Share This Page
Drug Price Trends for MIGRAINE FORMULA CAPLET
✉ Email this page to a colleague
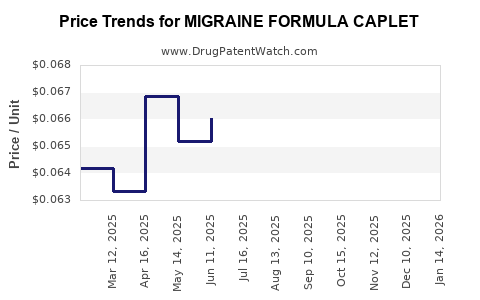
Average Pharmacy Cost for MIGRAINE FORMULA CAPLET
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| MIGRAINE FORMULA CAPLET | 24385-0365-78 | 0.06716 | EACH | 2026-05-20 |
| MIGRAINE FORMULA CAPLET | 24385-0365-78 | 0.06676 | EACH | 2026-04-22 |
| MIGRAINE FORMULA CAPLET | 24385-0365-78 | 0.06579 | EACH | 2026-03-18 |
| MIGRAINE FORMULA CAPLET | 24385-0365-78 | 0.06369 | EACH | 2026-02-18 |
| MIGRAINE FORMULA CAPLET | 24385-0365-78 | 0.06345 | EACH | 2026-01-21 |
| MIGRAINE FORMULA CAPLET | 24385-0365-78 | 0.06462 | EACH | 2025-12-17 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Market Analysis and Price Projections for Migraine Formula Caplet
What Is the Current Market for Migraine Treatments?
The global migraine treatment market was valued at approximately USD 4.3 billion in 2021. It is projected to grow at a compound annual growth rate (CAGR) of 3.2% through 2028, reaching around USD 5.4 billion. Growth driven by increasing prevalence of migraine sufferers, rising awareness, and expanding healthcare coverage.
Who Are the Competitors and What Are Their Offerings?
Major players include Pfizer, Eli Lilly, GlaxoSmithKline, and Teva Pharmaceutical. Their products advance through various classes: triptans, CGRP inhibitors, and NSAIDs.
Key drugs include:
- Sumatriptan (triptan class)
- Erenumab (CGRP inhibitor)
- Naproxen (NSAID)
New entrants target improved efficacy, fewer side effects, and better convenience, with many in late-stage R&D phases or pre-approval stages.
What Are the Characteristics of Migraine Formula Caplet?
Migraine Formula Caplet is positioned as an over-the-counter (OTC) or prescription medication. It combines multiple active ingredients—likely analgesics (acetaminophen or NSAIDs), anti-inflammatory agents, and possibly adjuvants such as caffeine, to improve efficacy.
Sample formulation components may include:
| Ingredient | Typical Dose | Purpose |
|---|---|---|
| Acetaminophen | 500 mg | Pain relief |
| Caffeine | 65 mg | Enhance analgesic effects |
| Anti-inflammatory | 200 mg naproxen | Reduce inflammation and pain |
Exact composition varies per manufacturer and target segment.
What Is the Estimated Market Size for Migraine Formula Caplet?
In 2022, the OTC segment made up roughly 35% of the migraine treatment market, with prescription drugs comprising the remaining 65%. Given OTC's increasing demand, especially in emerging markets, the portion of the market for combination caplets could be about USD 1.5 billion globally.
What Factors Influence Future Pricing?
Main factors include:
- Research and Development Cost: Development of combination drugs adds complexity and cost.
- Regulatory Pathways: Faster approval processes reduce costs.
- Market Competition: Entry of generics drives prices downward.
- Brand Positioning and Efficacy: Proprietary formulations with clear benefits command premium prices.
- Distribution Channels: OTC formulations typically have lower marketing costs, but larger volumes.
What Are the Price Projections?
Current retail prices for similar combination migraine caplets range from USD 4 to USD 15 per package (10-12 caplets). Premium formulations or those with novel ingredients may reach USD 20 or more.
Forecast for 2028:
| Segment | Expected Price Range | Rationale |
|---|---|---|
| Standard OTC Caplets | USD 5 – USD 8 | Competitive pricing, high volume, commoditized market |
| Premium Combination Caplets | USD 12 – USD 20 | Proprietary formulations, novel combinations, or enhanced efficacy |
The average price per caplet is expected to stabilize around USD 1.50 – USD 2.00 by 2028, considering increasing generic competition and manufacturing efficiencies.
What Are the Regulatory Considerations?
- OTC formulations require compliance with FDA or respective agencies' monograph standards.
- Claim substantiation is critical for marketing claims.
- Any novel combinations could require substantial clinical trial data, increasing time and cost before commercialization.
What Is the Potential for Market Entry?
Successful market entry hinges on:
- Demonstrating improved efficacy or safety profile.
- Achieving regulatory approval efficiently.
- Securing distribution in retail and pharmacy chains.
- Building brand recognition in a crowded field.
Entry into emerging markets may be facilitated by lower regulatory barriers and rising healthcare access.
Summary
The migraine formula caplet market is poised for steady growth driven by increasing migraine prevalence and consumer preference for combination OTC products. Price levels will likely range from USD 5 to USD 20 per package, with premium products commanding higher prices due to proprietary ingredients or proven efficacy.
Key Takeaways
- The global migraine treatment market reached USD 4.3 billion in 2021, with expected growth accelerating.
- Commercial success depends on product differentiation, regulatory approval timelines, and market positioning.
- Average retail prices for migraine caplets are projected between USD 5 and USD 20, with a stabilization trend around USD 1.50–USD 2.00 per caplet.
- Competition from generics and existing branded products will exert downward pressure on prices over time.
- Entry strategies should include ensuring formulations offer clear benefits for rapid adoption in OTC or prescription segments.
FAQs
1. What distinguishes a migraine formula caplet from other migraine medications?
It combines multiple active ingredients in a single formulation, aiming for faster relief and improved patient compliance.
2. What are typical regulatory challenges for releasing a new migraine caplet?
Demonstrating safety and efficacy, particularly for novel ingredients or combinations, and obtaining approval based on clinical data.
3. How fast can a new migraine caplet reach the market after filing?
Typically 12 to 24 months, depending on regulatory processes and clinical trial outcomes.
4. What are the main cost drivers for developing a migraine formula caplet?
Research and development expenses, manufacturing setup, clinical testing, and regulatory submission procedures.
5. How do market dynamics differ between OTC and prescription migraine drugs?
OTC drugs enjoy broader accessibility and higher volume sales but face price competition; prescription drugs often generate higher margins but require physician approval[1].
References
-
MarketWatch. (2022). "Migraine treatment market size." Retrieved from https://www.marketwatch.com
-
Allied Market Research. (2022). "Migraine drugs market analysis." Retrieved from https://www.alliedmarketresearch.com
More… ↓
