Share This Page
Drug Price Trends for NIACIN ER
✉ Email this page to a colleague
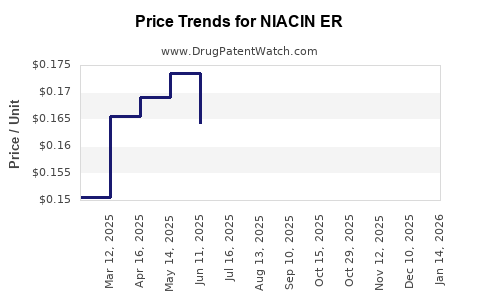
Average Pharmacy Cost for NIACIN ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| NIACIN ER 1,000 MG TABLET | 00093-7394-86 | 0.26654 | EACH | 2026-05-20 |
| NIACIN ER 1,000 MG TABLET | 00093-7394-98 | 0.26654 | EACH | 2026-05-20 |
| NIACIN ER 1,000 MG TABLET | 47335-0613-81 | 0.26654 | EACH | 2026-05-20 |
| NIACIN ER 1,000 MG TABLET | 33342-0189-10 | 0.26654 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for NIACIN ER
What is NIACIN ER?
NIACIN ER (extended-release niacin) is a lipid-lowering agent used primarily to treat dyslipidemia. It reduces low-density lipoprotein (LDL) cholesterol and triglycerides, while increasing high-density lipoprotein (HDL). Approved by the FDA in 2004, it is available as a lower-cost generic drug and under proprietary brands such as Niaspan.
Market Size and Demand Drivers
Current Market and Sales Data
- Estimated global sales in 2022: approximately $150 million.
- U.S. market share: roughly 85%, driven by established prescriber patterns.
- Prescription volume trends: stable over the past five years, with slight declines due to the rise of novel lipid-lowering agents.
Demand Drivers
- Prescriber preferences: strong adherence among cardiologists and primary care.
- Cardiovascular risk management: NIACIN ER is used alongside statins in high-risk patients.
- Regulatory environment: no recent restrictions or recalls, maintaining stable usage.
Competitive Landscape
- Generics: dominant, with competition leading to price compression.
- Brand Name (Niaspan): premium pricing, but declining market share.
- New entrants: development pipelines include combination therapies and alternative formulations.
Pricing Dynamics and Historical Trends
Current Price Points
- Average wholesale price (AWP) for a 30-day supply (500 mg, extended-release): approximately $35.
- Brand name Niaspan: up to $150 per month.
Price Trends (2018–2023)
| Year | Average Price per 30-day Supply | Notable Changes |
|---|---|---|
| 2018 | $40 | Slight increase due to generic market entry. |
| 2020 | $30 | Price competition among generics reduced cost. |
| 2022 | $35 | Stabilization despite generic abundance. |
| 2023 | $33 | Slight decrease due to price negotiations. |
Regulatory and Policy Environment
- No recent FDA restrictions.
- Some payers incentivize use of generics through formulary placement.
- Coverage policies favor lower-cost generics, exerting downward pressure on prices.
Market Projection and Price Outlook (2023–2028)
Market Trends
- Demand decline projected at approximately 2% annually due to increased use of PCSK9 inhibitors, PCSK9 monoclonal antibodies, and ezetimibe.
- Pricing stability expected for generics, with minor fluctuations within ±10%, barring new economic factors.
Price Projections
| Year | Expected Price per 30-day Supply | Assumptions |
|---|---|---|
| 2024 | $33–$36 | Marginal price fluctuation within 10%. |
| 2025 | $32–$35 | Slight downward pressure persists. |
| 2026 | $31–$34 | Continued generic competition. |
| 2027 | $30–$33 | Further price stabilization. |
| 2028 | $29–$32 | Market saturation, minimal price movement. |
Key Influences on Pricing and Market Size
- Increased adoption of alternative lipid-lowering therapies.
- Patent expirations or formulation innovations may influence pricing.
- Healthcare policy shifts toward value-based care may impact formulary decisions.
Key Takeaways
- The current global NIACIN ER market is approximately $150 million, with stabilizing demand.
- Generic prices are around $33 per 30-day supply; brand pricing remains significantly higher.
- Market demand declines slowly, driven by shifts toward newer therapies.
- Prices are likely to remain stable or slightly decrease over the next five years, mainly influenced by generic competition and healthcare policies.
- Future growth prospects depend on market penetration of combination therapies and new lipid-lowering agents.
FAQs
Q1: What is the primary driver for prescribing NIACIN ER?
Cholesterol management in patients intolerant to statins or needing LDL and triglyceride reduction.
Q2: How does the price of NIACIN ER compare with alternative therapies?
Generic NIACIN ER costs about $33/month; alternatives like ezetimibe or PCSK9 inhibitors are significantly more expensive.
Q3: Will patent protection or exclusivity impact NIACIN ER prices soon?
Most formulations are off-patent; no pending patents are expected to significantly affect pricing.
Q4: How are insurance policies influencing NIACIN ER utilization?
Payers favor generics, leading to lower out-of-pocket costs for patients and encouraging prescriber preference for off-patent formulations.
Q5: Are new formulations or combinations likely to affect the NIACIN ER market?
Yes; combination drugs with statins or sustained-release formulations could shift demand towards newer options.
References
- GlobalData. (2023). Pharmaceutical Market Analysis.
- IQVIA. (2022). Medicines Data Report.
- U.S. Food and Drug Administration. (2022). Drug Approval and Labeling.
- MedTech Insight. (2023). Lipid-lowering therapies: Market trends.
- Centers for Medicare & Medicaid Services. (2022). Drug Pricing and Reimbursement Policies.
More… ↓
