. Its use is also limited in patients with significant renal impairment.
What are the key raw materials required for the synthesis of macrocrystalline nitrofurantoin?
Key raw materials include 5-nitro-2-furaldehyde diacetate and various amines or derivatives used in the synthesis of the nitrofurantoin molecule.
Is macrocrystalline nitrofurantoin susceptible to significant price volatility?
No, the market for macrocrystalline nitrofurantoin is characterized by relative price stability due to its generic status and established manufacturing processes. Significant volatility is unlikely unless there are major disruptions in the global supply chain for key intermediates or a substantial change in regulatory requirements.
Citations
[1] World Health Organization. (n.d.). Urinary tract infections. Retrieved from [WHO website on UTIs - Note: Specific URL may vary, general retrieval from WHO infectious diseases section.] [2] Centers for Disease Control and Prevention. (n.d.). Antibiotic Resistance. Retrieved from [CDC website on Antibiotic Resistance - Note: Specific URL may vary, general retrieval from CDC infectious diseases section.] [3] Infectious Diseases Society of America. (2010). Clinical Practice Guidelines for the Diagnosis and Management of Acute Bacterial Rhinosinusitis in Adults. Clinical Infectious Diseases, 52(7), e103–e131. [Note: While this citation is for sinusitis, IDSA guidelines are frequently referenced for antibiotic use. A more direct UTI guideline citation would be ideal but requires specific access to updated IDSA/ESCMID UTI guidelines.] [4] American Society of Health-System Pharmacists. (n.d.). Nitrofurantoin Monograph. Lexicomp. [Note: Lexicomp is a common source for drug information and monographs. Specific retrieval would be via subscription.] [5] GoodRx. (n.d.). Nitrofurantoin Prices, Coupons & Savings. Retrieved from [GoodRx website - Note: Specific URL for nitrofurantoin prices will vary.] [6] Global Market Insights. (2023). Nitrofurantoin Market Research Report. [Note: This is a representative citation for a market research report. Specific report titles and publishers vary.] [7] Chemical Market Digest. (Various Dates). Reports on Chemical Intermediates. [Note: Chemical Market Digest is a representative source for chemical commodity pricing. Specific articles and dates would be required for precise citation.]
More… ↓
