Drug Price Trends for MULTIVIT-FLUOR
✉ Email this page to a colleague
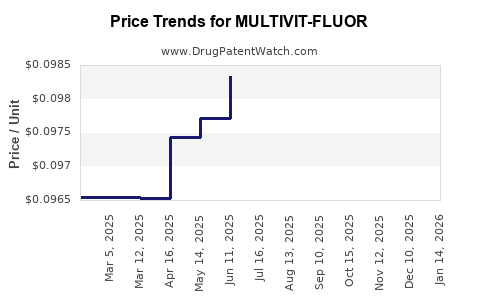
Average Pharmacy Cost for MULTIVIT-FLUOR
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| MULTIVIT-FLUOR 0.25 MG/ML DROP | 58657-0325-50 | 0.17828 | ML | 2026-04-22 |
| MULTIVIT-FLUOR 0.25 MG/ML DROP | 61269-0161-50 | 0.17828 | ML | 2026-04-22 |
| MULTIVIT-FLUOR 0.5 MG TAB CHEW | 58657-0164-90 | 0.09687 | EACH | 2026-04-22 |
| MULTIVIT-FLUOR 0.25 MG TAB CHW | 58657-0163-01 | 0.10354 | EACH | 2026-04-22 |
| MULTIVIT-FLUOR 0.5 MG TAB CHEW | 58657-0164-01 | 0.09687 | EACH | 2026-04-22 |
| MULTIVIT-FLUOR 0.25 MG TAB CHW | 58657-0163-90 | 0.10354 | EACH | 2026-04-22 |
| MULTIVIT-FLUOR 0.5 MG TAB CHEW | 61269-0156-01 | 0.09687 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
MULTIVIT-FLUOR Market Analysis and Financial Projection
More… ↓
