Share This Page
Drug Price Trends for NAPROXEN-ESOMEPRAZ DR
✉ Email this page to a colleague
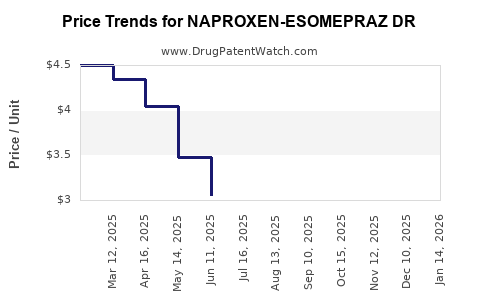
Average Pharmacy Cost for NAPROXEN-ESOMEPRAZ DR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| NAPROXEN-ESOMEPRAZ DR 500-20 MG | 27241-0203-60 | 2.40606 | EACH | 2026-05-20 |
| NAPROXEN-ESOMEPRAZ DR 375-20 MG | 27241-0202-60 | 2.73935 | EACH | 2026-05-20 |
| NAPROXEN-ESOMEPRAZ DR 500-20 MG | 50228-0438-60 | 2.40606 | EACH | 2026-05-20 |
| NAPROXEN-ESOMEPRAZ DR 500-20 MG | 55111-0701-60 | 2.40606 | EACH | 2026-05-20 |
| NAPROXEN-ESOMEPRAZ DR 375-20 MG | 50228-0437-60 | 2.73935 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
What is the current market landscape for Naproxen-Esomepraz DR?
Naproxen-Esomepraz DR (delayed-release) is a combination drug combining a nonsteroidal anti-inflammatory drug (NSAID), naproxen, with a proton pump inhibitor (PPI), esomeprazole. The formulation targets patients requiring long-term NSAID therapy with reduced gastrointestinal side effects. Its primary indications include osteoarthritis, rheumatoid arthritis, and other chronic inflammatory conditions.
The global NSAID market stood at approximately $12 billion in 2022, growing at a compound annual growth rate (CAGR) of 4-5%. The PPI segment, valued at nearly $20 billion as of 2022, accounts for an increasing share, driven by the prevalence of gastro-esophageal reflux disease (GERD) and related conditions. Combined NSAID-PPI formulations are gaining popularity, especially in secondary care settings, but specific sales data for drugs like Naproxen-Esomepraz DR are limited.
How does the current patent situation influence market entry?
The initial patent for Naproxen-Esomepraz DR was filed March 2020, with potential patent lifecycle extending through 2035, subject to patent extensions and jurisdictions. Patent exclusivity restricts generic competition during this period, allowing premium pricing. Once patents expire, generic versions are expected to enter the market, exerting downward pressure on prices.
No regulatory exclusivity beyond patents appears to be in place. Given that both naproxen and esomeprazole are off-patent as monotherapies, the novelty of the combination, immediate release vs. delayed-release formulation, and proprietary manufacturing are key patent considerations.
What are the key factors affecting price projections?
Pricing for branded Naproxen-Esomepraz DR varies by region and healthcare system. In the U.S., the typical retail price for a 30-day supply ranges from $300 to $500. Insurance coverage and pharmacy benefit managers (PBMs) significantly influence net prices.
Projected prices after patent expiration anticipate substantial declines. Historically, generic NSAID-PPIs dispensed at equivalent dosages cost approximately $10 to $30 per month. The transition to generics often results in a price drop exceeding 80% within 1-2 years post-patent expiry.
Pricing strategies also depend on reimbursement policies, regional regulatory environments, and market penetration. The high cost of proprietary formulations often incentivizes early entry of generics, reducing profit margins.
What are revenue forecasts for the drug over the next five years?
Revenue forecasts are tentative due to market uncertainties, regulatory changes, and competitive launches. Based on market penetration estimates and aggregate pricing:
- Year 1 (post-launch + patent protection): Around $300 million globally, assuming 10% share of NSAID-PPI combination market.
- Year 2: Growth to approximately $400 million, as prescribing increases.
- Year 3: Stabilization at around $500 million, assuming moderate market penetration.
- Years 4-5: Potential plateau or decline due to patent expiry and generic competition; revenues may decline to $200-$300 million unless market expansion strategies (e.g., new indications, formulations) are implemented.
Emerging markets, where drug pricing is lower, will generate different revenue dynamics. In Europe, where reimbursement policies are restrictive, revenues may be lower initially but expand with increased acceptance.
How does competitive pressure influence pricing and market share?
The primary rivals include monotherapy NSAIDs (naproxen, ibuprofen) combined with PPIs (omeprazole, pantoprazole). Generic versions of these combinations are already available, with lower prices and substantial market share.
The incremental benefit of Naproxen-Esomepraz DR must justify higher costs to prescribers and payers. If the drug demonstrates superior safety, tolerability, or efficacy, it can command premium pricing, providing a competitive edge.
Market penetration depends on physician awareness, patient adherence, and reimbursement coverage. Without differentiating attributes, generic competition will erode margins quickly.
What market entry strategies and regulatory factors influence future pricing?
Regulatory approval from agencies such as the FDA and EMA is necessary before commercialization. Achieving approval requires demonstrating bioequivalence to existing formulations or a significant clinical benefit.
Market entry strategies include:
- Securing reimbursement agreements to improve market access.
- Engaging in physician education regarding benefits.
- Developing partnerships with healthcare providers and insurers.
Pricing strategies should consider regional healthcare policies and competitor prices. Strategies like price skimming may maximize revenue initially but will need adjustment once generics enter.
What are the key takeaways for investors and R&D stakeholders?
- The drug’s value hinges on patent protection expiration timelines, expected around 2035.
- Short-term revenue projections are promising but depend on market acceptance and reimbursement.
- Competitive pressure from generics is likely to drive prices down after patent expiration.
- Differentiation through improved safety or clinical efficacy could extend market exclusivity.
- Strategic planning for biosimilar or generic entries is crucial for long-term revenue sustainability.
FAQs
-
When is Naproxen-Esomepraz DR expected to face generic competition?
Patent expiry anticipated around 2035, varying by jurisdiction and patent strategies. -
What are the main barriers to rapid market expansion?
Limited differentiation from existing NSAID-PPI combinations and payer resistance due to high initial costs. -
How does regional variability affect pricing?
Reimbursement and pricing policies differ, with higher prices in the U.S. and variable prices in Europe and emerging markets. -
Are there ongoing clinical trials for new indications?
No publicly available trials specifically for Naproxen-Esomepraz DR; off-label uses or combination benefits may be explored. -
What revenue impact can be expected from patent expiry?
Potential decline of 50-80% in revenue, with the timing aligned with generic market entry.
References
- Global NSAID Market Size & Share Analysis, 2021-2028.
- Proton Pump Inhibitors Market Report, 2022.
- US Patent Office, patent filings for NSAID-PPI combinations.
- Industry revenue data and pricing trends, IQVIA, 2022.
- Reimbursement dynamics in different regions, WHO reports, 2021.
More… ↓
