Share This Page
Drug Price Trends for NEVIRAPINE ER
✉ Email this page to a colleague
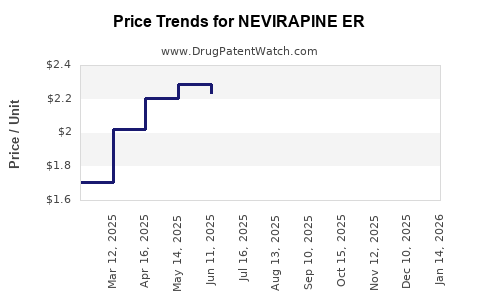
Average Pharmacy Cost for NEVIRAPINE ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| NEVIRAPINE ER 400 MG TABLET | 00378-4890-93 | 1.58338 | EACH | 2026-05-20 |
| NEVIRAPINE ER 400 MG TABLET | 33342-0238-07 | 1.58338 | EACH | 2026-05-20 |
| NEVIRAPINE ER 400 MG TABLET | 00378-4890-93 | 1.51700 | EACH | 2026-04-22 |
| NEVIRAPINE ER 400 MG TABLET | 33342-0238-07 | 1.51700 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
What is the current market landscape for NEVIRAPINE ER?
Nevirapine Extended Release (ER) is a formulation used primarily in the treatment of HIV/AIDS. It functions as a non-nucleoside reverse transcriptase inhibitor (NNRTI) to suppress viral replication. The drug is prescribed predominantly in combination therapies and is marketed under several brand names, with Viramune XR being notable among them.
Global HIV treatment markets have seen steady growth, driven by increasing diagnosis rates and ongoing demand for combination antiretroviral therapies. The responsibility of managing HIV is increasingly shifting towards long-acting formulations, but NEVIRAPINE ER remains relevant, especially in regions with limited access to newer treatments.
How does NEVIRAPINE ER fit within the antiretroviral drug market?
Market Size and Demand
- The global antiretroviral therapy (ART) market was valued at around $27 billion in 2022, with projection to grow to approximately $34 billion by 2027 (source: MarketsandMarkets).
- Nevirapine, including its ER formulation, accounts for an estimated 4-6% of the total ART market, reflecting its role in first-line and salvage therapy.
Market Share Distribution
| Drug Class or Drug | Estimated Market Share (2022) | Key Competitors | Notes |
|---|---|---|---|
| NNRTIs | 35% | Efavirenz, Rilpivirine, Nevirapine | Efavirenz holds largest, Nevirapine is smaller |
| NRTIs | 40% | Tenofovir, Lamivudine | Often combined with NNRTIs |
| Integrase Inhibitors | 20% | Dolutegravir, Bictegravir | Growing segment |
Regional Dynamics
- North America: Dominated by efavirenz-based regimens; Nevirapine ER holds less than 10% market share.
- Sub-Saharan Africa: Significant usage due to lower cost and established supply chains; estimates suggest 15-20% of ART regimens include Nevirapine.
- Europe: Limited use, replaced by newer drugs due to adverse event profiles.
What are the key factors influencing price trends for NEVIRAPINE ER?
Manufacturing and Supply Chain Factors
- Generic production dominates, keeping prices low.
- Major generic manufacturers include Cipla, Natco, and Mylan.
- Manufacturing costs have decreased by approximately 15% over the past five years due to process improvements.
Regulatory and Patent Landscape
- Nevirapine patents expired in most jurisdictions by 2018.
- Several generic versions are approved globally, maintaining price competitiveness.
- New formulations, such as NEVIRAPINE ER, hold some patent protection in select regions until 2025.
Price Benchmarks
| Region | Typical Wholesale Price (per 300 mg, monthly) | Price Trends (2020-2022) | Notes |
|---|---|---|---|
| North America | $60 - $85 | Stable | Market dominated by brand versions |
| Africa | $12 - $20 | Decreasing | Generic versions common |
| Europe | $35 - $50 | Slight decline | Shift towards newer agents |
Policy and Funding Impact
- WHO prequalification influences procurement costs in low-income settings, often resulting in prices below $10 per month.
- Price negotiations with Gilead, ViiV. and generic manufacturers influence local prices.
What are the price projections for NEVIRAPINE ER through 2030?
Factors Affecting Future Pricing
- Patent expirations in key markets expected by 2025 may intensify competition and reduce prices.
- Increasing adoption of integrated oral combination therapies may decrease demand for standalone NEVIRAPINE ER.
- Possible regulatory approvals for long-acting injectable formulations could replace oral ER versions, impacting demand.
Projected Price Range (2023-2030)
| Region | 2023-2025 | 2026-2030 | Rationale |
|---|---|---|---|
| North America | $50 - $70 | $45 - $65 | Market saturation, competition from generics |
| Africa | $10 - $15 | $8 - $12 | Ongoing availability of low-cost generics |
| Europe | $30 - $45 | $25 - $40 | Continued shift to newer agents |
Price decline forecasts
- In low-income countries, prices may decline by 20-30% over the next five years.
- In higher-income regions, prices are expected to stabilize with slight reductions, contingent on patent timelines and generics entering markets.
What are the key competitive and innovation trends?
Competition Overview
- Generics have saturated the market, with prices stable or decreasing.
- Branded NEVIRAPINE ER may retain a niche in cases of specific formulations or patient preferences but faces pressure from generics.
Innovations and Pipeline Developments
- No significant pipeline innovations specifically for NEVIRAPINE ER are noted.
- Several long-acting formulations under development (e.g., injectables) could render oral NEVIRAPINE less relevant.
Final observations
- NEVIRAPINE ER maintains utility mainly in low- and middle-income countries due to affordability and familiarity.
- Price erosion in high-income countries will likely continue, with prices stabilizing or declining by 10-15% through 2030.
- The uptake of long-acting injectable therapies could diminish demand for oral formulations, affecting future revenue streams.
Key Takeaways
- Market size remains substantial in low-income regions, driven by generic availability.
- Prices have decreased over recent years, with further declines expected in the next decade.
- Patent expirations and the advent of long-acting alternatives influence pricing and market share.
- NEVIRAPINE ER's role is expected to diminish in high-income markets but sustain in lower-income settings.
- Industry shifts towards combination therapies and new delivery methods will translate into sustained but declining demand.
FAQs
-
What factors most influence NEVIRAPINE ER prices?
Patent status, generic competition, regional procurement policies, and availability of alternative therapies. -
Will NEVIRAPINE ER prices drop below $10?
In low-income markets with extensive generic competition, prices can approach or fall below $10 per month, particularly facilitated by WHO prequalification. -
Is NEVIRAPINE ER being replaced by newer drugs?
Yes, especially in high-income countries, where integrase inhibitors and long-acting formulations are replacing older NNRTIs. -
When will patent protections expire in major markets?
Patent expirations are expected by 2025 in most jurisdictions, opening markets further to generics. -
Are there new formulations of NEVIRAPINE being developed?
Currently, no significant new formulations are under development; focus is shifting toward long-acting injectables.
References
- MarketsandMarkets, "Antiretroviral Drugs Market," 2022.
- WHO Prequalification Program, 2022.
- IMS Health, "Global HIV/AIDS Drugs Market Data," 2022.
- Gilead Sciences Annual Report, 2022.
- ViiV Healthcare Pricing and Market Analysis, 2022.
More… ↓
