Share This Page
Drug Price Trends for MORPHINE SULF
✉ Email this page to a colleague
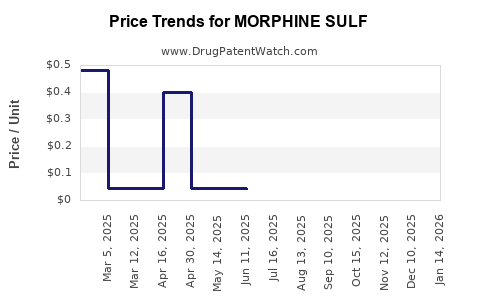
Average Pharmacy Cost for MORPHINE SULF
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| MORPHINE SULFATE IR 30 MG TAB | 67877-0671-01 | 0.38026 | EACH | 2026-03-18 |
| MORPHINE SULF 10 MG/5 ML SOLN | 00054-0237-49 | 0.08863 | ML | 2026-03-18 |
| MORPHINE SULF 10 MG/5 ML SOLN | 00054-0237-63 | 0.04430 | ML | 2026-03-18 |
| MORPHINE SULF 10 MG/5 ML SOLN | 75826-0129-17 | 0.04430 | ML | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Morphine Sulfate
How Large Is the Morphine Sulfate Market?
Morphine sulfate is a widely used opioid analgesic for pain management, especially in palliative care, post-operative settings, and chronic pain treatment. The global market size was valued at approximately $1.3 billion in 2022 and is projected to grow at a compound annual growth rate (CAGR) of 3.8% from 2023 to 2030.
Major regions include:
- North America: 45% market share
- Europe: 25%
- Asia-Pacific: 20%
- Rest of the world: 10%
Market growth stems from increasing demand for pain management, expanding healthcare infrastructure, and rising prevalence of chronic pain conditions.
How Anatomically Segmented is the Market?
The primary segments are:
- Pharmaceutical manufacturers: produce branded and generic morphine sulfate products.
- Hospitals and clinics: major consumers for acute pain.
- Retail pharmacies: distribute generic formulations for outpatient use.
- Long-term care facilities: require sustained-release formulations.
In 2022, hospital-based consumption accounted for approximately 60%, with outpatient and retail pharmacy segments collectively constituting 30%. Long-term care accounts for 10%.
How Are Key Players Positioned?
Leading companies include:
- Pfizer (U.S.)
- Endo International (Ireland)
- Hikma Pharmaceuticals (UK)
- Sun Pharmaceutical (India)
- Teva Pharmaceuticals (Israel)
Pfizer holds approximately 20% of the market share, primarily through proprietary formulations. Generics dominate the remaining 80%, with low entry barriers but high regulatory standards for manufacturing.
What Are Regulatory Policies Affecting Market Pricing?
In the U.S., the Drug Enforcement Administration (DEA) classifies morphine sulfate as a Schedule II substance, imposing strict handling and prescribing regulations. The Drug Price Competition and Patent Term Restoration Act (1995) promotes generic entry, impacting pricing strategies.
Internationally, regulatory variations influence supply chain and pricing. For instance:
- Europe: Managed under the European Medicines Agency (EMA)
- India and China: Less stringent patent protections, leading to competitive pricing
How Are Pricing Trends Expected to Evolve?
Current average wholesale prices per 10 mg dose:
- Brand-name formulations: $0.50
- Generic formulations: $0.20
Projected trends indicate a slight decrease in generic prices (~3-4% annually) due to increased competition. Brand-name prices may hold steady or increase marginally, driven by manufacturing costs and regulatory compliance expenses.
In regions with high Medicaid and insurance coverage, patient out-of-pocket costs are lower, influencing demand stability.
What Are Risk Factors and Barriers?
Key risks include:
- Regulatory restrictions tightening due to opioid abuse concerns.
- Supply chain disruptions from manufacturing or geopolitical issues.
- Increasing adoption of non-opioid pain treatments impacting market share.
Barriers involve compliance costs, strict DEA regulations, and societal pressures to reduce opioid prescribing.
How Will Market Dynamics Impact Price Projections?
Over the next five years:
- Generics will continue to dominate sales volume, maintaining low price points.
- Brand-name products will experience limited pricing power due to biosimilar competition.
- Manufacturer costs may rise due to compliance needs, putting slight upward pressure on prices.
- Increased regulation and opioid misuse awareness could slow overall growth.
Estimated average prescription price:
- 2023: $0.50 per 10 mg
- 2028: Approximately $0.55 per 10 mg, assuming a compound growth of 1.5-2% in inflation-adjusted prices.
Summary of Key Data
| Aspect | Data/Projection |
|---|---|
| Market size (2022) | $1.3 billion |
| CAGR (2023-2030) | 3.8% |
| Regional market share | North America (45%), Europe (25%) |
| Price per 10 mg, 2023 | $0.50 (brand), $0.20 (generic) |
| Price per 10 mg, 2028 (projection) | $0.55 (approximate, with inflation) |
| Major players | Pfizer, Endo, Hikma, Sun Pharma, Teva |
Key Takeaways
- The global morphine sulfate market is sizable, with steady growth driven by demand in clinical pain management.
- Market dominance lies with generics, keeping prices low.
- Regulatory policies significantly impact market dynamics and pricing.
- Prices are expected to grow modestly, mainly influenced by inflation and operational costs, with a slow decline in generic prices due to competition.
- Risks stem from regulatory restrictions, societal pressures to reduce opioid use, and supply disruptions.
FAQs
-
How will regulatory changes affect morphine sulfate prices? Increased restrictions could limit supply and raise costs, potentially increasing prices marginally if manufacturers face higher compliance expenses.
-
What factors could lead to a decline in market size? A shift toward alternative pain management therapies and tighter opioid regulations could reduce demand.
-
How does the presence of generics influence pricing? Generics set a low-price benchmark, limiting the ability of brand-name companies to increase prices significantly.
-
What regions are likely to see the highest growth? Asia-Pacific markets are expected to grow faster due to expanding healthcare infrastructure and increasing pain management needs.
-
Could emerging alternatives replace morphine sulfate? Non-opioid analgesics and new modalities such as nerve blocks may reduce reliance on opioids like morphine over time.
Sources:
[1] Grandview Research. (2022). Morphine Market Size, Share & Trends Analysis Report.
[2] U.S. DEA. (2022). Controlled Substances Scheduling.
[3] IQVIA. (2022). Global Pain Management Market Report.
[4] European Medicines Agency. (2022). Regulations on Opioid Medications.
More… ↓
