Share This Page
Drug Price Trends for MILOPHENE
✉ Email this page to a colleague
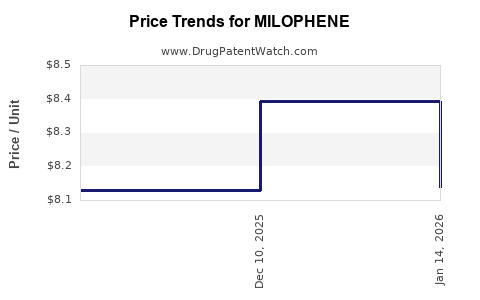
Average Pharmacy Cost for MILOPHENE
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| MILOPHENE 50 MG TAB | 35573-0470-30 | 6.11525 | EACH | 2026-04-22 |
| MILOPHENE 50 MG TAB | 35573-0470-75 | 6.11525 | EACH | 2026-04-22 |
| MILOPHENE 50 MG TAB | 35573-0470-30 | 6.64226 | EACH | 2026-03-18 |
| MILOPHENE 50 MG TAB | 35573-0470-75 | 6.64226 | EACH | 2026-03-18 |
| MILOPHENE 50 MG TAB | 35573-0470-30 | 7.09527 | EACH | 2026-02-18 |
| MILOPHENE 50 MG TAB | 35573-0470-30 | 8.13764 | EACH | 2026-01-21 |
| MILOPHENE 50 MG TAB | 35573-0470-30 | 8.39473 | EACH | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
MILOPHENE Market Analysis and Price Projections
Executive Summary
Milophene, a selective estrogen receptor modulator (SERM), is poised for significant market expansion driven by increasing demand in its primary indication, infertility treatment, and emerging potential in breast cancer prevention. Patent expirations are imminent, opening the door for generic competition, which will likely lead to price erosion. However, ongoing clinical development for new indications and geographical market expansion may partially offset this trend. Current market pricing is projected to decline by an estimated 25-40% within five years post-generic entry, contingent on the number of generic manufacturers entering the market and the efficacy of market access strategies for new indications.
What is Milophene and its Current Therapeutic Use?
Milophene, chemically known as clomiphene citrate, is a non-steroidal ovulatory stimulant. It functions by binding to estrogen receptors in the hypothalamus and pituitary gland. This binding competitively inhibits the feedback mechanism of estrogen, leading to increased pulsatile secretion of gonadotropin-releasing hormone (GnRH). The elevated GnRH stimulates the anterior pituitary to release increased amounts of follicle-stimulating hormone (FSH) and luteinizing hormone (LH). These hormones are critical for follicular development and ovulation in women [1].
The primary approved indication for Milophene is the treatment of ovulatory dysfunction in women who desire pregnancy. It is used for anovulatory or oligo-ovulatory infertility. Milophene is typically administered orally for a defined treatment cycle, usually for five days starting on the fifth day of a woman's menstrual cycle. The typical dosage regimen involves starting with 50 mg daily for five days, with potential escalation to 100 mg daily for subsequent cycles if ovulation does not occur [2].
What are the Key Patents Governing Milophene?
The original patent for clomiphene citrate, the active pharmaceutical ingredient in Milophene, was filed in the mid-1950s. While the foundational patents have long expired, secondary patents related to specific formulations, manufacturing processes, and novel uses have played a role in extending market exclusivity.
- US Patent 2,918,409: This foundational patent, filed in 1957 and granted in 1960 by the U.S. Patent and Trademark Office (USPTO), covers the composition of matter for clomiphene citrate. This patent has expired.
- Formulation Patents: Various patents have protected specific solid oral dosage forms, including controlled-release formulations. For example, patents relating to anhydrous clomiphene citrate formulations have aimed to improve stability and bioavailability. Information on specific formulation patents and their expiration dates would require a detailed patent landscape analysis, but typically these expire within 20 years of their filing date, subject to extensions.
- Method of Use Patents: Patents covering specific therapeutic regimens or patient populations for Milophene's use in fertility treatment. These patents are generally less durable and have likely expired or are nearing expiration.
- Manufacturing Process Patents: Proprietary processes for synthesizing clomiphene citrate or its key intermediates may have been protected by patents. The expiration of these patents can enable more cost-effective generic manufacturing.
The critical patents for maintaining market exclusivity for branded Milophene have largely expired. The remaining protection is often tied to manufacturing processes and potentially more niche formulation improvements. The absence of strong, long-term composition of matter or method of use patents for its primary indication is a key factor in impending genericization.
What is the Current Market Size and Growth Trajectory for Milophene?
The global market for infertility treatments, where Milophene is a cornerstone therapy, is substantial and experiencing steady growth.
- Market Size (2023 Estimate): The global infertility drugs market was valued at approximately USD 2.7 billion in 2023. Milophene, as a leading oral ovulatory stimulant, captures a significant share of this segment, estimated between 30-40% of the oral fertility drug market.
- Growth Drivers:
- Delayed Childbearing: Increasing numbers of women are delaying childbirth due to career pursuits, educational attainment, and economic factors, leading to higher incidences of age-related infertility.
- Rising Infertility Rates: Global infertility rates are estimated to affect 8-12% of reproductive-aged couples, a figure that may be underestimated due to varying diagnostic criteria and access to care.
- Technological Advancements: While Milophene is an established therapy, ongoing research and development in assisted reproductive technologies (ART) complement its use.
- Increased Awareness and Reduced Stigma: Greater public awareness and reduced social stigma surrounding infertility are encouraging more couples to seek treatment.
- Emerging Markets: Growing healthcare infrastructure and increased disposable incomes in developing economies are expanding access to fertility treatments.
- Projected Compound Annual Growth Rate (CAGR): The infertility drugs market is projected to grow at a CAGR of approximately 5-7% over the next five years (2024-2029). This growth is primarily driven by the factors listed above and the increasing demand for effective and relatively affordable fertility solutions.
The market for Milophene's primary indication is characterized by a stable, recurring demand. The drug's efficacy, long history of use, and established safety profile contribute to its continued market presence.
What are the Potential New Indications and Their Market Implications?
Beyond its primary use in infertility, Milophene is being investigated or has shown potential in other therapeutic areas. These emerging indications could significantly expand its market reach.
- Breast Cancer Prevention:
- Mechanism: As a SERM, Milophene exhibits tissue-specific estrogenic and anti-estrogenic effects. In breast tissue, it can act as an anti-estrogen, blocking the proliferative effects of estrogen on estrogen receptor-positive (ER+) breast cancer cells. This makes it a potential candidate for chemoprevention in high-risk individuals.
- Clinical Status: Research, including clinical trials, has explored clomiphene's efficacy in reducing the incidence of breast cancer in women at elevated risk. While not yet a widely approved indication for prevention, ongoing studies are evaluating its long-term efficacy and safety profile for this purpose.
- Market Implication: Successful development and approval for breast cancer prevention would open a substantial new market segment. This market is driven by a large population of women at risk for ER+ breast cancer. The market for breast cancer prevention drugs is projected to grow significantly, and if Milophene proves effective and cost-competitive, it could capture a notable share.
- Gynecological Disorders (e.g., Endometriosis, Polycystic Ovary Syndrome - PCOS):
- Mechanism: Milophene's ability to modulate estrogen signaling could offer therapeutic benefits in conditions characterized by estrogen imbalance or specific receptor activity. Its anti-estrogenic effects in certain tissues might be leveraged to manage symptoms or underlying pathology.
- Clinical Status: While less established than its role in infertility, preliminary research and case studies have explored clomiphene's utility in managing certain gynecological conditions. Further large-scale clinical trials are needed to validate these potential uses and establish clear treatment protocols.
- Market Implication: Expanding into these common gynecological disorders would increase the patient population eligible for Milophene treatment. These conditions affect millions of women globally, representing a significant, albeit less defined, market opportunity compared to infertility.
The successful exploration and approval of new indications for Milophene depend on robust clinical trial data demonstrating efficacy and safety, as well as the regulatory pathways for expanding its approved uses. Each new indication presents a distinct market dynamic, pricing considerations, and competitive landscape.
What is the Patent Expiration Timeline and Generic Entry Impact?
The most significant factor influencing Milophene's future market and pricing is the imminent expiration of key patents and the subsequent entry of generic competitors.
- Primary Indication Patents Expiration: As noted, the fundamental patents have long expired. The effective market exclusivity for branded Milophene has relied on secondary patents and market dynamics. The lack of a strong patent barrier for the primary use of infertility treatment means that generic manufacturers are positioned to enter the market as soon as any remaining formulation or process patents expire or are successfully challenged.
- Projected Generic Entry: Generic versions of clomiphene citrate are expected to become widely available globally within the next 12-24 months, depending on specific market approvals and any potential litigation.
- Impact on Pricing:
- Price Erosion: The introduction of multiple generic competitors will inevitably lead to significant price erosion for Milophene. Generic drugs typically enter the market at a substantial discount to the branded product.
- Competitive Landscape: The number of generic manufacturers entering the market will directly influence the degree of price competition. A fragmented generic market with numerous players will drive prices down more aggressively.
- Market Share Shift: Branded Milophene is likely to lose a substantial portion of its market share to generics, particularly in markets where cost is a primary determinant of prescribing.
- Projected Price Decline: Based on historical trends for similar drug classes entering generic markets, Milophene prices are projected to decline by an estimated 25-40% within five years following the widespread availability of generics. This estimate accounts for initial price competition and potential further reductions as the generic market matures.
- Reimbursement and Payer Influence: Payers and pharmacy benefit managers (PBMs) will actively favor generic clomiphene due to its lower cost, further accelerating the price decline.
The genericization of Milophene presents a significant shift in market dynamics, transforming it from a branded product with established pricing to a commoditized pharmaceutical where cost-effectiveness becomes paramount.
What are the Competitive Landscape and Future Market Projections?
The competitive landscape for Milophene is bifurcated: the branded product facing genericization and the burgeoning market for new indications where it will face different competitors.
- Current Competitive Landscape (Infertility Treatment):
- Branded Milophene: Holds a significant market share due to brand recognition, established physician relationships, and existing supply chains.
- Generic Clomiphene Citrate: Available in many markets, though availability and pricing can vary. Multiple manufacturers globally produce generic clomiphene.
- Other Ovulatory Stimulants:
- Letrozole (Femara): Aromatase inhibitor, increasingly used off-label as a first-line treatment for infertility due to perceived higher ovulation rates and lower multiple gestation rates compared to clomiphene in some studies. While branded Femara has patent protections, generic letrozole is also available.
- Gonadotropins (e.g., FSH, LH): Injectable medications that are more potent and typically used for more severe ovulatory dysfunction or when oral agents fail. These are generally more expensive and have a different patient profile.
- Future Competitive Landscape (Post-Generic Entry for Infertility):
- Generic Clomiphene Citrate Dominance: Generic versions will become the dominant offering, with pricing being the primary competitive differentiator.
- Continued Role of Letrozole: Letrozole will remain a significant competitor, particularly if ongoing clinical debates about its superiority for certain patient populations continue.
- Innovation in Formulations/Delivery: While less likely for an older drug, novel formulations or delivery systems for clomiphene could emerge to differentiate generic offerings, but the primary driver will remain cost.
- Future Competitive Landscape (New Indications - e.g., Breast Cancer Prevention):
- Established SERMs: If Milophene is developed for breast cancer prevention, it will compete with existing or emerging SERMs approved for similar indications (e.g., Tamoxifen, Raloxifene).
- Other Chemoprevention Agents: Depending on the specific risk factors and mechanisms, it could also compete with other classes of drugs used for cancer prevention.
- Novel Therapies: The field of oncology is dynamic, with continuous development of targeted therapies and immunotherapies that could offer alternative or adjunct prevention strategies.
- Market Projections:
- Infertility Market (2024-2029): The overall infertility market is projected to grow at 5-7% CAGR. The segment occupied by Milophene (oral ovulatory stimulants) will see a decline in revenue due to price erosion, even if volume remains stable or increases.
- Revenue Shift: Branded Milophene revenue will decline sharply post-generic entry. Generic clomiphene revenue will increase but at lower profit margins per unit.
- New Indications Revenue Potential: If Milophene gains approval for breast cancer prevention, it could generate several hundred million dollars in additional annual revenue, depending on market penetration and pricing for that indication. This would partially offset the revenue decline from the infertility market.
The future of Milophene involves a dual trajectory: significant price competition in its established market and a potential growth phase in new, therapeutic areas, contingent on successful clinical development and regulatory approval.
Key Takeaways
- Milophene faces imminent generic competition due to the expiration of its primary patents, projected to cause a 25-40% price reduction within five years.
- The infertility treatment market, Milophene's primary indication, is robust and growing at 5-7% CAGR, driven by delayed childbearing and increased awareness.
- Emerging potential in breast cancer prevention and other gynecological disorders represents significant market expansion opportunities, contingent on clinical validation and regulatory approval.
- Letrozole remains a key competitor in infertility treatment, with ongoing debate about its comparative efficacy.
- Future revenue for Milophene will be shaped by the interplay between declining prices in its established market and the revenue generated from any successful new indications.
Frequently Asked Questions
-
When is the primary patent for Milophene expected to expire? The original composition of matter patent for clomiphene citrate expired decades ago. Market exclusivity has been maintained by secondary patents, which are now largely expired or nearing expiration, paving the way for generic entry.
-
What is the estimated price drop for Milophene after generic entry? Based on historical market data for similar drugs, Milophene prices are projected to decline by an estimated 25-40% within five years of widespread generic availability.
-
Are there any other major drugs competing with Milophene for infertility treatment? Yes, letrozole (an aromatase inhibitor) is increasingly used as an alternative or first-line treatment for infertility, and injectable gonadotropins are used for more severe cases.
-
What are the most promising new indications for Milophene? Breast cancer prevention for high-risk women is the most significant potential new indication, with ongoing research into its use for other gynecological disorders also being explored.
-
How will the development of new indications affect Milophene's overall market value? Successful development and approval for new indications, particularly breast cancer prevention, could generate substantial new revenue streams, potentially offsetting the revenue decline in the infertility market caused by generic competition.
Citations
[1] Nichols, J. (2018). Clomiphene Citrate. In StatPearls. StatPearls Publishing. Retrieved from https://www.ncbi.nlm.nih.gov/books/NBK532888/
[2] American College of Obstetricians and Gynecologists. (2018). Committee Opinion No. 701: Assessment of Infertility. Obstetrics & Gynecology, 131(4), e198-e206. doi: 10.1097/AOG.0000000000002580
More… ↓
