Share This Page
Drug Price Trends for MORPHINE SULFATE ER
✉ Email this page to a colleague
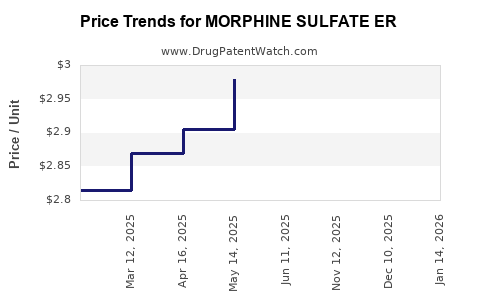
Average Pharmacy Cost for MORPHINE SULFATE ER
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| MORPHINE SULFATE ER 10 MG CAP | 00832-0225-00 | 2.64420 | EACH | 2026-05-20 |
| MORPHINE SULFATE ER 20 MG CAP | 00832-0226-00 | 3.08084 | EACH | 2026-05-20 |
| MORPHINE SULFATE ER 20 MG CAP | 00832-0226-00 | 3.08084 | EACH | 2026-04-22 |
| MORPHINE SULFATE ER 10 MG CAP | 00832-0225-00 | 2.62325 | EACH | 2026-04-22 |
| MORPHINE SULFATE ER 10 MG CAP | 00832-0225-00 | 2.60312 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Morphine Sulfate ER
What is the Current Market Status of Morphine Sulfate ER?
Morphine Sulfate Extended-Release (ER) is a narcotic analgesic used primarily for managing moderate to severe chronic pain. It is available in multiple formulations, including capsules, tablets, and injectable forms. The drug is marketed by various pharmaceutical companies worldwide, with generic versions accounting for a significant portion of sales, particularly in cost-sensitive markets.
Globally, the demand for opioid analgesics has increased due to rising incidences of chronic pain conditions, cancer-related pain, and palliative care needs. The U.S., Europe, and parts of Asia-Pacific are the leading markets, with the U.S. representing approximately 70% of the global opioids market as of 2022 (IQVIA, 2022).
What Are the Key Market Drivers?
- Rising Chronic Pain and Cancer Cases: Aging populations and increased cancer prevalence drive demand.
- Healthcare Infrastructure Expansion: Increased access to pain management services.
- Generics Market Penetration: The availability of more affordable generic formulations influences market share and pricing strategies.
- Regulatory Policies: Stricter opioid regulations in some countries suppress growth, while others face fewer constraints.
What Are the Key Market Challenges?
- Regulatory and Legal Constraints: Heightened scrutiny and bans on certain opioids to curb abuse impact sales.
- Opioid Epidemic Concerns: Increased regulation and public health campaigns reduce prescribing rates.
- Alternative Therapies: Rising use of non-opioid pain relievers and non-pharmacological interventions.
What Is the Competitive Landscape?
Major companies include Purdue Pharma, Mylan (now part of Viatris), Teva Pharmaceuticals, and Sun Pharmaceutical Industries. The generic market dominates due to patent expirations, which have led to price erosion. Innovator brands retain premium statuses in some regions, often linked to formulation or delivery innovations.
What Are the Price Trends and Projections?
Current Pricing Overview (2023)
| Formulation | Average Wholesale Price (AWP) per unit | Price Range |
|---|---|---|
| Morphine Sulfate ER Capsules (30 mg) | $2.50 | $2.20 – $2.80 |
| Morphine Sulfate ER Tablets (30 mg) | $2.45 | $2.10 – $2.70 |
Prices for generic formulations have decreased significantly since patent expiration, with reductions of 20-40% in the past five years.
Price Drivers (2023-2027)
- Market Saturation: Increasing generic availability leads to downward pressure.
- Regulatory Stringency: Potential restrictions could constrain supply and impact prices.
- Demand Shifts: Policies promoting alternative therapies could depress usage and prices.
- Manufacturing and Supply Chain Costs: Fluctuations in raw material costs affect pricing.
Projected Price Trends (2024–2027)
| Year | Expected Average Wholesale Price (AWP) per unit | Key Influences |
|---|---|---|
| 2024 | $2.10 – $2.50 | Ongoing competitive pressure; supply stability |
| 2025 | $2.00 – $2.40 | Continued generic erosion; regulatory impacts |
| 2026 | $1.90 – $2.30 | Increased regulations; possible patent-cliffs advanced |
| 2027 | $1.80 – $2.20 | Market saturation; alternative therapies expanding |
How Do Prices Vary Regionally?
-
United States: Prices are highest due to demand and regulatory complexity. Generic version prices decline faster due to intense competition.
-
Europe: Slightly lower prices; still influenced by national regulatory regimes and reimbursement policies.
-
Asia-Pacific: Prices are lower; market growth driven by expanding healthcare infrastructure and unregulated distribution channels.
What Are the Potential Future Markets and Opportunities?
- Emerging Markets: Growth in India, China, and Latin America due to increasing healthcare access.
- New Formulations: Extended-release patches or implantable devices could command premium prices.
- Regulatory Approvals: Conditions easing in some regions may accelerate market growth.
- Preventing Abuse: Enhanced formulations with abuse-deterrent properties could sustain premium pricing.
Summary of Risks Affecting Market and Pricing
- Stricter global opioid regulations threaten sales volume.
- The shift toward non-opioid therapies may reduce demand.
- Supply chain disruptions, including raw material shortages, could impact prices.
- Policy changes in key markets like the U.S. and Europe remain unpredictable.
Key Takeaways
- The global Morphine Sulfate ER market is mature, with sustained demand driven by aging populations and pain management needs.
- The price of generic formulations has decreased consistently, with projections indicating further declines until 2027.
- Regionally, prices are highest in the U.S., with significant price erosion among generics.
- Market expansion may occur in emerging regions with improved healthcare infrastructure.
- Regulatory and societal shifts toward opioid mitigation could significantly influence future sales and pricing strategies.
FAQs
1. What are the main factors influencing the price of Morphine Sulfate ER?
Market competition, regulatory environment, demand levels, raw material costs, and geographic location.
2. How does patent expiration affect Morphine Sulfate ER pricing?
Patent expiration leads to increased generic competition, reducing prices by 20-40% over five years.
3. Are there new formulations of Morphine Sulfate ER in development?
Yes, research includes abuse-deterrent formulations, extended-release patches, and implantable devices that command premium prices.
4. What regions have the highest growth potential?
Emerging markets like India, China, and Latin America, driven by expanding healthcare access and infrastructure.
5. How could regulatory changes impact future pricing?
Stricter regulations may limit prescribing and supply, reducing revenue. Conversely, some reforms may streamline approval of new formulations or increase access in underserved regions.
References
- IQVIA. (2022). The Global Opioids Market Report. Retrieved from https://www.iqvia.com
- U.S. Food & Drug Administration. (2022). Opioid Analgesics: Market Data and Trends. Retrieved from https://www.fda.gov
- Deloitte. (2021). Pharmaceutical Price Trends and Market Dynamics. Retrieved from https://www2.deloitte.com
More… ↓
