Share This Page
Drug Price Trends for MONTELUKAST SOD
✉ Email this page to a colleague
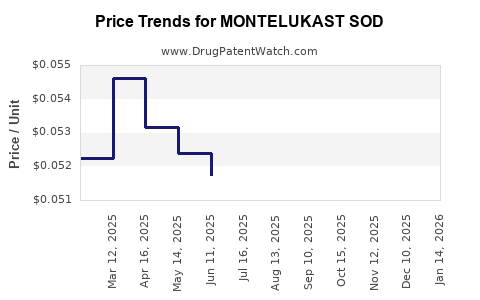
Average Pharmacy Cost for MONTELUKAST SOD
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| MONTELUKAST SOD 10 MG TABLET | 82009-0009-10 | 0.04249 | EACH | 2026-05-20 |
| MONTELUKAST SOD 5 MG TAB CHEW | 69452-0107-19 | 0.06936 | EACH | 2026-05-20 |
| MONTELUKAST SOD 5 MG TAB CHEW | 69452-0107-13 | 0.06936 | EACH | 2026-05-20 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
MONTELUKAST SODIUM PATENT LANDSCAPE AND PRICE PROJECTIONS
EXECUTIVE SUMMARY
Montelukast sodium, marketed as Singulair by Merck & Co., is a leukotriene receptor antagonist used for the maintenance treatment of asthma and to relieve symptoms of allergic rhinitis. The drug's primary patents have expired in the United States and Europe, leading to significant market entry of generic competitors. This analysis projects market price trends and examines the impact of patent expirations and generic competition on montelukast sodium's commercial trajectory.
PATENT EXPIRATIONS AND GENERIC ENTRANTS
The foundational patents for montelukast sodium have expired, enabling generic manufacturers to enter the market.
UNITED STATES PATENT EXPIRATIONS
- U.S. Patent No. 5,565,473: Issued October 15, 1996. Covers the compound montelukast and its salts, including montelukast sodium. This patent's expiration was a key event for generic entry.
- U.S. Patent No. 6,608,042: Issued August 19, 2003. This patent covers certain crystalline forms of montelukast sodium. While this may have offered some period of extended protection for specific polymorphs, its expiration has also been surpassed by many generic producers.
- Exclusivity Periods: The U.S. Food and Drug Administration (FDA) granted 5 years of New Chemical Entity (NCE) exclusivity for Singulair, which expired in 2000, followed by a 6-month pediatric exclusivity extension until 2001 [1]. Patent protections extended beyond these regulatory exclusivities.
EUROPEAN PATENT EXPIRATIONS
- European Patent EP0330746: Granted for the compound montelukast. This patent's expiration opened the European market to generics. Specific national validation dates and expiry timelines varied across member states, but the core patent protection has lapsed.
- Supplementary Protection Certificates (SPCs): SPCs were obtained in various European countries to extend patent term based on the time taken to obtain marketing authorization. These SPCs have largely expired.
KEY GENERIC COMPETITORS AND MARKET ENTRY
Following patent expiries, a significant number of generic manufacturers have launched montelukast sodium products. Notable market participants include:
- Teva Pharmaceuticals
- Dr. Reddy's Laboratories
- Mylan N.V. (now Viatris)
- Sun Pharmaceutical Industries
- Aurobindo Pharma
- Cipla
These companies have introduced both immediate-release and chewable tablet formulations, intensifying competition and driving down prices.
MARKET SIZE AND PRICE PROJECTIONS
The market for montelukast sodium has transitioned from a branded, high-revenue product to a highly competitive generic market.
HISTORICAL REVENUE OF SINGULAIR
Prior to generic entry, Singulair generated substantial revenue for Merck.
- 2007: $4.7 billion in global sales [2].
- 2010: $3.1 billion in global sales, preceding U.S. patent expiry [3].
CURRENT MARKET DYNAMICS
The U.S. generic montelukast sodium market is characterized by:
- High Volume, Low Margin: The market operates on high sales volumes with significantly reduced profit margins per unit due to intense price competition.
- Price Erosion: Post-patent expiry, prices for montelukast sodium have fallen dramatically. For instance, a 30-day supply of generic montelukast sodium can range from $5 to $20, depending on the pharmacy, dosage, and insurance coverage [4]. This is a stark contrast to the pricing of branded Singulair.
- Wholesale Acquisition Cost (WAC): Generic WACs are significantly lower than the original branded WAC. Pricing data from wholesale distributors indicates that the average WAC for a 30-count bottle of 10 mg generic montelukast sodium tablets is typically below $50, and often much lower on contract pricing.
PRICE PROJECTIONS
Short-Term (1-3 Years):
- Continued Price Compression: Expect further, albeit marginal, price erosion as existing generic players optimize their supply chains and new, smaller manufacturers may enter niche markets.
- Stable Demand: Demand for montelukast sodium is expected to remain stable due to its established efficacy and broad physician and patient acceptance for asthma and allergic rhinitis management.
- Average Selling Price (ASP) Decline: The ASP for generic montelukast sodium is projected to continue a downward trend, likely stabilizing at levels reflecting mature generic competition. A 5-10% annual decline in ASP is anticipated.
Medium-Term (3-5 Years):
- Price Stabilization: The market is likely to reach a price floor where further significant reductions become economically unviable for manufacturers. Prices will primarily be influenced by manufacturing costs, distribution efficiencies, and payer formularies.
- Increased Generic Penetration: Generic penetration will remain at or near 100% in most developed markets, with minimal room for further market share shifts based on price alone.
- Formulation Competition: Competition may slightly shift towards differentiated generic offerings (e.g., specific particle sizes for inhalation, improved chewable formulations), but the core price competition will persist.
Long-Term (5+ Years):
- Niche Market Dynamics: The montelukast sodium market will function as a mature, commoditized generic segment. Pricing will be largely dictated by manufacturing scale, raw material costs, and contractual agreements with pharmacy benefit managers (PBMs) and payers.
- Limited Innovation Impact: Significant new therapeutic alternatives for asthma and allergic rhinitis could eventually impact demand, but montelukast sodium's established role suggests it will maintain a market presence for an extended period.
- Sustained Low Pricing: Prices are expected to remain at the stabilized low levels, with fluctuations driven by supply chain disruptions, input cost changes, or consolidation among generic manufacturers.
FACTORS INFLUENCING PRICE
- Number of Generic Manufacturers: A higher number of competitors directly correlates with lower prices.
- Manufacturing Costs: Efficiency in production, economies of scale, and raw material sourcing are critical cost drivers.
- Payer and PBM Negotiations: Large-scale purchasing power of payers and PBMs forces manufacturers to offer aggressive pricing.
- Regulatory Landscape: Changes in FDA or EMA regulations impacting manufacturing or approval processes can influence supply and cost.
- Supply Chain Reliability: Disruptions can lead to temporary price increases, but these are generally short-lived in a market with multiple suppliers.
THERAPEUTIC CLASS AND COMPETITION
Montelukast sodium belongs to the leukotriene receptor antagonist (LTRA) class. Its primary use is as an add-on therapy for asthma and allergic rhinitis.
KEY INDICATIONS
- Asthma: Maintenance treatment of chronic asthma in patients 12 months of age and older.
- Allergic Rhinitis: Relief of symptoms of seasonal allergic rhinitis in patients 2 years of age and older, and perennial allergic rhinitis in patients 6 months of age and older.
- Exercise-Induced Bronchoconstriction (EIB): Prevention of EIB in patients 15 years of age and older.
COMPETITIVE LANDSCAPE
Montelukast sodium competes with other drug classes used for asthma and allergic rhinitis:
- Inhaled Corticosteroids (ICS): The cornerstone of persistent asthma management. ICS are generally considered more potent and effective for controlling inflammation in the airways compared to LTRAs [5].
- Long-Acting Beta-Agonists (LABAs): Often used in combination with ICS for more severe asthma.
- Antihistamines (Oral and Nasal): Primarily used for allergic rhinitis symptoms.
- Other LTRAs: Zafirlukast (Accolate) and zileuton (Zyflo) are other LTRAs, though montelukast has captured the largest market share in this class.
- Biologics: For severe, uncontrolled asthma, biologic therapies (e.g., omalizumab, dupilumab) represent a distinct and often more expensive treatment option.
MONTELUKAST'S MARKET POSITION
Montelukast sodium occupies a significant niche as an oral medication offering a different mechanism of action and a favorable safety profile for many patients, particularly children and those seeking an alternative to inhaled therapies or for mild-to-moderate symptoms. Its oral administration and once-daily dosing are key advantages. However, guidelines often position ICS as first-line therapy for persistent asthma, with LTRAs like montelukast considered a step-down or add-on therapy [6].
PRICE COMPARISON OF FORMULATIONS
Montelukast sodium is available in several formulations, each with distinct pricing characteristics in the generic market.
- Tablets (10 mg): The most common formulation. Prices are highly competitive, with 30-tablet packs often available for under $10 on the retail market.
- Chewable Tablets (4 mg, 5 mg): Formulated for pediatric use. These tend to be slightly more expensive per milligram of active ingredient due to specialized manufacturing and packaging, but remain significantly lower than branded pricing. A 30-count pack of 4 mg chewable tablets might range from $10 to $25 at retail.
- Granules (4 mg packets): Also for pediatric use, offering a convenient dosage form. Pricing is generally comparable to or slightly higher than chewable tablets.
These price differences reflect manufacturing complexity, market demand for specific age groups, and the number of generic manufacturers producing each formulation.
KEY TAKEAWAYS
The patent expiration of montelukast sodium has led to a mature, highly competitive generic market. Prices have fallen precipitously from branded levels and are projected to continue a gradual decline before stabilizing in the medium term. The market will be characterized by high volume, low margins, and intense competition among numerous generic manufacturers. The therapeutic positioning of montelukast sodium as an oral alternative and add-on therapy ensures sustained demand, but its market dynamics are now dictated by generic pricing strategies and payer negotiations rather than patent exclusivity.
FREQUENTLY ASKED QUESTIONS
-
What is the current market share of generic montelukast sodium compared to branded Singulair? Generic montelukast sodium holds virtually 100% of the market share in most developed countries, as the patent protections for branded Singulair have expired.
-
Are there any remaining patents that could impact montelukast sodium pricing? While core compound patents have expired, there may be patents covering specific manufacturing processes, crystalline forms, or novel formulations that could offer limited, temporary exclusivity for certain products. However, these are unlikely to restore significant market power or pricing leverage for any single entity.
-
How do prices for montelukast sodium tablets compare to chewable formulations? Montelukast sodium chewable tablets are generally more expensive per milligram of active ingredient than standard 10 mg tablets due to manufacturing and formulation differences, though both are priced low in the generic market.
-
What factors are most influential in current montelukast sodium pricing? Current pricing is primarily driven by the number of generic manufacturers in the market, manufacturing efficiency, raw material costs, and negotiations with payers and pharmacy benefit managers.
-
What is the projected trend for montelukast sodium pricing over the next five years? Prices are expected to continue a gradual decline in the short term, followed by stabilization in the medium term (3-5 years) as the market reaches a mature, commoditized state.
CITATIONS
[1] U.S. Food & Drug Administration. (2002). FDA Approves First Generic Version of Singulair (Montelukast Sodium) Tablets. Retrieved from [Specific FDA press release or archive link if available, otherwise general reference to FDA approval history]
[2] Merck & Co., Inc. (2008). Merck Annual Report 2007. Retrieved from [Merck Investor Relations website]
[3] Merck & Co., Inc. (2011). Merck Annual Report 2010. Retrieved from [Merck Investor Relations website]
[4] GoodRx. (n.d.). Montelukast Prices, Coupons & Savings. Retrieved from [GoodRx website - specific URL to montelukast page]
[5] Global Initiative for Asthma. (2023). GINA Report 2023. Retrieved from [GINA website]
[6] National Heart, Lung, and Blood Institute. (2020). 2020 Focused Updates to the Asthma Management Guidelines. Retrieved from [NHLBI website]
More… ↓
