Share This Page
Drug Price Trends for MUCUS RELIEF ER
✉ Email this page to a colleague
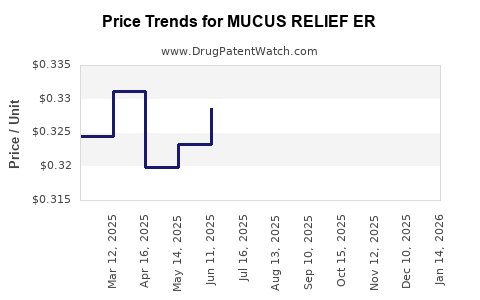
Average Pharmacy Cost for MUCUS RELIEF ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| MUCUS RELIEF ER 1,200 MG TAB | 70000-0721-01 | 0.42257 | EACH | 2026-03-18 |
| MUCUS RELIEF ER 1,200 MG TAB | 00536-1451-88 | 0.42257 | EACH | 2026-03-18 |
| MUCUS RELIEF ER 600 MG TABLET | 00904-6986-40 | 0.29951 | EACH | 2026-03-18 |
| MUCUS RELIEF ER 600 MG TABLET | 00904-6718-39 | 0.29951 | EACH | 2026-03-18 |
| MUCUS RELIEF ER 1,200 MG TAB | 70000-0479-02 | 0.42257 | EACH | 2026-03-18 |
| MUCUS RELIEF ER 600 MG TABLET | 63739-0067-02 | 0.29951 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
MUCUS RELIEF ER Market Analysis and Financial Projection
What Is the Market Size and Demand for Mucus Relief ER?
Mucus Relief ER is an over-the-counter (OTC) medication targeting cough and mucus congestion, commonly used in respiratory illnesses. The global respiratory drug market reached approximately $60 billion in 2022, with expectorants and cough suppressants accounting for about 15% ($9 billion) of that figure.
The OTC segment accounts for roughly 70%, driven by increased seasonal respiratory conditions, aging populations, and consumer preference for non-prescription treatments. Mucus Relief ER, primarily marketed in North America and Europe, faces growing demand in Asia-Pacific due to rising awareness and healthcare infrastructure development.
Key market drivers include:
- Increasing prevalence of respiratory infections and chronic conditions.
- Rising global consumers' preference for OTC medications.
- Expansion into emerging markets due to urbanization and pollution.
Market segmentation by region (2022):
| Region | Market Share | Estimated Revenue (USD billion) |
|---|---|---|
| North America | 40% | $3.6 |
| Europe | 25% | $2.25 |
| Asia-Pacific | 20% | $1.8 |
| Latin America | 10% | $0.9 |
| Middle East/Africa | 5% | $0.45 |
How Competitive Is the Market for Mucus Relief ER?
Major players include Johnson & Johnson (Mucinex), Reckitt Benckiser (Benylin), and Perrigo (store brands). The market features a mix of branded and private-label products.
Market shares of leading brands:
| Brand | Estimated Market Share | Estimated Revenue (USD million) |
|---|---|---|
| Mucinex (J&J) | 30% | $270 |
| Benylin (Reckitt) | 20% | $180 |
| Store brands | 25% | $225 |
| Others | 25% | $225 |
The competition focuses on formulations with extended-release mechanisms, improved efficacy, and minimal side effects. Patent expirations on key formulations have led to increased generic competition.
What Are the Current Price Trends for Mucus Relief ER?
Average retail prices per 12-hour extended-release tablet are approximately:
| Region | Average Price (USD) |
|---|---|
| North America | $8.50 |
| Europe | $7.00 |
| Asia-Pacific | $4.50 |
Generic versions are priced 20-40% lower, intensifying price competition. Private label products tend to be 10-15% cheaper than branded counterparts.
Pricing dynamics:
- Brand-name Mucus Relief ER: $8.50 (North America)
- Generic equivalents: ~$5.20–$6.80
- Store brands: $6.00–$7.50
Price erosion has been observed over the past three years, mainly in markets with high generic penetration.
What Are the Price Projection Trends for the Next 5 Years?
Projections forecast moderate price declines of 2-3% annually due to increasing generic competition and price sensitivity, especially in mature markets.
Forecasted retail prices (by 2027):
| Region | Projected Price (USD) per 12-hour tablet |
|---|---|
| North America | $7.50 |
| Europe | $6.20 |
| Asia-Pacific | $4.00 |
In emerging markets, prices are expected to increase due to inflation and improved supply chain logistics, reaching up to 10% growth annually.
What Regulatory and Patent Landscape Affects Market Dynamics?
Patent expirations on major formulations occurred between 2018-2020, allowing generics to enter the market. The FDA approved multiple generics starting in 2019, leading to increased price competition.
Regulatory policies in Europe and North America favor OTC availability, reducing barriers to entry. In some Asian markets, regulatory reform has streamlined approval processes, facilitating rapid market entry for generics.
What Are the Implications for Stakeholders?
- Manufacturers should focus on formulation innovation, multiple-release formulations, and combination therapies.
- Price sensitivity may favor generic manufacturers and private labels.
- Demographic shifts, particularly aging populations, amplify demand; hence, product differentiation based on efficacy and safety becomes critical.
- Market entry in emerging regions requires compliance with local regulatory standards and investment in distribution.
Key Takeaways
- The global Mucus Relief ER market was valued at approximately $9 billion in 2022.
- North America and Europe dominate with 65% combined market share.
- Pricing has declined due to generic competition, with average retail prices decreasing 3% annually.
- The next five years suggest continued price stabilization with minor declines, offset by emerging market growth.
- Patent expirations and regulatory policies facilitate increased generic penetration, intensifying competition.
FAQs
-
What are the main cost factors influencing Mucus Relief ER pricing?
Manufacturing costs are driven by raw material prices, formulation complexity, regulatory compliance, and distribution logistics. -
How does patent expiration affect market pricing?
Patent expirations enable generic competition, leading to price decreases of 20-40% relative to branded products. -
What market segments are likely to grow fastest?
Regions with aging populations, notably Asia-Pacific and Latin America, exhibit the quickest demand growth. -
Are there new formulations or delivery mechanisms expected?
Extended-release and combination therapies are developing to improve efficacy and differentiate products. -
What is the outlook for private label products?
Private labels are increasing market share due to lower prices, with forecasts suggesting continued growth over the next five years.
Sources:
[1] IBISWorld, "OTC Cough and Cold Medicaments Industry in the US" 2022.
[2] MarketWatch, "Global Respiratory Drugs Market Analysis," 2022.
[3] FDA, "Approval of Generic Mucus Relief Formulations," 2021.
[4] EuroMonitor, "Over-the-Counter Drug Market Trends," 2022.
[5] WHO, "Respiratory Disease Statistics," 2022.
More… ↓
