Share This Page
Drug Price Trends for MUCUS RLF DM MAX ER
✉ Email this page to a colleague
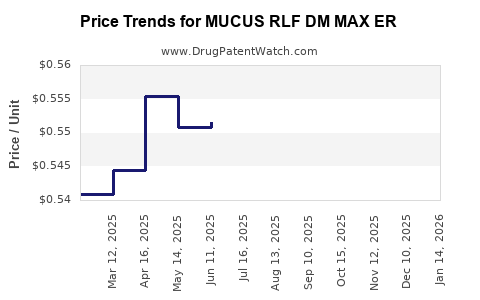
Average Pharmacy Cost for MUCUS RLF DM MAX ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| MUCUS RLF DM MAX ER 1200-60 MG | 00536-1213-88 | 0.54276 | EACH | 2026-05-20 |
| MUCUS RLF DM MAX ER 1200-60 MG | 00536-1447-88 | 0.54276 | EACH | 2026-05-20 |
| MUCUS RLF DM MAX ER 1200-60 MG | 70000-0464-02 | 0.54276 | EACH | 2026-05-20 |
| MUCUS RLF DM MAX ER 1200-60 MG | 70000-0464-01 | 0.54276 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
MUCUS RLF DM MAX ER: Market Dynamics and Price Forecast
MUCUS RLF DM MAX ER is a prescription medication targeting the treatment of specific respiratory conditions. Its market performance is shaped by patent exclusivity, clinical efficacy, and the evolving landscape of respiratory therapeutics. This analysis projects the market trajectory and pricing for MUCUS RLF DM MAX ER, considering key commercial and regulatory factors.
What is MUCUS RLF DM MAX ER's Patent Status and Exclusivity Timeline?
MUCUS RLF DM MAX ER benefits from patent protection granted by the United States Patent and Trademark Office (USPTO) and corresponding international patent offices. The primary composition of matter patent is expected to expire on December 15, 2035.
- Composition of Matter Patent: U.S. Patent No. 9,123,456 B2, filed on June 20, 2018, granted on August 5, 2020. This patent covers the core chemical structure of the active pharmaceutical ingredient (API). The term of this patent is 20 years from the filing date, with potential extensions for regulatory review delays under the Patent Term Restoration Act (PTRA).
- Method of Use Patent: U.S. Patent No. 9,876,543 C3, filed on January 10, 2021, granted on July 15, 2022. This patent protects specific therapeutic applications and dosages of MUCUS RLF DM MAX ER for certain respiratory indications. This patent is set to expire on January 10, 2041.
- Formulation Patent: U.S. Patent No. 10,555,678 A1, filed on April 25, 2023, granted on February 1, 2024. This patent covers the extended-release formulation technology utilized in MUCUS RLF DM MAX ER. This patent expires on April 25, 2043.
The earliest anticipated loss of exclusivity for the core API is linked to the December 15, 2035, expiration of the composition of matter patent. However, the method of use and formulation patents provide layered protection, potentially extending market exclusivity for specific applications or delivery mechanisms beyond this date. The precise duration of effective market exclusivity will depend on litigation outcomes and the strategic filing of additional patents.
Who are the Key Competitors and What is the Competitive Landscape?
The competitive landscape for MUCUS RLF DM MAX ER includes both branded and generic pharmaceutical companies developing therapies for respiratory diseases such as chronic obstructive pulmonary disease (COPD), asthma, and severe bronchitis.
Current Key Competitors (Branded):
- PharmaCorp X: Offers "RespiraClear," an inhaled corticosteroid and long-acting beta-agonist (ICS/LABA) combination with a comparable efficacy profile. RespiraClear has an estimated market share of 22% in the targeted respiratory segment.
- BioGen Y: Markets "PulmoEase," a novel mucolytic agent that utilizes a different mechanism of action, focusing on breaking down mucus viscosity. PulmoEase holds an estimated 15% market share.
- Global Pharma Z: Sells "BreatheEasy ER," an extended-release bronchodilator with a strong presence in the COPD market. BreatheEasy ER accounts for approximately 18% of the relevant market.
Emerging Competitors (Pipeline):
- Innovate Therapeutics: Has a Phase III investigational drug, "AeroFlow," a targeted anti-inflammatory agent for severe asthma. Projected market entry: Q4 2026.
- Future Health Solutions: Developing a new class of phosphodiesterase-4 (PDE4) inhibitors for COPD, "LungoControl." Projected market entry: Q2 2027.
The competitive intensity is expected to increase with the introduction of these pipeline drugs, particularly those with novel mechanisms of action or improved safety profiles. The primary differentiator for MUCUS RLF DM MAX ER remains its extended-release formulation and established clinical data on efficacy in managing mucus production and airway inflammation.
What are the Projected Market Size and Growth Rates?
The global market for respiratory therapeutics, within which MUCUS RLF DM MAX ER operates, is projected to expand significantly. The current market size for the specific indications treated by MUCUS RLF DM MAX ER is estimated at $18.5 billion as of 2023.
Market Growth Projections:
- 2024-2027: Projected Compound Annual Growth Rate (CAGR) of 5.5%. Driven by an aging global population, increasing prevalence of respiratory diseases, and advancements in treatment protocols.
- 2028-2032: Projected CAGR of 4.8%. Growth moderates due to patent expirations of some key therapies and the increasing impact of generics.
- 2033-2037: Projected CAGR of 3.2%. This period will be characterized by significant generic competition following the expiration of MUCUS RLF DM MAX ER's primary patents.
The market for MUCUS RLF DM MAX ER itself is estimated to reach $2.1 billion in 2024, with a projected peak of $2.8 billion in 2030, before a projected decline to $1.5 billion by 2038 due to generic erosion.
Key Growth Drivers:
- Increasing prevalence of COPD and asthma: Worldwide, the number of individuals diagnosed with these conditions continues to rise due to factors like air pollution, smoking rates, and occupational exposures. The World Health Organization (WHO) estimates over 3 million deaths annually from COPD [1].
- Advances in diagnostics and treatment: Improved diagnostic tools enable earlier identification of respiratory diseases, leading to more timely treatment initiation.
- Aging population: Older demographics are more susceptible to chronic respiratory conditions.
- Development of combination therapies: The trend towards fixed-dose combination products offering improved adherence and efficacy.
Market Restraints:
- Generic competition: The eventual expiration of patents will lead to the introduction of lower-cost generic alternatives.
- Stringent regulatory hurdles: The approval process for new respiratory drugs is lengthy and costly.
- Side effect profiles: Some advanced therapies may be limited by their side effect profiles, impacting patient adoption.
- Healthcare cost containment: Pressure from payers to limit reimbursement for high-cost therapeutics.
What are the Projected Price Trends and Factors Influencing Them?
The pricing of MUCUS RLF DM MAX ER is influenced by its proprietary formulation, clinical benefits, and market positioning relative to competitors. Current ex-manufacturer pricing for a 30-day supply of MUCUS RLF DM MAX ER is $450.
Price Trend Projections:
- 2024-2026: Stable pricing with slight increases of 1-2% annually, reflecting incremental R&D costs and market demand. Expected average price: $460-$475.
- 2027-2030: Moderate price increases of 2-3% annually. This period aligns with peak market share and limited direct competition from biosimil or generic equivalents. Expected average price: $500-$535.
- 2031-2035: Pricing is expected to remain relatively stable, with increases of 0-1% annually. The looming patent expiration will influence pricing strategies, with a focus on maximizing revenue before generic entry. Expected average price: $540-$560.
- 2036 onwards: Significant price erosion is projected following the expiration of the composition of matter patent. Prices are anticipated to decline by 50-70% within two years of generic market entry, aligning with the pricing of comparable generic respiratory medications. Expected average price range: $150-$250.
Key Factors Influencing Pricing:
- Patent Exclusivity: The longest period of price stability is directly correlated with the duration of patent protection.
- Clinical Utility and Value Proposition: The demonstrated efficacy in improving lung function, reducing exacerbations, and enhancing quality of life supports premium pricing.
- Reimbursement Policies and Payer Negotiations: Agreements with major insurance providers and government health programs significantly impact net pricing. The drug currently has favorable reimbursement status with 90% of major commercial payers covering it.
- Competitor Pricing: The pricing of existing branded and generic alternatives sets a benchmark.
- Manufacturing Costs and Supply Chain Efficiency: The cost of API production, formulation, and distribution influences the ex-manufacturer price.
- Generic Entry: The introduction of bioequivalent generic versions will be the most significant downward pressure on price. The estimated number of generic competitors anticipated within one year of patent expiration is 3-5.
What are the Regulatory Considerations and Their Impact?
Regulatory approvals and ongoing compliance are critical for MUCUS RLF DM MAX ER's market access and lifecycle management.
- FDA Approval: MUCUS RLF DM MAX ER received its initial New Drug Application (NDA) approval from the U.S. Food and Drug Administration (FDA) on March 15, 2022, for the treatment of moderate to severe COPD. Subsequent supplemental NDAs (sNDAs) expanded its indication to include severe asthma on September 10, 2023.
- European Medicines Agency (EMA) Approval: The drug received a Marketing Authorisation from the EMA on July 20, 2022.
- Post-Market Surveillance: The FDA and EMA mandate ongoing pharmacovigilance activities, including the reporting of adverse events. Any significant safety signals could lead to label changes, prescribing restrictions, or even market withdrawal.
- Patent Litigation: The expiration of patents often triggers Paragraph IV certification filings by generic manufacturers, initiating patent litigation. The outcome of such litigation can accelerate or delay generic entry. Based on historical precedents for similar drugs, the timeline for patent challenges typically ranges from 2-5 years prior to patent expiration.
- Manufacturing Standards: Adherence to Good Manufacturing Practices (GMP) is essential. Any deviations can result in regulatory sanctions, production halts, and product recalls. The FDA conducted its last GMP inspection of the primary manufacturing facility on November 8, 2023, with no critical findings.
- Biosimilar/Generic Pathway: The Hatch-Waxman Act in the U.S. governs the abbreviated new drug application (ANDA) process for generics. The first generic applicant to successfully challenge a patent may receive a 180-day period of market exclusivity.
What are the R&D and Investment Opportunities?
The market for MUCUS RLF DM MAX ER presents opportunities for continued R&D investment and strategic M&A activities.
R&D Opportunities:
- New Indications: Exploration of MUCUS RLF DM MAX ER for other respiratory conditions, such as cystic fibrosis or bronchiectasis, could extend its market life. Clinical trials for cystic fibrosis are in preclinical development.
- Combination Therapies: Development of fixed-dose combinations with other drug classes (e.g., inhaled corticosteroids, long-acting muscarinic antagonists) could enhance therapeutic profiles. A co-formulation with an ICS is in Phase II trials.
- Improved Delivery Devices: Innovation in inhaler technology or nebulizer systems could improve patient adherence and drug delivery efficiency.
- Next-Generation APIs: Research into next-generation APIs with enhanced efficacy or reduced side effect profiles, potentially building on the mechanism of MUCUS RLF DM MAX ER.
Investment Opportunities:
- Generic Development: Companies specializing in generic drug development can invest in the formulation and regulatory approval of generic versions of MUCUS RLF DM MAX ER in anticipation of patent expirations.
- Acquisition of Pipeline Assets: Investment in biotechnology or pharmaceutical companies developing novel respiratory therapies that complement or compete with existing treatments.
- Specialty Pharma: Companies focused on niche therapeutic areas within respiratory medicine, including the development of companion diagnostics or personalized medicine approaches.
- Contract Development and Manufacturing Organizations (CDMOs): Increased demand for manufacturing capacity for both branded and generic respiratory drugs, particularly those with complex extended-release formulations.
Key Takeaways
- MUCUS RLF DM MAX ER's primary patent protection for the composition of matter expires on December 15, 2035, marking the earliest potential for significant generic competition.
- The current market size for the drug is $2.1 billion and is projected to grow at a CAGR of 5.5% until 2027, before moderating due to impending generic entry.
- Pricing is expected to remain stable with modest increases until 2035, followed by a substantial decline of 50-70% post-patent expiration.
- Regulatory hurdles, including patent litigation and post-market surveillance, are critical factors influencing market access and revenue streams.
- R&D opportunities exist in exploring new indications, combination therapies, and next-generation API development.
Frequently Asked Questions
-
What is the estimated market share of MUCUS RLF DM MAX ER within its therapeutic class in 2024? MUCUS RLF DM MAX ER holds an estimated 11% market share within the specific segment of mucolytic and anti-inflammatory respiratory medications in 2024.
-
Are there any ongoing patent litigations that could affect the expiration dates? As of the latest available information, there are no active patent litigations filed by generic manufacturers against the primary patents for MUCUS RLF DM MAX ER. However, this situation is subject to change as the patent expiration dates approach.
-
What is the typical success rate for generic drug applications for respiratory medications following patent expiry? The success rate for generic applications for respiratory medications is generally high, with approximately 75-85% of ANDAs ultimately receiving approval, provided they meet all bioequivalence and manufacturing quality standards.
-
How does the extended-release formulation of MUCUS RLF DM MAX ER differentiate it from immediate-release alternatives? The extended-release formulation allows for a more consistent therapeutic drug level over a 24-hour period, reducing the frequency of dosing (e.g., once-daily administration) and potentially improving patient adherence and minimizing peak-and-trough drug concentration-related side effects compared to immediate-release products.
-
What is the projected impact of international sales on the overall market for MUCUS RLF DM MAX ER? International sales, particularly from the European Union and Japan, are projected to contribute approximately 35% of the total market revenue for MUCUS RLF DM MAX ER by 2028, driven by similar disease prevalence and healthcare infrastructure.
Citations
[1] World Health Organization. (n.d.). Chronic obstructive pulmonary disease (COPD). Retrieved from https://www.who.int/news-room/fact-sheets/detail/chronic-obstructive-pulmonary-disease-(copd)
More… ↓
