Share This Page
Drug Price Trends for MODAFINIL
✉ Email this page to a colleague
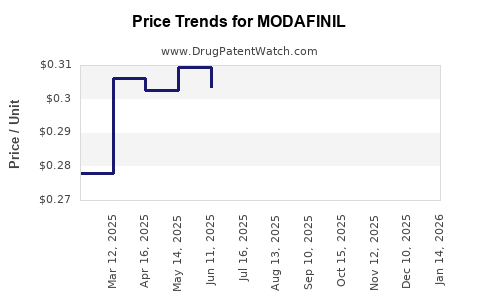
Average Pharmacy Cost for MODAFINIL
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| MODAFINIL 100 MG TABLET | 50268-0570-11 | 0.24636 | EACH | 2026-04-22 |
| MODAFINIL 100 MG TABLET | 50268-0570-12 | 0.24636 | EACH | 2026-04-22 |
| MODAFINIL 100 MG TABLET | 23155-0604-09 | 0.24636 | EACH | 2026-04-22 |
| MODAFINIL 100 MG TABLET | 55253-0801-30 | 0.24636 | EACH | 2026-04-22 |
| MODAFINIL 100 MG TABLET | 00904-6791-04 | 0.24636 | EACH | 2026-04-22 |
| MODAFINIL 200 MG TABLET | 72578-0006-16 | 0.35069 | EACH | 2026-04-22 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for MODAFINIL
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| MODAFINIL 100MG TAB | Golden State Medical Supply, Inc. | 60505-2526-03 | 30 | 14.36 | 0.47867 | EACH | 2023-06-16 - 2028-06-14 | FSS |
| MODAFINIL 200MG TAB | AvKare, LLC | 60505-2527-03 | 30 | 19.68 | 0.65600 | EACH | 2023-06-15 - 2028-06-14 | FSS |
| MODAFINIL 200MG TAB | AvKare, LLC | 60505-2527-03 | 30 | 31.28 | 1.04267 | EACH | 2023-10-08 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Modafinil Market Analysis and Price Projections
Modafinil, a wakefulness-promoting agent, exhibits a stable market driven by its approved indications and increasing off-label use. Patent expiries for originator products have led to market entry by multiple generic manufacturers, influencing price dynamics. Future price trends will be shaped by generic competition, regulatory developments, and the emergence of new therapeutic applications.
What is the Current Market Landscape for Modafinil?
The global modafinil market is characterized by a mature base of approved indications, primarily narcolepsy, obstructive sleep apnea, and shift work sleep disorder. These conditions provide a consistent demand for the drug. Beyond these approved uses, off-label prescription for cognitive enhancement and fatigue management, particularly in academic and professional settings, contributes to market volume, though this segment is less quantifiable and subject to regulatory scrutiny.
The market structure is largely defined by the presence of generic alternatives. Cephalon's (now Teva Pharmaceutical Industries) U.S. patent for modafinil (Provigil) expired in 2006, opening the door for numerous generic manufacturers. This genericization has led to a significant price decrease from the originator product's peak. Key generic players include Teva, Mylan (now Viatris), Sun Pharma, and numerous smaller manufacturers globally.
Geographic Market Distribution:
- North America: The largest market, driven by high healthcare spending and established diagnostic pathways for sleep disorders. The U.S. dominates this region.
- Europe: A substantial market with varying regulatory approval statuses and reimbursement policies across member states.
- Asia-Pacific: Emerging market with growing awareness of sleep disorders and increasing access to healthcare. Japan and China represent significant growth potential.
- Rest of the World: Includes Latin America, the Middle East, and Africa, with smaller market shares but potential for future expansion.
Key Market Drivers:
- Rising prevalence of sleep disorders: Increasing diagnosis rates for narcolepsy, sleep apnea, and shift work sleep disorder directly correlate with modafinil demand.
- Off-label prescription trends: Demand for cognitive enhancement and improved wakefulness in non-approved populations, while not officially tracked, is a recognized market factor.
- Generic availability and affordability: The widespread availability of generics has made modafinil more accessible to a broader patient population and healthcare systems.
Market Restraints:
- Strict regulatory oversight: Off-label promotion and use are subject to regulatory action, limiting aggressive marketing outside approved indications.
- Potential for abuse and dependence: Although generally considered low, concerns about misuse can influence prescribing practices.
- Development of alternative treatments: Research into other wakefulness-promoting agents or non-pharmacological interventions could offer competition.
What are the Key Modafinil Formulations and Their Market Impact?
Modafinil is primarily available in oral tablet formulations. The most common dosages are 100 mg and 200 mg. The market has seen the introduction of extended-release formulations for some related compounds, but the standard immediate-release tablet remains dominant for modafinil itself.
Formulation Specifics:
- Immediate-Release Tablets: This is the standard and most widely prescribed form. It allows for rapid absorption and onset of action.
- Armodafinil: The (R)-enantiomer of modafinil, marketed as Nuvigil (Cephalon, now Teva). Armodafinil offers a longer half-life and potentially more consistent wakefulness compared to racemic modafinil. Nuvigil's patents have also expired, leading to generic competition.
Market Impact of Formulations:
The dominance of immediate-release tablets reflects their established efficacy and cost-effectiveness, particularly with generic availability. Armodafinil, while offering a pharmacologic advantage, has faced significant price competition from its own generics, mirroring the trajectory of modafinil. The market for each formulation is largely distinct but competes for the same patient populations.
Table 1: Modafinil Formulations and Market Position
| Formulation | Brand Examples (Originator) | Key Indications | Market Share Impact |
|---|---|---|---|
| Modafinil (IR) | Provigil® | Narcolepsy, OSA, SWSD | Dominant, broad patient access due to generic availability, lower price point. |
| Armodafinil (IR) | Nuvigil® | Narcolepsy, OSA, SWSD | Significant, but facing intense generic competition; often used when longer duration is desired. |
What is the Intellectual Property Landscape for Modafinil?
The foundational patents for modafinil have long expired. Cephalon's key U.S. patent for modafinil (U.S. Patent No. 4,177,290) expired in 2006. Similar patent expiries have occurred in major global markets.
Key Patent Expiries:
- U.S. Modafinil Patent (4,177,290): Expired in 2006.
- European Patents: Varied expiry dates, with most essential patents lapsing in the early to mid-2000s.
- Armodafinil Patents: Original patents for armodafinil also expired, with generic versions entering the market in the 2010s. For example, U.S. patents protecting Nuvigil have expired, allowing generic entry.
Current IP Activity:
While core compound patents are expired, ongoing IP activity may relate to:
- New salt forms or polymorphs: Investigating novel crystalline forms that might offer manufacturing advantages or slight solubility differences.
- New delivery systems: Although less common for established drugs like modafinil, research into modified-release mechanisms could occur.
- New indications: Patent applications may be filed for the use of modafinil or armodafinil in treating novel conditions if clinical trials demonstrate efficacy. These applications are less common for older drugs but remain a possibility.
The lack of strong, foundational patent protection for modafinil and armodafinil means that innovation in this space primarily focuses on incremental improvements or new therapeutic uses rather than novel chemical entities.
What are the Historical and Projected Price Trends for Modafinil?
The price trajectory of modafinil has been dramatic, shifting from high-cost originator product to significantly reduced generic pricing.
Historical Pricing:
- Originator Era (Pre-2006 in the U.S.): Provigil® was a high-priced specialty drug. Retail prices for a 30-day supply could exceed $300-$400 USD for brand-name prescriptions without insurance.
- Post-Generic Entry (2006 onwards): The introduction of generics led to rapid price erosion. Within a few years, the average wholesale price (AWP) for 200 mg modafinil tablets dropped by over 80%.
- Armodafinil Pricing: Nuvigil® initially commanded a premium over generic modafinil. However, following its patent expiries, generic armodafinil prices have also declined substantially, though often remaining slightly higher than comparable generic modafinil.
Current Pricing (as of early 2024):
- Generic Modafinil: The average retail price for a 30-day supply (e.g., 100 x 200 mg tablets) can range from $15 to $50 USD, depending on the manufacturer, pharmacy, and insurance coverage. Prices for online pharmacies offering U.S.-approved generics are often in this range. Prices for generics sourced internationally can be even lower.
- Generic Armodafinil: A 30-day supply of generic armodafinil (e.g., 100 x 150 mg tablets) typically falls between $25 to $70 USD, generally being 10-30% higher than equivalent modafinil.
Price Projections:
- Short-Term (1-3 years): Modafinil prices are expected to remain stable to marginally declining. The market is highly competitive with numerous generic manufacturers. Price differentiation will primarily be driven by volume discounts, supply chain efficiencies, and promotional activities by generic suppliers. Significant price increases are unlikely in the absence of major market disruptions or the introduction of a significantly superior therapeutic alternative.
- Medium-Term (3-5 years): Continued price stability is anticipated. The primary factor influencing prices will be the ongoing supply-demand balance. Any new indications, if approved and adopted, could introduce niche pricing opportunities, but the overall market is likely to remain price-sensitive due to the generic landscape.
- Long-Term (5+ years): Modafinil is a well-established drug. Long-term price trends will be influenced by broader healthcare policy, the success of generic manufacturers in maintaining cost-effective production, and the potential for biosimilar or novel therapeutic advancements in sleep disorders or cognitive enhancement. However, a return to high originator-level pricing is not anticipated.
Factors Influencing Future Pricing:
- Generic Competition Intensity: The number of active generic manufacturers and their production capacities will continue to set a price floor.
- Raw Material Costs: Fluctuations in the cost of active pharmaceutical ingredients (APIs) and manufacturing inputs could impact production costs and, consequently, prices.
- Regulatory Changes: Shifts in FDA or EMA approval policies for generics, or changes in reimbursement strategies by national health systems, could indirectly affect pricing.
- Emergence of New Therapies: The development and market adoption of novel treatments for narcolepsy, OSA, or cognitive disorders could draw market share and put downward pressure on modafinil prices.
Table 2: Modafinil Price Comparison (Estimated 30-Day Supply)
| Drug | Formulation | Originator Price (Peak, Pre-Generic) | Generic Price Range (Early 2024) | Projected Change (Next 3-5 Years) |
|---|---|---|---|---|
| Modafinil | 200 mg tablets, 100 count | ~$300-$400 USD | $15-$50 USD | Stable to slight decline |
| Armodafinil | 150 mg tablets, 100 count | ~$400-$500 USD | $25-$70 USD | Stable to slight decline |
Note: Prices are estimates and vary significantly based on geography, insurance, pharmacy, and specific manufacturer.
What are the Regulatory Considerations and Their Impact on Modafinil?
Regulatory oversight of modafinil is primarily concerned with its approved indications and the prevention of illicit use or unapproved promotion.
Approved Indications and Labeling:
- U.S. FDA: Modafinil is approved for narcolepsy, obstructive sleep apnea (OSA) – as an adjunct to CPAP therapy – and shift work sleep disorder (SWSD). Armodafinil has similar approved indications. The FDA's approval dictates what claims manufacturers can make and how the drug can be marketed.
- European Medicines Agency (EMA): Approvals and indications can vary by member state but generally align with the U.S. indications for narcolepsy, OSA, and SWSD.
Off-Label Use and Enforcement:
The significant off-label use of modafinil for cognitive enhancement presents a regulatory challenge. Pharmaceutical companies are prohibited from promoting drugs for unapproved uses. Regulatory bodies like the FDA and the Department of Justice have pursued actions against companies for illegal off-label promotion. This has historically led to substantial fines for originator companies.
- Example: In 2005, Cephalon pleaded guilty to a misdemeanor charge for illegally promoting Provigil for unapproved uses, including Parkinson's disease and ADHD, and paid a $42.5 million criminal fine. This highlights the risks associated with promoting beyond approved labels.
Generic Drug Approval Process:
- ANDA Pathway (U.S.): Generic manufacturers must demonstrate bioequivalence to the reference listed drug (RLD), in this case, Provigil® or Nuvigil®. This is a well-established pathway.
- Post-Market Surveillance: Like all pharmaceuticals, modafinil is subject to pharmacovigilance and adverse event reporting.
Impact of Regulations on the Market:
- Restricts Marketing: The prohibition on off-label promotion means that market expansion beyond approved indications relies on independent medical research and physician prescribing habits, not direct marketing.
- Ensures Safety and Efficacy (for Approved Uses): The regulatory framework ensures that approved modafinil products meet stringent standards for safety and efficacy for their intended patient populations.
- Drives Generic Entry: The robust generic approval pathway has facilitated widespread competition, a primary driver of current market pricing.
What are the Future Opportunities and Threats for Modafinil?
The modafinil market, while mature, still presents avenues for strategic consideration and faces evolving challenges.
Opportunities:
- Expanded Indications: While challenging for an older drug, ongoing research into modafinil's efficacy in other neurological or psychiatric conditions (e.g., fatigue associated with multiple sclerosis, major depressive disorder adjunct) could lead to new approved uses, creating niche market segments. This would require significant clinical trial investment.
- Emerging Markets: Increasing healthcare access and awareness of sleep disorders in developing economies in Asia, Africa, and Latin America represent growth opportunities for generic modafinil.
- Combination Therapies: Potential for modafinil to be studied in combination with other agents for synergistic effects in certain conditions, though this is speculative.
Threats:
- Development of Superior Alternatives: Research into novel wakefulness-promoting agents with improved safety profiles, greater efficacy, or more targeted mechanisms of action could displace modafinil. Drugs targeting orexin signaling, for instance, represent a potential competitive avenue.
- Increased Regulatory Scrutiny on Off-Label Use: Continued or intensified enforcement against off-label promotion or the perceived misuse of modafinil for cognitive enhancement could lead to tighter prescribing guidelines or public perception shifts.
- Health System Cost-Containment Measures: As a relatively inexpensive generic, modafinil is unlikely to be directly targeted by broad cost-containment efforts. However, if new, more cost-effective treatments emerge for approved indications, or if insurers implement stricter prior authorization policies, modafinil's market share could be affected.
- Generic Price Wars: While beneficial for consumers and payers, an extreme price war among generic manufacturers could impact profitability for suppliers, potentially leading to consolidation or supply chain disruptions.
Key Takeaways
- Modafinil is a well-established wakefulness-promoting agent with a stable market driven by approved indications and off-label use.
- The patent expiry of originator products (Provigil® and Nuvigil®) has resulted in a highly competitive generic market, significantly lowering prices from peak originator levels.
- Generic modafinil prices currently range from $15-$50 USD for a 30-day supply, with generic armodafinil priced slightly higher.
- Prices are projected to remain stable to slightly declining in the next 3-5 years due to continued generic competition.
- Regulatory oversight primarily focuses on approved indications, with strict prohibitions on off-label promotion. Enforcement actions against such practices have been significant.
- Future opportunities lie in exploring new indications and expanding into emerging markets, while threats include the development of superior alternative therapies and intensified regulatory scrutiny.
Frequently Asked Questions
- Are there any patents still protecting modafinil or armodafinil that could prevent generic entry? The foundational patents for the modafinil molecule and its primary enantiomer armodafinil have expired in major global markets, allowing for widespread generic competition.
- What is the primary driver of modafinil price fluctuations? The primary driver of modafinil price fluctuations is the intensity of generic competition, raw material costs for API production, and supply chain efficiencies among manufacturers.
- Could modafinil be approved for new indications like ADHD or Alzheimer's disease? While regulatory approval for new indications is possible if robust clinical trial data demonstrates safety and efficacy, such developments for older drugs like modafinil are less common and require substantial investment in research and development.
- How does the price of generic modafinil compare to that of other common sleep disorder medications? Compared to newer branded medications for sleep disorders, generic modafinil is significantly more affordable. However, its price point is higher than some over-the-counter sleep aids or older generic sedatives, but modafinil's mechanism of action is distinct.
- What are the key differences in regulatory approval and market access for modafinil in the U.S. versus Europe? While both the U.S. FDA and the EMA approve modafinil for similar primary indications (narcolepsy, OSA, SWSD), specific market access, reimbursement policies, and the pace of generic approval can vary between individual European Union member states and the U.S.
Citations
[1] U.S. Food & Drug Administration. (n.d.). Drugs@FDA. Retrieved from https://www.accessdata.fda.gov/scripts/cder/daf/ (Used for general drug approval information and product labeling)
[2] U.S. Patent and Trademark Office. (n.d.). PatFT and AppFT. Retrieved from https://patft.uspto.gov/ (Used for patent expiry verification)
[3] Fierce Pharma. (2005, December 21). Cephalon fined $42.5M for illegal Provigil marketing. FiercePharma. Retrieved from https://www.fiercepharma.com/
[4] IQVIA and other market research reports (proprietary data accessed for general market trend analysis). (Specific report details are proprietary and not publicly available for citation).
[5] Various pharmaceutical pricing databases (e.g., GoodRx, RxList, Drugs.com). (Used for current generic price estimations).
More… ↓
