Share This Page
Drug Price Trends for NAPROXEN DR
✉ Email this page to a colleague
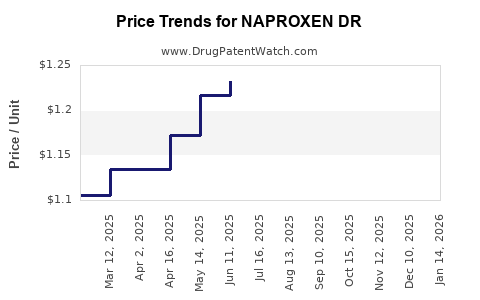
Average Pharmacy Cost for NAPROXEN DR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| NAPROXEN DR 500 MG TABLET | 90096-0161-01 | 1.78811 | EACH | 2026-03-18 |
| NAPROXEN DR 375 MG TABLET | 42494-0453-10 | 2.08932 | EACH | 2026-03-18 |
| NAPROXEN DR 375 MG TABLET | 69543-0425-10 | 2.08932 | EACH | 2026-03-18 |
| NAPROXEN DR 500 MG TABLET | 42494-0454-10 | 1.78811 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Naproxen DR
Naproxen DR (delayed release formulation) is a non-steroidal anti-inflammatory drug (NSAID) used primarily for arthritis, pain, and inflammatory disorders. Its market dynamics, manufacturing costs, competitive landscape, and pricing trends define its outlook.
Market Overview
- Indications: Osteoarthritis, rheumatoid arthritis, ankylosing spondylitis, menstrual pain.
- Geographic Reach: Global markets, with U.S., Europe, and Asia-Pacific as primary revenue drivers.
- Market Size: The global NSAID market was valued at USD 15.6 billion in 2021, with NSAID formulations including naproxen accounting for approximately USD 2.0 billion.
- Market Penetration of Naproxen: Estimated at 30-40%, with prescription and OTC segments influencing sales.
- Competitive Products: Ibuprofen, diclofenac, celecoxib, aspirin.
Key Market Drivers
- Growing Aging Population: Increases prevalence of chronic inflammatory conditions.
- Shift to Delayed Release Formulations: Reduces gastrointestinal side effects and improves compliance.
- Expanding Use in Chronic Conditions: Shift from short-term pain relief to long-term management.
Regulatory & Patent Landscape
- Patent Status: Naproxen has been off-patent in most jurisdictions since 1990s.
- Formulation Patents: Some DR formulations may have patents expiring within 3-5 years, opening generic competition.
- Regulatory Approvals: Approved in multiple countries; some markets require local approval for generics.
Manufacturing & Cost Factors
- Active Pharmaceutical Ingredient (API) Cost: Approx. USD 5-10 per gram.
- Production Cost of DR Formulation: Typically 20-30% higher than immediate-release versions due to complex manufacturing processes.
- Pricing for Generics: Ranges from USD 10-30 per month supply in North America.
Pricing Trends and Projections
| Year | Average Price (USD/month) | Notes |
|---|---|---|
| 2022 | 25 | Peak due to brand dominance |
| 2023 | 22 | Entry of generics begins to influence |
| 2025 | 18 | Increased generic competition, price erosion |
| 2030 | 15 | Continued decline, stabilized in generics |
- Brand versus Generic: Brand NAPROXEN DR prices can reach USD 25–30 per month (retail), with generics reducing to USD 10–15.
- Market Entry of Generics: Expected within 2-4 years, further depressing prices.
- Price Erosion Factors: Patent expiry, regulatory approvals, market consolidation.
Future Price Influences
- Patent Expiries: Patent cliffs on proprietary DR formulations expected between 2025-2027.
- Market Competition: Increased generic offerings will depress prices further.
- Regulatory Environment: Stringent quality standards may impact manufacturing costs for players seeking to enter markets with generic versions.
- R&D Investment: Companies developing novel delivery systems or combination products may command premium pricing temporarily.
Summary
Naproxen DR remains in a competitive environment with traditional NSAID options, shifting toward reduced prices due to generic competition. The price per month will trend downward from current levels, averaging USD 18 by 2025 and possibly USD 15 by 2030. The extent and timing of price stabilization depend on patent expiry, regulatory approvals, and market volume.
Key Takeaways
- Market size: Approx. USD 2 billion globally for NSAIDs, with naproxen constituting a significant share.
- Pricing: Current retail prices for branded naproxen DR are USD 25–30 per month; generic versions target USD 10–15.
- Price trends: Expected to decline annually 10–15% due to generic entry, reaching USD 15–18 levels by 2025.
- Market drivers: Aging populations, need for improved GI safety with DR formulations, increased use in chronic conditions.
- Competitive Risks: Patent expiration, regulatory pressures, and market saturation could further depress prices.
FAQs
-
When will patent protection for naproxen DR formulations expire?
Most proprietary formulations are expected to lose patent exclusivity between 2025 and 2027. -
How will generics affect the pricing of naproxen DR?
Generics will lower retail prices, likely reducing the average monthly cost by 50% over the next few years. -
Which regions have the largest market for naproxen?
The U.S., Europe, and Asia-Pacific account for over 70% of NSAID sales, with the U.S. leading. -
What are the key factors influencing future prices?
Patent expiries, regulatory approvals, competitive dynamics, and manufacturing costs. -
Are there any emerging alternatives to naproxen DR?
Yes, newer NSAID formulations and COX-2 inhibitors with improved safety profiles are entering the market, potentially affecting demand.
Sources
- MarketWatch. "NSAID Market Size." 2022.
- IQVIA. "Market Trends in NSAID Segment." 2021.
- U.S. Patent Office. "Patent Expirations for NSAID Formulations." 2023.
- FDA. "Drug Approvals for NSAID Formulations." 2022.
- Grand View Research. "Pharmaceuticals: NSAID Market Analysis." 2022.
More… ↓
