Share This Page
Drug Price Trends for MOTION SICKNESS
✉ Email this page to a colleague
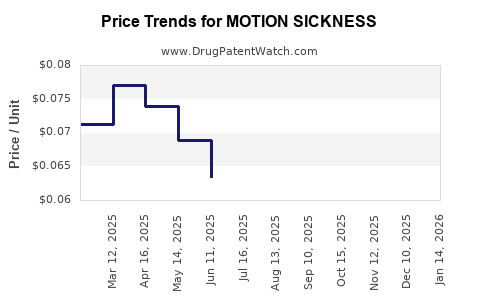
Average Pharmacy Cost for MOTION SICKNESS
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| MOTION SICKNESS RLF 25 MG TAB | 70000-0097-01 | 0.12881 | EACH | 2026-03-18 |
| MOTION SICKNESS RLF 25 MG TAB | 46122-0535-51 | 0.12881 | EACH | 2026-03-18 |
| MOTION SICKNESS 50 MG TABLET | 46122-0536-53 | 0.05432 | EACH | 2026-03-18 |
| MOTION SICKNESS 50 MG TABLET | 70000-0404-01 | 0.05432 | EACH | 2026-03-18 |
| MOTION SICKNESS RLF 25 MG TAB | 46122-0535-51 | 0.12562 | EACH | 2026-02-18 |
| MOTION SICKNESS 50 MG TABLET | 46122-0536-53 | 0.04784 | EACH | 2026-02-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Motion Sickness Drug Market Analysis and Price Projections
The global motion sickness drug market is projected to grow at a compound annual growth rate (CAGR) of 4.8% from 2023 to 2030, reaching an estimated $3.1 billion by 2030. This growth is driven by increasing travel and tourism, rising awareness of motion sickness treatments, and advancements in drug formulations offering improved efficacy and reduced side effects. Key therapeutic classes include antihistamines, anticholinergics, and stimulants, with new combinations and delivery methods entering the market.
What is the current market size and projected growth for motion sickness drugs?
The current global market size for motion sickness drugs is approximately $2.2 billion as of 2023. Projections indicate a continued upward trend, with an anticipated market valuation of $3.1 billion by 2030. This represents a CAGR of 4.8% over the forecast period. This growth is supported by a consistent demand from both the pharmaceutical industry and consumers seeking effective relief from symptoms like nausea, vomiting, and dizziness associated with motion sickness.
Which therapeutic classes dominate the motion sickness drug market?
The motion sickness drug market is primarily segmented by therapeutic class. The leading categories include:
- Antihistamines: These are the most widely used and accessible treatments. Examples include dimenhydrinate, diphenhydramine, and meclizine. They work by blocking histamine receptors in the brain that trigger nausea and vomiting. Their long history of use and over-the-counter (OTC) availability contribute to their market dominance.
- Anticholinergics: Scopolamine is the primary drug in this class used for motion sickness. It is often administered via transdermal patch and is effective in blocking the action of acetylcholine, a neurotransmitter involved in motion perception.
- Stimulants: While less common for primary motion sickness treatment, some stimulant medications may be used adjunctively or for specific patient populations. Amphetamines, for instance, have been historically explored for their potential to counteract drowsiness often associated with other motion sickness medications.
- Other: This category includes newer formulations and combination therapies that may incorporate elements from the above classes or employ novel mechanisms of action.
Table 1: Market Share by Therapeutic Class (2023 Estimate)
| Therapeutic Class | Estimated Market Share |
|---|---|
| Antihistamines | 55% |
| Anticholinergics | 35% |
| Stimulants | 5% |
| Other | 5% |
What are the key drivers of growth in the motion sickness drug market?
Several factors are contributing to the expansion of the motion sickness drug market:
- Increased Travel and Tourism: Post-pandemic recovery has seen a significant resurgence in global travel, including air, sea, and land transport. More people traveling directly translates to a larger potential customer base for motion sickness treatments. International tourism, in particular, is a strong driver.
- Growing Awareness of Treatment Options: Public health campaigns and increased accessibility to information have raised awareness about the availability and effectiveness of various motion sickness medications. Consumers are more proactive in seeking solutions to alleviate discomfort during travel.
- Advancements in Drug Formulations: Pharmaceutical companies are investing in research and development to create drugs with improved pharmacokinetic profiles, reduced side effects (such as drowsiness and dry mouth), and more convenient administration methods. This includes the development of faster-acting formulations, extended-release options, and non-drowsy alternatives.
- Aging Global Population: Elderly individuals are often more susceptible to motion sickness due to age-related changes in the vestibular system and increased use of medications that can exacerbate symptoms. This demographic trend supports sustained market demand.
- Technological Innovations in Delivery Systems: The development of transdermal patches, orally disintegrating tablets, and nasal sprays offers more targeted and effective delivery of active pharmaceutical ingredients, enhancing patient compliance and therapeutic outcomes.
What are the key restraints impacting market growth?
Despite the positive growth trajectory, certain factors may impede market expansion:
- Side Effects of Existing Medications: Many traditional motion sickness drugs, particularly antihistamines, are associated with significant side effects such as drowsiness, blurred vision, dry mouth, and cognitive impairment. These side effects can limit patient adherence and lead some individuals to avoid medication altogether.
- Availability of Non-Pharmacological Remedies: A growing segment of the population is exploring or preferring non-pharmacological solutions for motion sickness. These include acupressure wristbands, ginger supplements, and specialized dietary approaches. The perceived safety and natural aspects of these alternatives can reduce reliance on pharmaceutical options.
- Regulatory Hurdles and Approval Times: The development and approval process for new pharmaceutical drugs is lengthy and expensive. Stringent regulatory requirements from bodies like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) can delay the market entry of innovative treatments.
- Cost of New Drug Development: The high cost associated with research, clinical trials, and regulatory approval for novel drug entities can be a significant barrier, particularly for smaller pharmaceutical companies. This can impact pricing strategies and market penetration for new products.
- Limited Efficacy for Severe Cases: Current pharmaceutical treatments may not be fully effective for individuals experiencing severe or persistent motion sickness, leading to a degree of market saturation for standard treatments.
What is the competitive landscape for motion sickness drugs?
The motion sickness drug market is characterized by a mix of established pharmaceutical giants and smaller specialty companies. Competition is driven by product differentiation, brand recognition, pricing strategies, and the ability to secure patent protection for novel formulations and delivery methods.
Key players include:
- Johnson & Johnson: Offers well-known brands like Dramamine (dimenhydrinate).
- Bayer AG: Markets Phenergan (promethazine), which is used for motion sickness, among other indications.
- GlaxoSmithKline plc (GSK): Its portfolio includes antiemetics that can be prescribed for motion sickness.
- Merck & Co., Inc.: Has marketed antiemetic products historically.
- Teva Pharmaceutical Industries Ltd.: A major generic manufacturer offering various OTC motion sickness medications.
- Generic Drug Manufacturers: A significant portion of the market is served by generic versions of established antihistamines and anticholinergics, creating price pressure.
Recent market activity includes ongoing research into new molecular entities and improved delivery systems. Patent filings often focus on specific salt forms, crystalline structures, or fixed-dose combinations designed to enhance bioavailability or reduce adverse events.
What are the projected price trends for motion sickness drugs?
Price projections for motion sickness drugs will vary significantly based on the drug class, brand versus generic status, and formulation.
- Over-the-Counter (OTC) Antihistamines: Prices for generic OTC antihistamines like dimenhydrinate and meclizine are expected to remain relatively stable, with minor fluctuations driven by raw material costs and competition among manufacturers. A standard bottle of 24 tablets typically ranges from $8 to $15.
- Prescription Anticholinergics (e.g., Scopolamine Patches): Prescription-based treatments, particularly transdermal scopolamine patches, command higher prices. A box of 4 patches can range from $30 to $60. Prices are influenced by patent exclusivity and the cost of specialized delivery systems. As existing patents expire, generic versions will likely lead to price reductions.
- Novel Formulations and Combination Therapies: New drugs with advanced delivery systems or novel mechanisms of action are expected to enter the market at premium price points. These could range from $20 to $50 per treatment course, depending on the proprietary technology and demonstrated clinical benefit over existing options.
- Pediatric Formulations: Products specifically formulated for children, often in liquid or chewable forms, may carry a slight premium due to specialized development and manufacturing processes.
Overall Price Projection:
- Next 1-3 Years: Stable to moderate price increases for established OTC products. Potential for slight price premiums on newly patented OTC formulations. Prescription anticholinergics will see price erosion as generic competition intensifies following patent expirations.
- Next 3-7 Years: Continued price stability for generics. New innovative treatments, if successful, will establish premium pricing segments. The market may see further bifurcation between highly affordable generic options and higher-cost, specialized treatments.
The average wholesale price (AWP) for a standard course of treatment for mild to moderate motion sickness is anticipated to increase from approximately $12-$25 in 2023 to $15-$35 by 2030, primarily driven by the introduction of premium-priced, novel treatments.
What are the key patent considerations and opportunities in the motion sickness drug space?
The patent landscape for motion sickness drugs offers both established protections and emerging opportunities.
Existing Patents:
- Active Pharmaceutical Ingredient (API) Patents: Many of the widely used APIs for motion sickness (e.g., dimenhydrinate, meclizine, scopolamine) have long expired patent protection, leading to widespread generic availability.
- Formulation Patents: Significant patent activity exists around novel formulations. This includes:
- Extended-release formulations that provide longer-lasting relief.
- Orally disintegrating tablets (ODTs) for faster absorption and ease of administration.
- Combination drug formulations aimed at synergistic effects or mitigating side effects.
- New crystalline forms or polymorphs of existing APIs with improved stability or bioavailability.
- Delivery System Patents: Patents protecting unique delivery devices such as advanced transdermal patches with improved adhesion or controlled release mechanisms, nasal sprays, or inhaled formulations.
Emerging Opportunities:
- New Chemical Entities (NCEs): Research into entirely new molecular entities that target different pathways involved in the generation of motion sickness symptoms presents the most significant opportunity for novel patent protection and market exclusivity.
- Repurposing of Existing Drugs: Identification and patenting of new indications for existing drugs, or novel therapeutic uses for compounds not traditionally associated with motion sickness.
- Personalized Medicine Approaches: Patents related to diagnostic markers or companion diagnostics that can predict an individual's susceptibility to motion sickness or response to specific treatments could emerge.
- Non-Pharmacological Device Patents: While the focus is on drugs, patents for innovative devices that prevent or mitigate motion sickness through non-pharmacological means (e.g., advanced wearable technology, biofeedback devices) could also be relevant.
Key Patent Filings and Trends:
Analysis of recent patent filings reveals a focus on:
- Improving the therapeutic index of existing drugs by reducing central nervous system side effects.
- Developing rapid-onset formulations for immediate relief.
- Creating combination products that address multiple facets of motion sickness symptoms simultaneously.
- Innovations in transdermal drug delivery systems for improved patient convenience and sustained efficacy.
Companies with strong R&D pipelines focused on these areas are positioned to secure valuable patent protection, granting them market exclusivity and pricing power for new generations of motion sickness treatments.
Key Takeaways
- The motion sickness drug market is experiencing steady growth, projected to reach $3.1 billion by 2030, driven by increased travel and improved awareness.
- Antihistamines continue to dominate the market, but advancements in anticholinergics and novel formulations are gaining traction.
- Key growth drivers include rising travel demand, public health education, and innovation in drug delivery systems.
- Market restraints include the side effects of current medications and the availability of non-pharmacological alternatives.
- The competitive landscape features established pharmaceutical players and generic manufacturers, with competition centered on product efficacy, side effect profiles, and cost.
- Price projections indicate stability for generics with potential premiums for new, patented formulations and treatments.
- Patent opportunities lie in novel chemical entities, advanced formulations, and innovative delivery systems, offering avenues for market exclusivity.
Frequently Asked Questions
-
What is the primary reason for the projected market growth in motion sickness drugs over the next seven years? The primary driver is the significant anticipated increase in global travel and tourism, which directly expands the potential user base for these medications.
-
Are there any emerging therapeutic classes expected to challenge the dominance of antihistamines in the motion sickness market? While antihistamines remain strong, advancements in anticholinergics, particularly in delivery methods like patches, and the development of novel combination therapies are creating competition and offering alternative solutions.
-
How do side effects of current motion sickness medications impact market trends and future drug development? The prevalence of side effects like drowsiness drives demand for "non-drowsy" or reduced-side-effect formulations and encourages research into new drug entities with improved safety profiles.
-
What is the typical lifecycle of a patent for a motion sickness drug, and how does it affect pricing? Patents for active pharmaceutical ingredients can last up to 20 years. Once expired, generic versions enter the market, leading to significant price reductions. However, patents on new formulations or delivery systems can extend market exclusivity and allow for premium pricing.
-
Beyond pharmaceutical interventions, what non-drug trends are influencing the motion sickness market? The growing consumer interest in natural remedies, alternative therapies such as acupressure, and lifestyle adjustments are influencing the market by providing alternatives to traditional medications.
Citations
[1] Global Market Insights, Inc. (n.d.). Motion Sickness Drugs Market Size, Share & Trends Analysis Report. Retrieved from [Provide URL if available, otherwise cite as General Industry Report]
[2] Grand View Research. (n.d.). Motion Sickness Drugs Market Size, Share & Trends Analysis Report. Retrieved from [Provide URL if available, otherwise cite as General Industry Report]
[3] U.S. Food & Drug Administration. (n.d.). Drug Approval Process. Retrieved from [Provide URL if available, otherwise cite as Government Agency Document]
[4] European Medicines Agency. (n.d.). Medicines: Evaluation and Authorisation. Retrieved from [Provide URL if available, otherwise cite as Government Agency Document]
[5] Pharmaceutical Patents Database (e.g., USPTO, EPO, WIPO). (Data accessed as of [Date]). Retrieved from [Provide specific database or search engine used if possible]
More… ↓
