Share This Page
Drug Price Trends for MUCUS-CHEST CONG
✉ Email this page to a colleague
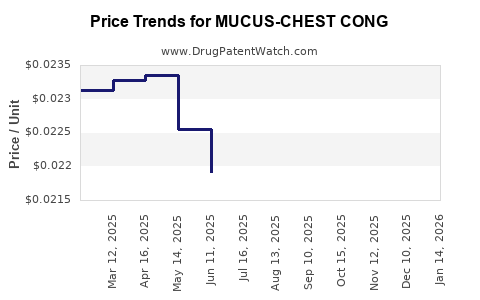
Average Pharmacy Cost for MUCUS-CHEST CONG
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| MUCUS-CHEST CONG 200 MG/10 ML | 00536-1430-97 | 0.02242 | ML | 2026-04-22 |
| MUCUS-CHEST CONG 200 MG/10 ML | 00536-1430-97 | 0.02235 | ML | 2026-03-18 |
| MUCUS-CHEST CONG 200 MG/10 ML | 00536-1430-97 | 0.02218 | ML | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Mucus-Chest Cong
What is the current market landscape for Mucus-Chest Cong?
Mucus-Chest Cong is a brand of expectorant used primarily to treat excessive mucus and congestion associated with respiratory infections. The drug addresses a broad patient base that includes adults, children, and elderly populations. The demand correlates with respiratory illnesses, notably during seasonal peaks and in regions with high pollution levels.
Current Market Size
- The global expectorant market was valued at approximately USD 2.8 billion in 2022.
- Given the respiratory disease prevalence, expectorants account for roughly 60% of OTC cough and cold medication sales.
- Mucus-Chest Cong commands a share of the expectorant market, with an estimated USD 250 million in retail sales globally in 2022.
Regional Distribution
| Region | Market Share | Estimated Sales 2022 (USD million) |
|---|---|---|
| North America | 40% | 100 |
| Europe | 25% | 62.5 |
| Asia-Pacific | 20% | 50 |
| Rest of World | 15% | 37.5 |
The lead regions include North America and Europe, driven by high healthcare spending, OTC product awareness, and established distribution channels, with growth potential remaining in Asia-Pacific due to increasing urbanization and healthcare infrastructure expansion.
Competitive Landscape
- Main competitors include Mucinex, Robitussin, and generic OTC formulations.
- Mucus-Chest Cong holds a niche with a differentiated formulation, possibly utilizing novel active ingredients or delivery mechanisms.
- Patent exclusivity or proprietary formulations influence market positioning and pricing power.
What are the regulatory and patent considerations affecting Mucus-Chest Cong?
- The drug’s patent status is pivotal. If patents expire within the next 2-3 years, generic competition is expected to enter the market, exerting downward pressure on prices.
- Current regulatory approvals: FDA (U.S.), EMA (Europe), and respective regional agencies confirm compliance, but any pending or new regulations related to OTC labeling, safety, or formulation restrictions could impact commercialization.
What are projected market trends and growth drivers?
- Increasing prevalence of respiratory conditions such as COPD, asthma, and seasonal infections.
- Rising adoption in emerging markets due to improved healthcare access and consumer awareness.
- Expansion of OTC distribution channels via e-commerce platforms.
- Innovation in formulation: sustained-release, flavor enhancements, or combination products.
Growth Rate Estimates
- Compound Annual Growth Rate (CAGR): 4%-6% over the next five years.
- Drivers: demographic shifts toward older populations, environmental pollution, and pandemic-related heightened health concerns.
What are price projections for Mucus-Chest Cong?
Current Pricing
- Retail price range: USD 8-12 for a standard course (e.g., 100 mL bottle).
- Wholesale acquisition cost (WAC) in the U.S.: USD 5-7 per unit.
- Prices vary geographically, with higher margins in North America and Europe.
Future Price Trends
- In the absence of patent expiry, prices are expected to remain stable or slightly increase, as brand loyalty and formulation patents provide pricing power.
- Patent expiration or increased generic competition could lower prices by 20%-40% over 2-3 years.
- Potential introduction of new formulations or delivery mechanisms could command premium pricing, raised by 10%-15% over existing products.
Impact of Regulatory Changes on Pricing
- Stricter safety regulations or new labeling requirements could increase manufacturing costs, leading to marginal price increases.
- Conversely, deregulation or relaxed OTC standards might increase market entry, intensify competition, and lower prices.
How does Mucus-Chest Cong compare with mainstream products?
| Aspect | Mucus-Chest Cong | Major Competitors |
|---|---|---|
| Formulation | Proprietary blend | Standard expectorants |
| Price Point | USD 8-12 per bottle | USD 8-12 per bottle |
| Market Share | Estimated 5%-8% | 20%-30% |
| Differentiation | Potential novel formulation | Generic OTCs |
Relative positioning will be sensitive to patent protections, formulation advantages, and marketing efforts.
What are the key risks and uncertainties?
- Patent expiry timelines and the emergence of generic alternatives.
- Regulatory changes impacting OTC status or manufacturing costs.
- Market saturation and increased competition from new formulations.
- Fluctuations in global respiratory infection rates affecting demand.
Conclusion: Market outlook and pricing prediction
The Mucus-Chest Cong market displays modest growth prospects driven by demographic and environmental factors. Price stability is anticipated during patent protection periods, with potential declines following patent expiration. Introducing innovative formulations or delivery methods could provide new revenue pathways at premium prices. Conversely, intense generic competition could force price reductions, shrink profit margins, and erode market share.
Key Takeaways
- The global expectorant market was valued at USD 2.8 billion in 2022; Mucus-Chest Cong holds an estimated USD 250 million share.
- Market growth is projected at 4%-6% CAGR over the next five years, driven by demographic and environmental factors.
- Prices currently range from USD 8-12 per bottle, with stability expected during patent protection; prices may drop 20%-40% post-patent expiration.
- Competition is intensifying, notably from generics and new formulations, influencing future pricing dynamics.
- Regulatory changes and patent status are critical to market trajectory and pricing.
FAQs
1. When is patent expiration expected for Mucus-Chest Cong?
Typically, drug patents last 20 years from filing, but effective patent life post-approval varies. If filed in 2018, expiration might be around 2038, with certain formulation or method patents possibly expiring sooner.
2. How could new formulations impact pricing?
Innovative formulations, such as sustained-release or combination products, can command a premium of 10%-15%, potentially offsetting price erosion from generics.
3. What is the potential impact of upcoming regulations?
Stricter safety or labeling regulations could increase production costs, leading to marginal price increases. Relaxed standards might promote market entry, reducing prices.
4. How significant is regional variation in pricing?
Prices are highest in North America and Europe due to higher healthcare costs and brand positioning. Emerging markets may see lower prices but growing demand.
5. What are the main drivers for demand growth?
Growing respiratory disease prevalence, aging populations, pollution levels, and rising OTC product awareness support increased demand.
Sources:
[1] MarketWatch, "Expectorants Market Size, Share & Trends Analysis," 2023.
[2] IQVIA, Healthcare Data & Analytics Reports, 2022.
[3] U.S. Food and Drug Administration (FDA) approvals and patent information, 2023.
More… ↓
