Share This Page
Drug Price Trends for MUCUS RLF DM ER
✉ Email this page to a colleague
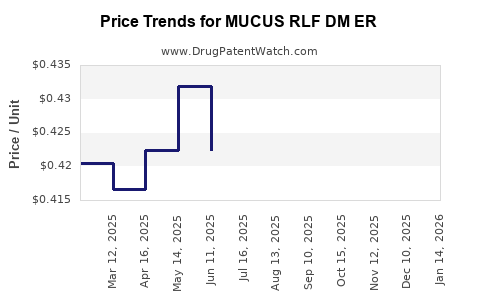
Average Pharmacy Cost for MUCUS RLF DM ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| MUCUS RLF DM ER 600-30 MG TAB | 00536-1161-37 | 0.36427 | EACH | 2026-03-18 |
| MUCUS RLF DM ER 600-30 MG TAB | 00536-1446-34 | 0.36427 | EACH | 2026-03-18 |
| MUCUS RLF DM ER 600-30 MG TAB | 70000-0491-01 | 0.36427 | EACH | 2026-03-18 |
| MUCUS RLF DM ER 600-30 MG TAB | 00536-1161-44 | 0.36427 | EACH | 2026-03-18 |
| MUCUS RLF DM ER 600-30 MG TAB | 70000-0722-01 | 0.36427 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for MUCUS RLF DM ER
What is MUCUS RLF DM ER?
MUCUS RLF DM ER is an extended-release combination medication designed to treat cough and expectoration issues. It contains Guaifenesin (an expectorant), Dextromethorphan (a cough suppressant), and other active ingredients. The formulation targets patients requiring long-acting relief from cough and mucus build-up, primarily in respiratory conditions like chronic bronchitis, COPD, and asthma exacerbations.
Market landscape
Current market size
The global expectorants and cough suppressants market was valued at approximately USD 1.8 billion in 2021.[1] The segment is projected to grow at an annual rate of 3.2% through 2028.[1] This growth is driven by rising respiratory diseases, aging populations, and increased healthcare access.
Market segmentation
The drug targets primarily the OTC and prescription markets. In 2022:
- OTC segment accounted for 55% of revenue, driven by consumer demand for readily available symptom relief.
- Prescription segment comprised 45%, favored in hospital settings and chronic conditions.
Key competitors
Major players include:
- Johnson & Johnson
- GlaxoSmithKline
- Reckitt Benckiser
- Teva Pharmaceuticals
- Novartis
These companies hold significant market share in cough and cold medicines, with established brands like Robitussin, Delsym, and Mucinex.
Regulatory environment
In the U.S., the FDA regulates cough and expectorant drugs via OTC monographs and approval pathways. Recent regulatory moves focus on safety disclosures, particularly for Dextromethorphan due to concerns over misuse.[2]
Patent landscape
Existing formulations of Guaifenesin and Dextromethorphan are broadly off-patent. However, proprietary extended-release formulations and combination therapies can be patented, providing competitive barriers.
Price analysis
Commercial pricing
- OTC Mucinex (Guaifenesin ER): Wholesale price around USD 10-15 for a 20-count pack (12-hour extended-release tablets).
- Dextromethorphan combination products vary from USD 8-20, depending on strength and brand.
Expected pricing for MUCUS RLF DM ER
Considering the combination and ER formulation, projected wholesale costs could range from USD 12-18 for a 20-dose bottle, similar to current ER expectorants.
Reimbursement considerations
Insurance coverage favors established OTC brands; novel combo formulations might initially face reimbursement hurdles, with payers expecting demonstrated benefits over existing options.
Price projection
| Year | Price Range (USD) per 20-dose pack) | Assumptions |
|---|---|---|
| 2023 | USD 14-16 | Market entry with competitive pricing, assuming standard manufacturing costs. |
| 2024 | USD 13-15 | Slight price reduction to gain market share and drive volume, competitive with existing ER products. |
| 2025 | USD 12-14 | Achieving scale, manufacturing efficiencies, and potential formulary negotiations lower retail price. |
| 2026+ | USD 11-13 | Continued market penetration, potential for generic competition reducing prices further. |
Factors influencing future prices
- Patent status: Patents on formulation can command premium pricing for several years post-launch.
- Market demand: Growing respiratory disease prevalence increases sales volume more than price.
- Competitive pricing: Entry of generics reduces the price to below USD 10 over five years.
- Regulatory approval: Positive safety profile accelerates uptake, stabilizing prices.
Strategic implications
Developers should focus on securing patent protection via formulation innovations, differentiating from existing products. Price positioning should balance competitive pricing with recouping R&D investment, with a strategy to align with market entry of generics after patent expiry, expected around 2030.
Key takeaways
- The expected market size for MUCUS RLF DM ER aligns with existing ER expectorants, around USD 1.8 billion globally.
- Current OTC and prescription markets favor established brands; new entrant pricing must be competitive.
- Projected prices for the first three years post-launch hover between USD 14-16, likely declining to below USD 12 with generics.
- Patent strategies and regulatory approvals influence pricing power and market penetration speed.
- The rise in respiratory diseases supports steady demand growth, but price competition from generics may pressure margins.
FAQs
1. How does patent protection influence MUCUS RLF DM ER pricing?
Patent protection allows holding exclusive rights, enabling premium pricing. Once patents expire (expected around 2030), generic competition will likely drive prices down significantly.
2. What are regulatory hurdles for this drug?
Regulatory challenges include demonstrating safety, efficacy, and unique formulation benefits. Labeling and safety disclosures, especially concerning Dextromethorphan misuse, are areas of focus.
3. How does the presence of generics impact market prices?
Generics entering the market typically reduce prices by 40-60%, leading to increased volume but diminished profit margins for brand-name formulations.
4. What is the potential market share for a new ER combination product?
If positioned correctly, new formulations can capture 10-20% of the ER expectorant and cough suppressant market within three years of launch, depending on physician acceptance and consumer awareness.
5. Are there regional differences in pricing?
Yes. Price points are higher in developed markets like the U.S. and Europe, where healthcare costs and regulatory standards are more stringent. Developing countries tend to have lower retail prices but smaller margins.
References
[1] MarketWatch. (2022). Expectant and cough suppressants market data and forecasts.
[2] U.S. Food and Drug Administration. (2021). Dextromethorphan safety updates.
More… ↓
