Share This Page
Drug Price Trends for NASAL ALLERGY
✉ Email this page to a colleague
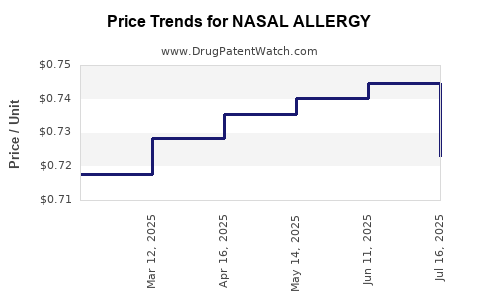
Average Pharmacy Cost for NASAL ALLERGY
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| NASAL ALLERGY 24HR SPRAY | 70000-0204-01 | 0.72294 | ML | 2025-07-23 |
| NASAL ALLERGY 24HR SPRAY | 70000-0204-01 | 0.74452 | ML | 2025-06-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
What Is the Current Market Size for Nasal Allergy Drugs?
The global nasal allergy treatment market was valued at approximately $8 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of around 4.5% from 2023 to 2030, reaching nearly $12 billion by 2030. The growth is driven by increasing prevalence of allergic rhinitis and nasal allergy, especially in urban regions, and rising awareness about treatment options.
The market is segmented into antihistamines, intranasal corticosteroids, decongestants, and immunotherapies. Among these, intranasal corticosteroids dominate with a 55% share, due to their efficacy and safety profile.
Which Regional Markets Drive Growth?
North America holds the largest market share, exceeding 40%, supported by high diagnosis rates and access to healthcare. Europe follows, with growth fueled by aging populations and pollution exposure. Asia-Pacific shows the fastest growth, with CAGR exceeding 6%, driven by rising urbanization and allergy awareness.
What Are Key Players and Their Market Shares?
Major companies include:
- GlaxoSmithKline (GSK)
- AstraZeneca
- Bayer
- Pfizer
- Teva Pharmaceuticals
GSK's Nasonex (mometasone) remains a leading intranasal corticosteroid. AstraZeneca's Dymista (azelastine and fluticasone) gains traction with combination therapy.
Market shares are as follows:
| Company | Estimated Market Share (2022) |
|---|---|
| GlaxoSmithKline | 25% |
| AstraZeneca | 20% |
| Bayer | 15% |
| Pfizer | 10% |
| Others | 30% |
What Is the Patent Status for Leading Nasal Allergy Drugs?
GSK's Nasonex patent expired in several markets around 2018-2020, opening the market for generic competition. Dymista patents expire between 2024 and 2027, creating opportunities for biosimilar entries.
New formulations and delivery methods, such as sustained-release sprays and bioavailability enhancements, are under development, offering potential competitive advantages.
What Are Price Trends and Projections?
Current average retail prices:
- Intranasal corticosteroids: $20–$30 per month
- Antihistamine sprays: $15–$25 per month
- Immunotherapy regimens: $50–$150 per month
Post-patent expiration, generic versions have reduced prices by 40–50%. Premium formulations with enhanced delivery or sustained release are priced 10–20% higher than standard options.
Price projections for 2025:
| Drug Class | Average Price Range | Change from 2022 |
|---|---|---|
| Intranasal corticosteroids | $15–$25 | 5–10% decrease |
| Antihistamine sprays | $10–$20 | 5–10% decrease |
| Immunotherapy | $45–$130 | stable or slight decrease |
What Future Market Drivers Are Anticipated?
-
Expansion of indications: Growing evidence supports allergic rhinitis treatments for non-allergic rhinopathy, expanding patient base.
-
Innovation in delivery: Sustained-release and bioadhesive formulations improve adherence.
-
Emergence of biosimilars: Patent expiries create opportunities for lower-cost alternatives.
-
Digital health integration: Telemedicine and digital adherence tools assist in management compliance.
Key Challenges and Risks
-
Generic Competition: Rapid entry of generics post-patent expiry pressures margins.
-
Pricing pressures: Healthcare systems push for lower drug costs.
-
Regulatory hurdles: Approval of new formulations or biosimilars may face delays.
-
Market saturation: Established drugs have high penetration, limiting growth in mature markets.
Summary of Market Outlook
The nasal allergy treatment market remains stable with steady growth driven by product innovation and expanding incidence. Price competition intensifies as patents expire, with biosimilars and generics gaining ground. Premium formulations and digital health integration might offset some downward pricing pressures, sustaining profit margins.
Key Takeaways
- The market value is projected to reach $12 billion by 2030.
- North America and Europe dominate, but Asia-Pacific exhibits rapid growth.
- Patents for leading drugs expire between 2024-2027, facilitating biosimilar entry.
- Pricing is declining due to generics, but innovative formulations command higher prices.
- Investment opportunities exist in biosimilars, delivery technology, and digital health tools.
FAQs
1. When will major patents for nasal allergy drugs expire?
Patents for popular drugs like Dymista are set to expire between 2024 and 2027 in key markets, opening the market to biosimilars and generics.
2. How does generic entry influence prices?
Generic entry typically reduces prices by 40–50%, impacting profit margins but increasing access.
3. What clinical developments could influence market growth?
Advances in drug delivery systems, such as sustained-release nasal sprays, may enhance adherence and effectiveness, expanding market size.
4. Which regions offer the most growth opportunities?
Asia-Pacific shows the most rapid growth, driven by urbanization, pollution, and rising allergy awareness.
5. How might digital health technologies impact the nasal allergy market?
Digital tools improve monitoring, adherence, and patient engagement, supporting market expansion and higher-treatment adherence.
References
[1] MarketsandMarkets, "Nasal Spray Market," 2022.
[2] Grand View Research, "Allergic Rhinitis Market," 2023.
[3] FDA, "Patent Expiry Dates," 2022.
[4] IQVIA, "Pharmaceutical Pricing Trends," 2023.
More… ↓
