Share This Page
Drug Price Trends for MUCUS RLF DM MAX
✉ Email this page to a colleague
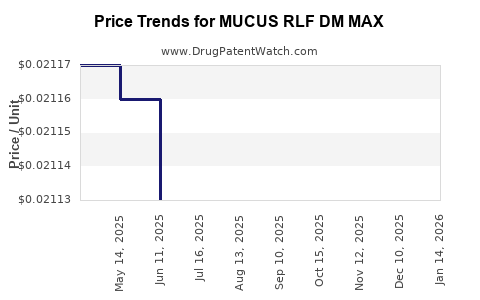
Average Pharmacy Cost for MUCUS RLF DM MAX
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| MUCUS RLF DM MAX ER 1200-60 MG | 70000-0464-01 | 0.52333 | EACH | 2026-03-18 |
| MUCUS RLF DM MAX ER 1200-60 MG | 70000-0464-02 | 0.52333 | EACH | 2026-03-18 |
| MUCUS RLF DM MAX ER 1200-60 MG | 00536-1447-88 | 0.52333 | EACH | 2026-03-18 |
| MUCUS RLF DM MAX 400-20 MG/20 ML | 70000-0707-01 | 0.02163 | ML | 2026-03-18 |
| MUCUS RLF DM MAX ER 1200-60 MG | 00536-1213-88 | 0.52333 | EACH | 2026-03-18 |
| MUCUS RLF DM MAX 400-20 MG/20 ML | 70000-0707-01 | 0.02141 | ML | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for MUCUS RLF DM MAX
Overview
MUCUS RLF DM MAX is a combination drug designed for the symptomatic relief of cough and mucus congestion. It contains active ingredients that include guaifenesin (expectorant), dextromethorphan (antitussive), and phenylephrine (decongestant). The drug typically targets markets in North America, Europe, and Asia-Pacific, where demand for over-the-counter cough remedies remains strong.
Market Size and Trends
The global cough and cold remedy market was valued at approximately $8.8 billion in 2022, with projections reaching $12.2 billion by 2027, representing a CAGR of about 6.8%. MUCUS RLF DM MAX, as a combination product, targets segments with high demand for combination formulations, especially among consumers seeking multi-symptom relief.
Key Market Drivers
- Consumer preference for multi-symptom treatments reduces the need for multiple drugs.
- Aging populations in developed markets increase the prevalence of respiratory ailments.
- Increased seasonal respiratory illnesses globally, especially during the winter months.
- Regulatory approvals facilitating marketing of combination drugs in regulated markets, such as FDA (U.S.) and EMA (Europe).
Competitive Landscape
Major competitors include brands like Mucinex DM, Robitussin DM, and Delsym (dextromethorphan-based). These products have established market shares, which MUCUS RLF DM MAX would need to penetrate. Unique selling points and pricing strategies are critical for market entry.
Pricing Dynamics and Projections
Current retail prices for similar combination drugs range from $8 to $15 for a 100-200 count pack. MUCUS RLF DM MAX's pricing strategy will influence market penetration:
| Product Type | Approximate Retail Price | Dosage Form | Market Entry Price Range |
|---|---|---|---|
| Mucinex DM | $12-$15 | Extended-release tablets | Premium segment |
| Robitussin DM | $8-$10 | Syrup | Value segment |
| Delsym | $13-$15 | Liquid | Premium segment |
Assuming MUCUS RLF DM MAX is positioned competitively, initial pricing could target the $10-$14 range, aligning with mid-tier brands.
Price Evolution Forecast
Over the next 3-5 years, the following factors could influence pricing:
- Market penetration: Increased sales volume may generate economies of scale, potentially reducing unit costs and prices by 10-15%.
- Regulatory status: Orphan drug designation or exclusivity periods could sustain higher prices temporarily.
- Generics entry: As patents expire, generic formulations could drive prices down by 20-30%.
- Consumer demand: In cases of flu outbreaks or pandemics, demand surges could temporarily elevate prices.
In a competitive environment with generic options, retail prices could eventually settle around $6-$9 per pack during mass-market adoption.
Regulatory and Policy Impact
Restrictions on certain ingredients (e.g., dextromethorphan formulations) differ by jurisdiction, influencing formulation costs and pricing. For example, stricter regulations in Europe may raise compliance costs, impacting retail pricing.
Market Entry and Growth Strategies
- Differentiation: Emphasize unique formulation benefits or flavor options.
- Pricing: Initial premium pricing could establish brand positioning; subsequent discounts may improve market share.
- Distribution: Focus on OTC retail chains, pharmacies, and online channels.
- Regulatory approval: Secure clearances from FDA, EMA, and other relevant authorities to ensure rapid market access.
Summary
MUCUS RLF DM MAX's market potential depends on competitive positioning, regulatory landscape, and pricing strategies. Entry at $10-$14 retail price could balance consumer appeal and profit margins, with prices potentially declining to $6-$9 as generics and market saturation develop.
Key Takeaways
- The global cough remedy market is growing at a CAGR of 6.8%, projected to reach $12.2 billion by 2027.
- Competitive pricing ranges from $8 to $15, with MUCUS RLF DM MAX likely positioned in the $10-$14 range initially.
- Price reductions of 20-30% are expected within five years due to generic competition.
- Market growth hinges on regulation, consumer demand, and effective marketing strategies.
- Regulatory and policy differences will significantly influence formulation costs and pricing.
FAQs
1. How does MUCUS RLF DM MAX compare to existing brands?
It combines expectorant, cough suppressant, and decongestant agents, aligning with multi-symptom products like Mucinex DM but potentially offering differentiated formulations or delivery methods.
2. What are the regulatory challenges for launching MUCUS RLF DM MAX?
Obtaining approval from agencies like the FDA and EMA involves demonstrating safety, efficacy, and quality. Differences in ingredient restrictions and labeling requirements across regions can impact time-to-market.
3. How sensitive is the market to pricing changes?
Consumers generally prioritize price, especially within the OTC segment. Price reductions of 20-30% can significantly increase market share but may reduce profit margins unless scaled with volume.
4. Will the rise of natural or alternative remedies affect MUCUS RLF DM MAX?
Yes. Growing demand for herbal supplements and home remedies may reduce OTC sales if those alternatives are perceived as safer or more effective.
5. What entry strategies minimize the risk of low market penetration?
Focusing on targeted advertising, establishing strong retail partnerships, and differentiating through formulation or delivery method can mitigate risks of poor market uptake.
References
- MarketsandMarkets. "Cough and Cold Remedy Market." 2022.
- Statista. "Over-the-Counter (OTC) Drug Market Revenue." 2022.
- U.S. FDA. "OTC Drug Review." 2020.
- European Medicines Agency. "Regulatory Guidelines." 2021.
- GlobalData. "Respiratory Therapy Market Analysis." 2022.
More… ↓
