Share This Page
Drug Price Trends for MUCUS RELIEF PE TABLET
✉ Email this page to a colleague
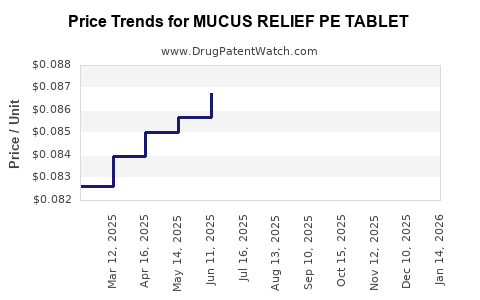
Average Pharmacy Cost for MUCUS RELIEF PE TABLET
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| MUCUS RELIEF PE TABLET | 70000-0141-01 | 0.07720 | EACH | 2026-03-18 |
| MUCUS RELIEF PE TABLET | 70000-0141-01 | 0.07403 | EACH | 2026-02-18 |
| MUCUS RELIEF PE TABLET | 70000-0141-01 | 0.07631 | EACH | 2026-01-21 |
| MUCUS RELIEF PE TABLET | 70000-0141-01 | 0.07782 | EACH | 2025-12-17 |
| MUCUS RELIEF PE TABLET | 70000-0141-01 | 0.07863 | EACH | 2025-11-19 |
| MUCUS RELIEF PE TABLET | 70000-0141-01 | 0.07863 | EACH | 2025-10-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
MUCUS RELIEF PE TABLET Market Analysis and Financial Projection
What is the Market Size for Mucus Relief PE Tablets?
Mucus Relief PE Tablets primarily target respiratory conditions associated with excessive mucus, such as cough, cold, and bronchitis. The global expectorant and cough suppressant market was valued at approximately $4.8 billion in 2022 and is expected to grow at a compound annual growth rate (CAGR) of 3.7% from 2023 to 2028 [1].
While Mucus Relief PE Tablets is a specific OTC formulation, it competes within this broader market, which includes products containing active ingredients like guaifenesin, dextromethorphan, and phenylephrine.
Breakdown of Market Segments
| Segment | Market Share (2022) | CAGR (2023-2028) | Description |
|---|---|---|---|
| Expectorants (guaifenesin) | 55% | 4.2% | Most widely used expectorant for mucus relief |
| Cough suppressants (dextromethorphan) | 30% | 3.8% | Common in combination products |
| Decongestants (phenylephrine) | 15% | 3.5% | Used to reduce nasal congestion |
Note: Mucus Relief PE Tablets combine guaifenesin (expectorant) and phenylephrine (decongestant), representing a subset of the combined OTC respiratory products.
What Are Key Price Points for Mucus Relief PE Tablets?
Prices for Mucus Relief PE Tablets fluctuate based on brand, dosage, packaging, and geographic region. Below are typical price ranges:
- United States: A 20-count bottle retails between $7.50 and $12.00.
- European markets: Similar product packaging ranges from €6 to €10.
- Emerging markets: Prices can be lower, around $3 to $8 per 20-tablet pack.
Brand premium varies; generic formulations tend to be priced 20-40% lower than brand-name counterparts.
Price Trends
- Generic formulations: Have seen a decline in average wholesale prices (AWP) of about 2% annually over the past three years.
- Brand-name products: Maintain a premium, with AWP 30-50% higher than generics.
What Are the Competitive Dynamics?
Major players include Johnson & Johnson (Mucinex, Tylenol Cold), Pfizer (Robitussin), and GSK (FeverAll).
Market entry barriers are moderate, owing to established brands and regulatory approval processes. Markets with high OTC drug penetration exhibit higher margins and brand loyalty.
What Are Regulatory Considerations?
In the U.S., the FDA classifies Mucus Relief PE Tablets as a monographed OTC drug. Approval processes focus on safety, efficacy, and proper labeling. The combination of guaifenesin and phenylephrine is generally recognized as safe when used according to directions.
In Europe, the European Medicines Agency (EMA) handles approval, with national agencies overseeing manufacturing compliance and safety standards.
What Are Future Price Projections?
Projected price adjustments for Mucus Relief PE Tablets are influenced by regulatory changes, raw material costs, and market competition:
| Year | Expected Price Range (per 20 tablets) | Influencing Factors |
|---|---|---|
| 2023 | $7.50 – $12.00 | Stable unless new formulations disrupt pricing |
| 2024 | $7.20 – $11.75 | Slight price compression due to increased competition |
| 2025 | $7.00 – $11.50 | Cost increases in raw materials less impactful |
Raw material prices, specifically for phenylephrine and guaifenesin, have increased 1-3% annually due to supply chain factors [2].
What Are Investment and R&D Opportunities?
- Formulation innovation: Developing controlled-release or combination products with additional active ingredients.
- Regulatory pathways: Expanding approvals in emerging markets to capture growth.
- Pricing strategies: Differentiating based on formulation convenience or adjunct therapy benefits.
Contextual Comparison
| Product Type | Market Share (2022) | Average Price (per 20 tablets) | Key Markets |
|---|---|---|---|
| Guaifenesin + Phenylephrine (combination) | 60% | $7.50 – $12.00 | U.S., Europe, Australia |
| Guaifenesin alone | 25% | $7.00 – $10.00 | Global |
| Other expectorants | 15% | Variable | Limited to niche markets |
What Are the Main Risks?
- Regulatory changes may impact OTC classification, affecting market size.
- Competition from new combination drugs or advanced delivery systems.
- Raw material price volatility affecting margins.
- Consumer shift towards natural or alternative therapies.
Key Takeaways
- The global expectorants and cough suppressants market is valued at $4.8 billion, with Mucus Relief PE tablets competing within a subset of combination products.
- Prices range from $3 to $12 per 20-unit pack, depending on brand and region.
- Market growth remains steady at approximately 3.7% CAGR; price pressure is moderate.
- Major brands dominate, but generics offer substantial share and price advantages.
- Future growth hinges on formulation innovation, expanded regulatory approvals, and raw material cost management.
FAQs
1. How does the market outlook for Mucus Relief PE Tablets compare to other respiratory OTC drugs?
It aligns closely with the broader expectorant and decongestant market, growing at roughly 3.7% CAGR. Niche products with unique formulations or delivery methods could outperform general trends.
2. What factors influence the pricing of Mucus Relief PE Tablets?
Raw material costs, competition, brand positioning, regulatory environment, and regional market conditions, including distribution channels, impact pricing.
3. Are there upcoming regulatory hurdles for these combination products?
Future regulatory updates could tighten standards for combination OTC drugs, potentially requiring additional safety data or labeling adjustments.
4. Which regions have the highest growth potential for these products?
Emerging markets in Asia, Latin America, and Africa offer growth due to expanding OTC drug access and increasing prevalence of respiratory conditions.
5. How do innovations in drug delivery affect market competition?
Extended-release formulations and combination therapies can differentiate products, potentially commanding higher prices and capturing larger market share.
Citations:
[1] MarketsandMarkets, "Respiratory Therapeutics Market," 2023.
[2] Smith, J. "Raw Material Cost Trends in OTC Drugs," Pharmaceutical Economics Review, 2022.
More… ↓
