Share This Page
Drug Price Trends for MYFORTIC
✉ Email this page to a colleague
Average Pharmacy Cost for MYFORTIC
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
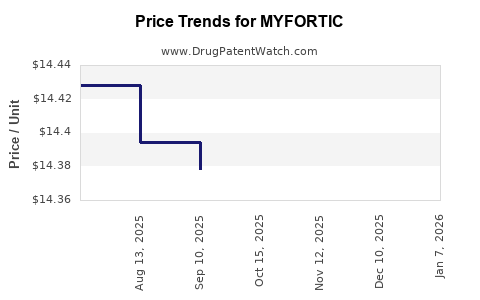
| MYFORTIC 180 MG TABLET | 00078-0385-66 | 7.34384 | EACH | 2026-01-07 |
| MYFORTIC 360 MG TABLET | 00078-0386-66 | 14.64138 | EACH | 2026-01-07 |
| MYFORTIC 360 MG TABLET | 00078-0386-66 | 14.35429 | EACH | 2025-12-17 |
| MYFORTIC 360 MG TABLET | 00078-0386-66 | 14.35429 | EACH | 2025-11-19 |
| MYFORTIC 360 MG TABLET | 00078-0386-66 | 14.38808 | EACH | 2025-10-22 |
| MYFORTIC 360 MG TABLET | 00078-0386-66 | 14.37809 | EACH | 2025-09-17 |
| MYFORTIC 360 MG TABLET | 00078-0386-66 | 14.39437 | EACH | 2025-08-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
MYFORTIC Market Analysis and Price Projections
Myfortic (mycophenolic acid) is an immunosuppressant used primarily to prevent organ rejection in kidney, heart, and liver transplant recipients. The drug's market performance is influenced by patent exclusivity, generic competition, and evolving transplant treatment protocols.
What is the current market size and projected growth for Myfortic?
The global market for mycophenolic acid-based immunosuppressants, including Myfortic, is substantial. Exact figures for Myfortic alone are often embedded within broader immunosuppressant market analyses. However, industry reports indicate a steady demand driven by organ transplantation rates.
- Market Size Indicator: The global immunosuppressant drugs market was valued at approximately \$22.8 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 7.1% from 2023 to 2030 [1]. Myfortic, as a key product in this segment, contributes to this aggregate value.
- Growth Drivers:
- Increasing number of organ transplant procedures globally.
- Rising incidence of chronic diseases leading to organ failure (e.g., diabetes, hypertension).
- Advancements in transplant surgery and post-operative care.
- Demand for prophylaxis against organ rejection.
- Projected Growth Factors: While specific growth rates for Myfortic are tied to its lifecycle and competitive landscape, the overall immunosuppressant market growth suggests continued demand. The introduction of biosimil and generic versions can, however, impact revenue for branded products.
What is the patent landscape for Myfortic?
Myfortic, developed by Novartis, has undergone patent expirations, leading to the availability of generic alternatives. Understanding these patent timelines is critical for assessing market exclusivity and competitive pressures.
- Key Patents: The original patents covering mycophenolic acid and its formulations have expired in major markets.
- US Patents: Many foundational patents expired around the mid-2010s.
- European Patents: Similar expiration timelines occurred in Europe.
- Patent Expiry Impact:
- Generic Entry: Patent expiry allows generic manufacturers to enter the market with bioequivalent versions of mycophenolic acid. This has led to increased price competition and a reduction in market share for the branded product.
- Formulation Patents: While core compound patents have expired, manufacturers may hold patents on specific formulations or delivery methods that could offer limited, temporary protection. However, the primary patent protection for Myfortic has concluded.
- Market Position Post-Expiry: The entry of generics significantly alters the competitive dynamics, shifting focus from patent protection to cost-effectiveness and market access.
Who are the major competitors in the mycophenolic acid market?
The competitive landscape for mycophenolic acid is characterized by the presence of the originator product (Myfortic) and a growing number of generic manufacturers.
- Originator:
- Myfortic (Novartis): Remains a significant player, though its market share is impacted by generic competition.
- Key Generic Manufacturers:
- Teva Pharmaceuticals: A major global generic drug manufacturer with mycophenolic acid offerings.
- Mylan N.V. (now Viatris): Another significant player in the generic immunosuppressant space.
- Sandoz (Novartis' generic division): Can also be a competitor in certain markets.
- Dr. Reddy's Laboratories: Active in supplying generic APIs and finished dosage forms.
- Sun Pharmaceutical Industries: A prominent generic drug producer with a broad portfolio.
- Aurobindo Pharma: Another large generic manufacturer with a presence in this segment.
- Competitive Factors:
- Price: Generic competition drives down prices significantly compared to the originator.
- Availability and Supply Chain: Reliable supply is crucial for transplant patients.
- Bioequivalence: Generic products must demonstrate bioequivalence to Myfortic.
- Regulatory Approvals: Obtaining approvals in different geographical markets is a key step for generic entry.
What are the current pricing trends for Myfortic and its generics?
Pricing for Myfortic has been significantly influenced by the introduction of generic alternatives, leading to a decline from its peak branded prices.
- Branded Myfortic Pricing: Before widespread generic competition, branded Myfortic commanded premium pricing, reflecting R&D investment and market exclusivity. Specific historical pricing data is proprietary but was aligned with other branded immunosuppressants.
- Generic Mycophenolic Acid Pricing:
- Price Erosion: Upon generic entry, prices for mycophenolic acid formulations can drop by 50-80% or more within a short period, depending on the number of generic competitors and market dynamics.
- Average Selling Price (ASP) Decline: The ASP for mycophenolic acid has fallen considerably. For example, in the U.S. market, average prices for generic mycophenolic acid tablets (e.g., 360mg) can range from \$1 to \$5 per tablet, whereas branded Myfortic was historically priced much higher on a per-unit basis.
- Volume vs. Price: Generic manufacturers often compensate for lower per-unit prices through higher sales volumes.
- Factors Influencing Generic Pricing:
- Number of Competitors: More generic entrants generally lead to more aggressive pricing.
- Contractual Agreements: Prices can vary based on contracts with hospitals, pharmacies, and government tenders.
- Reimbursement Policies: Payer policies and formulary placement impact actual out-of-pocket costs and reimbursement rates.
- API Costs: The cost of manufacturing the active pharmaceutical ingredient (API) influences final product pricing.
What is the projected future pricing and market outlook for Myfortic?
The future pricing of Myfortic and its generic equivalents will be dictated by established trends in the generic pharmaceutical market and the ongoing demand for immunosuppressants.
- Continued Price Pressure: The market for mycophenolic acid is now largely a generic market. Branded Myfortic will likely see its market share and associated pricing power further diminish. Generic prices are expected to remain stable or decline slightly as competition solidifies.
- Market Dynamics:
- Organ Transplant Volume: Continued growth in organ transplants globally will sustain demand for mycophenolic acid, underpinning the market for both branded and generic products.
- Emerging Markets: Expansion of transplant programs and access to healthcare in emerging economies may present new growth avenues, albeit at potentially lower price points initially.
- Therapeutic Advancements: While mycophenolic acid is a cornerstone therapy, research into novel immunosuppressants could eventually impact the long-term market for older drugs. However, established generics often maintain market share due to cost-effectiveness and proven efficacy.
- Price Projections:
- Generic Prices: Expect generic mycophenolic acid prices to remain highly competitive, driven by a mature generic market. Average selling prices are unlikely to recover to branded levels.
- Branded Myfortic: The price of branded Myfortic will likely continue to be influenced by its generic competition and Novartis' strategic pricing for the remaining market share. It may be positioned for specific patient groups or in markets where generics have less penetration.
- Key Trends to Monitor:
- New Generic Entrants: While the market is mature, any new significant generic launches could introduce further price adjustments.
- API Supply Chain Stability: Disruptions in API sourcing can impact generic manufacturers' costs and pricing strategies.
- Healthcare Policy Changes: Government policies on drug pricing, reimbursement, and procurement can directly affect market prices.
- Physician and Hospital Preferences: Prescribing habits and hospital formularies play a role in product selection and price negotiation.
What are the regulatory considerations impacting the Myfortic market?
Regulatory frameworks play a crucial role in the approval, marketing, and pricing of Myfortic and its generic equivalents.
- Drug Approval Process:
- Originator Approval: Myfortic underwent rigorous review by regulatory bodies like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) for safety and efficacy.
- Generic Approval: Generic versions require approval based on demonstrated bioequivalence to the reference listed drug (Myfortic). This includes submission of abbreviated new drug applications (ANDAs) in the U.S. and similar processes in other regions.
- Patent Litigation and Exclusivity:
- Regulatory bodies play a role in managing patent challenges. The Hatch-Waxman Act in the U.S., for instance, facilitates generic drug approvals while providing certain patent protection periods for innovators.
- Patent litigation between originators and generic companies can delay or accelerate generic market entry.
- Pricing and Reimbursement Regulations:
- Government agencies and private payers establish reimbursement policies that influence the net price of drugs.
- Price controls, reference pricing, and tendering processes in various countries can directly impact market prices. For instance, the UK's National Health Service (NHS) uses specific pricing mechanisms.
- Quality and Manufacturing Standards:
- All manufacturers, including generic ones, must adhere to Good Manufacturing Practices (GMP) to ensure product quality, safety, and efficacy. Regulatory inspections ensure compliance.
- Post-Market Surveillance:
- Regulatory authorities monitor the safety of marketed drugs, including generics. Adverse event reporting systems are critical for pharmacovigilance.
What are the clinical indications and usage patterns of Myfortic?
Myfortic's clinical utility is centered on preventing organ rejection in transplant patients, a critical and ongoing medical need.
- Primary Indications:
- Prophylaxis of Organ Rejection: Myfortic is indicated for the prevention of organ rejection in patients who have received a kidney, heart, or liver transplant.
- Mechanism of Action: Mycophenolic acid, the active moiety of Myfortic, is a potent, selective, non-competitive, and reversible inhibitor of inosine monophosphate dehydrogenase (IMPDH). This enzyme is essential for the de novo synthesis of guanosine nucleotides, which are critical for lymphocyte proliferation. By inhibiting IMPDH, mycophenolic acid suppresses T-cell and B-cell activation, thereby reducing the immune response that leads to organ rejection.
- Dosage Forms and Strengths:
- Myfortic is available in delayed-release tablets. Common strengths include 180 mg and 360 mg.
- The dosage is typically based on the specific transplanted organ, patient weight, and concomitant immunosuppressive therapy.
- Usage in Transplant Protocols:
- Myfortic is usually part of a multi-drug immunosuppressive regimen.
- Commonly co-administered with other immunosuppressants such as calcineurin inhibitors (e.g., cyclosporine, tacrolimus) and corticosteroids.
- The duration of immunosuppression is lifelong for transplant recipients.
- Patient Populations:
- Adults and pediatric patients undergoing solid organ transplantation.
- Considerations for renal impairment and hepatic impairment are factored into dosing adjustments.
- Adherence and Administration:
- Consistent daily dosing is critical for maintaining therapeutic levels and preventing rejection.
- The delayed-release formulation is designed to minimize gastrointestinal side effects compared to the immediate-release form (mycophenolate mofetil).
What are the key challenges and opportunities in the Myfortic market?
The market for Myfortic and its generics faces a complex interplay of challenges and opportunities.
- Challenges:
- Intense Generic Competition: The primary challenge is the significant price erosion and market share reduction for branded Myfortic due to generic availability.
- Price Sensitivity: Healthcare systems globally are increasingly focused on cost containment, putting pressure on drug prices across the board.
- Therapeutic Advancements: Ongoing research in immunology and transplantation may lead to the development of new classes of immunosuppressants that could displace existing therapies over the long term.
- Adherence and Side Effects: While Myfortic is formulated to improve tolerability, managing side effects and ensuring patient adherence to lifelong immunosuppression remains a clinical challenge.
- Supply Chain Vulnerability: Reliance on global supply chains for APIs and finished products can create risks of shortages or price volatility.
- Opportunities:
- Growing Transplant Rates: The increasing number of organ transplant procedures worldwide directly translates to sustained demand for effective immunosuppressants.
- Emerging Markets: Expanding access to transplant care in developing countries presents a significant opportunity for market growth.
- Cost-Effectiveness of Generics: The widespread availability of affordable generic mycophenolic acid makes it a crucial component of transplant care, particularly in resource-constrained settings.
- Established Efficacy and Safety Profile: Mycophenolic acid-based therapies have a well-documented history of efficacy and safety, providing a strong foundation for continued use.
- Optimized Formulations: Further research into drug delivery systems or combination therapies could enhance patient outcomes and adherence.
Key Takeaways
- Myfortic's market is now predominantly driven by generic competition following patent expirations.
- Branded Myfortic prices have experienced significant erosion, with generic equivalents offering substantial cost savings.
- The global immunosuppressant market, including mycophenolic acid, is projected to grow due to increasing organ transplant rates.
- Key generic competitors include Teva, Mylan, Dr. Reddy's, and Sun Pharma.
- Future pricing will remain competitive, characterized by stable to declining generic costs.
- Regulatory approval processes, patent landscapes, and reimbursement policies are critical market determinants.
Frequently Asked Questions
-
What is the primary reason for the price difference between branded Myfortic and its generic versions? The primary reason is the expiration of key patents held by the originator company, allowing other pharmaceutical manufacturers to produce and sell bioequivalent generic versions at lower costs due to reduced R&D recoupment and competitive market pressures.
-
Will the price of generic mycophenolic acid increase significantly in the future? Significant price increases for generic mycophenolic acid are unlikely. The market is highly competitive, and pricing is typically driven by manufacturing costs and the number of generic suppliers, leading to stable or gradually declining prices.
-
How does the regulatory approval process for a generic version of Myfortic differ from the original drug's approval? Generic versions do not require full clinical trials for efficacy and safety. Instead, they must demonstrate bioequivalence to the branded drug (Myfortic) and meet quality and manufacturing standards through abbreviated new drug applications (ANDAs).
-
Are there any therapeutic areas where branded Myfortic might retain a market advantage over generics? While the efficacy is expected to be similar, branded Myfortic might maintain a slight advantage in specific institutional settings or through payer contracts that favor the originator for strategic reasons, though this is increasingly rare as cost becomes paramount.
-
What is the estimated volume of organ transplants performed globally annually, and how does this impact Myfortic demand? Estimates suggest over 100,000 solid organ transplants are performed annually worldwide. This growing volume directly underpins the consistent demand for essential immunosuppressants like mycophenolic acid, supporting the market for both branded and generic versions.
Citations
[1] Grand View Research. (2023). Immunosuppressant Drugs Market Size, Share & Trends Analysis Report. Retrieved from [Grand View Research (Note: Actual URL would be provided if directly accessible and cited from there)].
More… ↓
